Summary
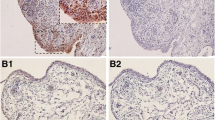
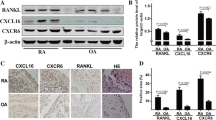
Chemokine-like factor 1 (CKLF1) is a newly cloned chemotactic cytokine with CCR4 being its functional receptor. Recent evidence demonstrates a role of CKLF1 in arthritis. The aim of this study was to quantify the expression of CKLF1 as well as assess the correlation between CKLF1 and plasma acute-phase markers. Synovium was obtained from 16 osteoarthritis (OA), 15 rheumatoid arthritis (RA) and 10 ankylosing spondylitis (AS) patients undergoing total joint arthroplasty, with other 11 patients treated for meniscal tears during sport accidents serving as normal controls. Levels of CKLF1 and CCR4 mRNA were detected by qRT-PCR, and the expression of CKLF1 was investigated by immunohistochemistry staining, subsequently analyzed with semiquantitative scores. Plasma acute-phase markers of inflammation were determined by ELISA. CKLF1 was found with a particularly up-regulated expression in synovim from AS and RA patients, and CCR4 mRNA levels increased in RA patients, not in OA or AS patients. Elevated levels of plasma markers of inflammation including CRP, ESR and Ddimer were observed in RA. Further, significantly positive correlations between relative expression levels of CKLF1 and CRP/ESR in RA patients and a positive correlation between CKLF1 and ESR in AS patients were found. There was no detectable correlation between CKLF1 and plasma D-dimer. This study confirms an increased but different level of CKLF1 in RA, OA and AS patients, all significantly higher than that in controls. Additionally, the significant positive correlations between CKLF1 levels and CRP/ESR in RA and between CKLF1 and ESR suggest that CKLF1 might contribute to the inflammation state and clinical symptoms in these rheumatic diseases. Further studies are required to investigate the utility of targeting specific CKLF1 for symptom control or disease modification in RA and AS.
Similar content being viewed by others
References
Bijlsma JW, Berenbaum F, Lafeber FP. Osteoarthritis: an update with relevance for clinical practice. Lancet, 2011,377(9783):2115–2126
Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet, 2010,376(9746):1094–1108
Braun J, Sieper J. Ankylosing spondylitis. Lancet, 2007,369(9570):1379–1390
Vander Cruyssen B, Vastesaeger N, Collantes-Estévez E. Hip disease in ankylosing spondylitis. Curr Opin Rheumatol, 2013,25(4):448–454
Sellam J, Berenbaum F. The role of synovitis in pathophysiology and clinical symptoms of osteoarthritis. Nat Rev Rheumatol, 2010,6(11):625–635
Scanzello CR, Goldring SR. The role of synovitis in osteoarthritis pathogenesis. Bone, 2012,51(2):249–257
Boissier MC, Semerano L, Challal S, et al. Rheumatoid arthritis: from autoimmunity to synovitis and joint destruction. J Autoimmun, 2012,39(3):222–228
Chen WS, Chen CH, Lin KC, et al. Immunohistological features of hip synovitis in ankylosing spondylitis with advanced hip involvement. Scand J Rheumatol, 2009, 38(2):154–155
Schett G. Synovitis—an inflammation of joints destroying the bone. Swiss Med Wkly, 2012,142:w13692
Szekanecz Z, Vegvari A, Szabo Z, et al. Chemokines and chemokine receptors in arthritis. Front Biosci (Schol Ed), 2010,2:153–167
Vergunst CE, van de Sande MG, Lebre MC, et al. The role of chemokines in rheumatoid arthritis and osteoarthritis. Scand J Rheumatol, 2005,34(6):415–425
Yang PT, Kasai H, Zhao LJ, et al. Increased CCR4 expression on circulating CD4(+) T cells in ankylosing spondylitis, rheumatoid arthritis and systemic lupus erythematosus. Clin Exp Immunol, 2004,138(2):342–347
Szekanecz Z, Koch AE, Tak PP. Chemokine and chemokine receptor blockade in arthritis, a prototype of immune-mediated inflammatory diseases. Neth J Med, 2011,69(9):356–366
Yang GY, Chen X, Sun YC, et al. Chemokine-like factor 1 (CLFK1) is over-expressed in patients with atopic dermatitis. Int J Biol Sci, 2013,9(8):759–765
Keith JC Jr, Albert LM, Leathurby Y, et al. The utility of pathway selective estrogen receptor ligands that inhibit nuclear factor-kappa B transcriptional activity in models of rheumatoid arthritis. Arthritis Res Ther, 2005,7(3): R427–438
Shao L, Li T, Mo X, et al. Expressional and functional studies of CKLF1 during dendritic cell maturation. Cell Immunol, 2010,263(2):188–195
Wang Y, Zhang Y, Yang X, et al. Chemokine-like factor 1 is a functional ligand for CC chemokine receptor 4 (CCR4). Life Sci, 2006,78(6):614–621
Li G, Li GY, Wang ZZ, et al. The chemokine-like factor 1 induces asthmatic pathological change by activating nuclear factor-?B signaling pathway. Int Immunopharmacol, 2014,20(1):81–88
Kong LL, Wang ZY, Han N, et al. Neutralization of chemokine-like factor 1, a novel C-C chemokine, protects against focal cerebral ischemia by inhibiting neutrophil infiltration via MAPK pathways in rats. J Neuroinflammation, 2014,11:112
Li T, Zhong J, Chen Y, et al. Expression of chemokinelike factor 1 is up-regulated during T lymphocyte activation. Life Sci, 2006,79(6):519–524
Altman R, Asch E, Bloch D, et al. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum, 1986,29(8): 1039–1049
Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum, 1988,31(3):315–324
Dougados M, van der Linden S, Juhlin R, et al. The European Spondylarthropathy Study Group preliminary criteria for the classification of spondylarthropathy. Arthritis Rheum, 1991,34(10):1218–1227
Tao K, Zeng H, Xiao DM, et al. Influences of IL-6R antibody on PMMA bone cement-mediated expression of OPG and RANKL in synovial fibroblasts. J Huazhong Univ Sci Technolog Med Sci, 2014,34(2):241–246
Hou WS, Li W, Keyszer G, et al. Comparison of cathepsins K and S expression within the rheumatoid and osteoarthritic synovium. Arthritis Rheum, 2002,46(3):663–674
Katschke KJ Jr, Rottman JB, Ruth JH, et al. Differential expression of chemokine receptors on peripheral blood, synovial fluid, and synovial tissue monocytes/macrophages in rheumatoid arthritis. Arthritis Rheum, 2001,44(5):1022–1032
Pearle AD, Scanzello CR, George S, et al. Elevated highsensitivity C-reactive protein levels are associated with local inflammatory findings in patients with osteoarthritis. Osteoarthritis Cartilage, 2007,15(5):516–523
Yildirim K, Karatay S, Melikoglu MA, et al. Associations between acute phase reactant levels and disease activity score (DAS28) in patients with rheumatoid arthritis. Ann Clin Lab Sci, 2004,34(4):423–426
Arvidsson NG, Gudbjörnsson B, Hällgren R, et al. Concordant message of different inflammatory markers in patients with rheumatoid arthritis. Ups J Med Sci, 1998,103(1):35–42
de Andrade KR, de Castro GR, Vicente G, et al. Evaluation of circulating levels of inflammatory and bone formation markers in axial spondyloarthritis. Int Immunopharmacol, 2014,21(2):481–486
Keenan RT, Swearingen CJ, Yazici Y, et al. Erythrocyte sedimentation rate and C-reactive protein levels are poorly correlated with clinical measures of disease activity in rheumatoid arthritis, systemic lupus erythematosus and osteoarthritis patients. Clin Exp Rheumatol, 2008,26(5):814–819
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by National Natural Science Foundation of China (No. 81441056 and 81541134).
Rights and permissions
About this article
Cite this article
Tao, K., Tang, X., Wang, B. et al. Distinct expression of chemokine-like factor 1 in synovium of osteoarthritis, rheumatoid arthritis and ankylosing spondylitis. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 70–76 (2016). https://doi.org/10.1007/s11596-016-1544-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1544-4