Summary
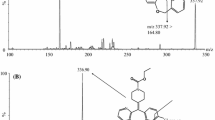
A rapid and highly selective liquid chromatography-tandem mass spectrometric (LC-MS/MS) method for the determination of megestrol in human plasma was described using medrysone as internal standard (IS). Blood samples were collected from 20 healthy volunteers after oral administration of 160 mg megestrol acetate dispersible tablets. The analytes were extracted by liquid-liquid extraction procedure and separated on a hanbon lichrospher column with the mobile phase of methanol and water containing 0.1% formic acid and 20 mmol/L ammonium acetate (5:1, v/v). Positive ion electrospray ionization with multiple reaction-monitoring mode (MRM) was employed by monitoring the transitions m/z 385.5–325.4 and m/z 387.5–327.4 for megestrol and medrysone, respectively. Under the isocratic separation conditions, the chromatographic run time was approximately 2.54 min for megestrol and 2.59 min for medrysone. The calibration curve range was from 0.5 to 200.0 ng/mL. The inter-batch and intra-batch precision and accuracy were less than 5.2% relative standard deviation (RSD) and 6.4% relative error (RE). The proposed method was successfully applied in the bioequivalence study of megestrol acetate dispersible tablets.
Similar content being viewed by others
References
Logmans A, Mous HVH, Bontenbal M, et al. Results of curettage for postmenopausal vaginal bleeding in women treated with tamoxifen and megestrol acetate for progressive metastatic breast carcinoma. Eur J Obstet Gyn R B, 1994,56(3):173–176
Jonat W, Howell A, Blomqvist C, et al. A randomized trial comparing two doses of the new selective aromatase inhibitor anastrozole (arimidex) with megestrol acetate in postmenopausal patients with advanced breast cancer. Euro J Cancer, 1996,32(3):404–412
Stuart NS, Warwick J, Blackledge GR, et al. A randomised phase III cross-over study of tamoxifen versus megestrol acetate in advanced and recurrent breast cancer. Euro J Cancer, 1996,32(11):1888–1892
Licchetta A, Correale P, Migali C, et al. Oral metronomic chemo-hormonal-therapy of metastatic breast cancer with cyclophosphamide and megestrol acetate. J Chemother, 2010,22(3):201–204
Mateen F, Jatoi A. Megestrol acetate for the palliation of anorexia in advanced, incurable cancer patients. Clin Nutr, 2006,25(5):711–715
Westman G, Bergman B, Albertsson M, et al. Megestrol acetate in advanced, progressive, hormone-insensitive cancer. Effects on the quality of life: a placebo-controlled, randomised, multicentre trial. Euro J Cancer, 1999,35(4):586–595
De Conno F, Martini C, Zecca E, et al. Megestrol acetate for anorexia in patients with far-advanced cancer: a double-blind controlled clinical trial. Euro J Cancer, 1998,34(11):1705–1709
Pascual López A, Roqué i Figuls M, Urrútia Cuchi G, et al. Systematic review of megestrol acetate in the treatment of anorexia-cachxia syndrome. J Pain Sym Man, 2004,27(4):360–369
Fietkau R, Riepl M, Kettner H, et al. Supportive use of magestrol acetate in patients with head and neck cancer during radio(chemo)therapy. Euro J Cancer, 1997,33(1):75–79
Chen HC, Leung SW, Wang CJ, et al. Effect of megestrol acetate and prepulsid on nutritional improvement in patients with head and neck cancers undergoing radiotherapy. Radiother Oncol, 1997,43(1):75–79
Hong S, Jeong IG, You D, et al. Safty of megestrol acetate in palliating anorexia-cachexia syndrome in patients with castration resistant prostate cancer. J Hematol Oncol, 2013,28(5):687–692
Adlercreutz H, Eriksen PB, Christensen MS. Plasma concentrations of megestrol acetate and medroxyprogesterone acetate after single oral administration to healthy subjects. J Pharmac Biomed Anal, 1983,1(2):153–162
Gaver RC, Movahhed HS, Farmen RH, et al. Liquid chromatographic procedure for the quantitative analysis of megestrol acetate in human plasma. J Pharm Sci, 1985,74(6):664–667
Joseph G. Timpone DJW, Ning Li, et al. The safety and pharmacokinetics of single-agent and combination therapy with megestrol acetate and dronabinol for the treatment of HIV wasting syndrom. AIDS Res Human Retroviruses, 1997,13(4):305–315
Guedes-Alonso R, Sosa-Ferrera Z, Santana-Rodríguez JJ. Simultaneous determination of hormonal residues in treated waters using ultrahigh performance liquid chromatography-tandem mass spectrometry. J Anal Methods Chem, 2013,2013:210653
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
This project was supported by the Foundation of the Zhejiang Xianju Pharmaceutical Co., Ltd (China).
Rights and permissions
About this article
Cite this article
Li, F., Zou, Xj., Zheng, H. et al. LC-MS/MS method for determination of megestrol in human plasma and its application in bioequivalence study. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 33, 912–916 (2013). https://doi.org/10.1007/s11596-013-1221-9
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-013-1221-9