Abstract
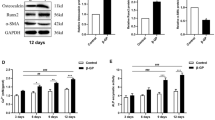
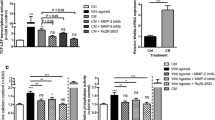
In this study, the hypothesis that Wnt/β-catenin pathway is involved in the arterial calcification by regulating the osteoprotegerin (OPG)/receptor activator of NF-κB ligand (RANKL) system was tested. The β-catenin expression was measured in the warfarin-induced calcified arteries and the osteoblast-like cells differentiating from smooth muscle cells (SMCs) by immunohistochemistry and Western blotting. The Wnt/β-catenin pathway was activated or inhibited by lithium chloride (LiCl) or dickkopf 1 (DKK1) in vitro and in vivo. Then the calcification level was determined by von Kossa staining, Ca2+ content assay, and alkaline phosphatase (ALP) activity assay. The expression levels of osteocalcin, OPG and RANKL were detected by Western blotting or real-time PCR. The results showed that in calcified arteries and OBL cells, the activation of Wnt/β-catenin pathway significantly enhanced the calcification as evidenced by increased von Kossa stains, Ca2+ contents, ALP activities, and osteocalcin expression levels (P<0.05), and it promoted the RANKL expression (P<0.05), but slightly affected the OPG expression. These results indicated that the activation of Wnt/β-catenin pathway worsens the arterial calcification, probably by promoting the RANKL expression.
Similar content being viewed by others
References
Rennenberg RJ, Schurgers LJ, Kroon AA, et al. Arterial calcifications. J Cell Mol Med, 2010,14(9):2203–2210
Valdivielso JM. Vascular calcification: types and mechanisms. Nefrologia, 2011,31(2):142–147
Tsushima M. Aortic calcification and calcium. Clin Calcium, 2010,20(11):1627–1635
Harper E, Forde H, Davenport C, et al. Vascular calcification in type–2 diabetes and cardiovascular disease: Integrative roles for OPG, RANKL and TRAIL. Vascul Pharmacol, 2016,7(82):30–40
Neven E, De Schutter TM, De Broe ME, et al. Cell biological and physicochemical aspects of arterial calcification. Kidney Int, 2011,79(11):1166–1177
Leopold JA. Vascular calcification: Mechanisms of vascular smooth muscle cell calcification. Trends Cardiovasc Med, 2015,25(4):267–274
Ogawa T, Nitta K. Pathogenesis and Management of Vascular Calcification in Patients with End–Stage Renal Disease. Contrib Nephrol, 2018,196:71–77
Bardeesi ASA, Gao J, Zhang K, et al. A novel role of cellular interactions in vascular calcification. J Transl Med, 2017,15(1):95
Nusse R, Clevers H. Wnt/ß–Catenin signaling, disease, and emerging therapeutic modalities. Cell, 2017,169(6):985–999
Kramer I, Halleux C, Keller H, et al. Osteocyte Wnt/ beta–catenin signaling is required for normal bone homeostasis. Mol Cell Biol, 2010,30(12):3071–3085
Kubota T, Michigami T, Ozono K. Wnt signaling in bone metabolism. J Bone Miner Metab, 2009,27(3):265–271
Trouvin AP, Goëb V. Receptor activator of nuclear factor–?B ligand and osteoprotegerin: maintaining the balance to prevent bone loss, Clin Interv Aging, 2010,19(5):345–354
Boyce BF, Xing L. The RANKL/RANK/OPG pathway. Curr Osteoporos Rep, 2007,5(3):98–104
D’Amelio P, Isaia G, Isaia GC. The osteoprotegerin/RANK/RANKL system: a bone key to vascular disease. J Endocrinol Invest, 2009,32(4):6–9
Shen C, Yuan Y, Li F, et al. Mechanism of genistein regulating the differentiation of vascular smooth muscle cells into osteoblasts via the OPG/RANKL pathway. Oncotarget, 2017,8(44):76857–76864
Xie H, Xie PL, Wu XP, et al. Omentin–1 attenuates arterial calcification and bone loss in osteoprotegerindeficient mice by inhibition of RANKL expression. Cardiovasc Res, 2011,92(2):296–306
Helas S, Goettsch C, Schoppet M, et al. Inhibition of receptor activator of NF–kappaB ligand by denosumab attenuates vascular calcium deposition in mice. Am J Pathol, 2009,175(2):473–478
Van Campenhout A, Golledge J. Osteoprotegerin, vascular calcification and atherosclerosis. Atherosclerosis, 2009, 204(2):321–329
Schoppet M, Kavurma MM, Hofbauer LC, et al. Crystallizing nanoparticles derived from vascular smooth muscle cells contain the calcification inhibitor osteoprotegerin. Biochem Biophys Res Commun, 2011,407(1):103–107
Liu C, Wan J, Yang Q, et al. Effects of atorvastatin on warfarin–induced aortic medial calcification and systolic blood pressure in rats. J Huazhong Univ Sci Technolog Med Sci, 2008,28(5):535–538
Clément–Lacroix P, Ai M, Morvan F, et al. Lrp5–independent activation of Wnt signaling by lithium chloride increases bone formation and bone mass in mice. Proc Natl Acad Sci USA, 2005,102(48):17406–17411
Castellot J, Favreau LV, Karnovsky MJ, et al. Inhibition of vascular smooth muscle cell growth by endothelial cell–derived heparin. Possible role of a platelet endoglycosidase. J Biol Chem, 1982,257(19):11256–11260
Olesen P, Nguyen K, Wogensen L, et al. Calcification of human vascular smooth muscle cells: associations with osteoprotegerin expression and acceleration by high–dose insulin. Am J Physiol Heart Circ Physiol, 2007,292(2):1058–1064
Heo JS, Lee SY, Lee JC. Wnt/ß–catenin signaling enhances osteoblastogenic differentiation from human periodontal ligament fibroblasts. Mol Cells, 2010,30(5): 449–454
Demer LL, Tintut Y. Inflammatory, metabolic, and genetic mechanisms of vascular calcification. Arterioscler Thromb Vasc Biol, 2014,34(4):715–723
Eijken M, Meijer IM, Westbroek I, et al. Wnt signaling acts and is regulated in a human osteoblast differentiation dependent manner. J Cell Biochem, 2008,104(2):568–579
Li X, Liu P, Liu W, et al. Dkk2 has a role in terminal osteoblast differentiation and mineralized matrix formation. Nat Genet, 2005,37(9):945–952
Osako MK, Nakagami H, Koibuchi N, et al. Estrogen inhibits vascular calcification via vascular RANKL system: common mechanism of osteoporosis and vascular calcification. Circ Res, 2010,107(4):466–475
Panizo S, Cardus A, Encinas M, et al. RANKL increases vascular smooth muscle cell calcification through a RANK–BMP4–dependent pathway. Circ Res, 2009,104(9):1041–1048
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nie, B., Zhang, Sy., Guan, Sm. et al. Role of Wnt/β-Catenin Pathway in the Arterial Medial Calcification and Its Effect on the OPG/RANKL System. CURR MED SCI 39, 28–36 (2019). https://doi.org/10.1007/s11596-019-1996-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-019-1996-4