Summary
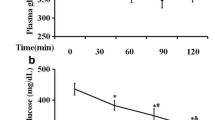
The effect of Fructus Mume formula and its separated prescription extract on insulin resistance in type 2 diabetic rats was investigated. The rat model of type 2 diabetes was established by feeding on a high-fat diet for 8 weeks and by subsequently intravenous injection of small doses of streptozotocin. Rats in treatment groups, including the Fructus Mume formula treatment group (FM), the cold property herbs of Fructus Mume formula treatment group (CFM), the warm property herbs of Fructus Mume formula treatment group (WFM), were administrated with Fructus Mume formula and its separated prescription extract by gavage, while the rats in diabetic model group (DM) and metformin group (MET) were given by gavage with normal saline and metformin correspondingly. The body weight before and after treatment was measured, and the oral glucose tolerance test (OGTT) and the insulin release test (IRT) were performed. The homeostasis model assessment-insulin resistance index (HOMA-IR) was calculated. The protein and mRNA expression levels of Insr, β-arrestin-2, Irs-1 and Glut-4 in the liver, skeletal muscle and fat tissues were detected by using Western blotting and RT-PCR respectively. The results demonstrated that, as compared with DM group, OGTT, IRT (0 h, 1 h) levels and HOMR-IR in treatment groups were all reduced, meanwhile their protein and mRNA expression levels of Insr, Irs-1 and Glut-4 in the liver, skeletal muscle and fat tissues were obviously increased, and their protein and mRNA expression levels of β-arrestin-2 in the liver and skeletal muscle tissues were also markedly increased. It was suggested that the Fructus Mume formula and its separated prescription extracts could effectively improve insulin resistance in type 2 diabetic rats, which might be related to the up-regulated expression of Insr, Irs-1 and Glut-4 in the liver, skeletal muscle and fat tissues, and β-arrestin-2 in the liver and skeletal muscle tissues.
Similar content being viewed by others
References
Yang WY, Lu JM, Weng JP, et al. Prevalence of diabetes among men and women in China. N Engl J Med, 2010,362(12):1091–1101
Gastaldelli A. Role of beta-cell dysfunction, ectopic fat accumulation and insulin resistance in the pathomRNAsis of type 2 diabetes mellitus. Diabetes Res Clin Pract, 2011,93(1):S60–S65
American Diabetes Association. Standards of Medical Care in Diabetes—2013. Diabetes Care, 2013,36(Suppl 1):S11–S66
Gagliardi L, Wittert G. Management of obesity in patients with type 2 diabetes mellitus. Curr Diabetes Rev, 2007,3(2):95–101
Church T. Exercise in obesity, metabolic syndrome, and diabetes. Prog Cardiovasc Dis, 2011,53(6):412–418
Pandey A, Tripathi P, Pandey R, et al. Alternative therapies useful in the management of diabetes: A systematic review. J Pharm Bioallied Sci, 2011,3(4):504–512
Tu X, Xie CG, Wang F, et al. Fructus MUME formula in the treatment of type 2 diabetes mellitus: a randomized controlled pilot trial. Evid Based Complemen Alternat Med, 2013,2013:787 459
Moy GA, McNay EC. Caffeine prevents weight gain and cognitive caused by a high-diet while elevating hippocampal BDNF. Physiol Behav, 2013,109:69–74
Kotronen A, Seppälä-Lindroos A, Bergholm R, et al. Tissue specificity of insulin resistance in humans: fat in the liver rather than muscle is associated with features of the metabolic syndrome. Diabetologia, 2008,51(1):130–138
Lalli CA, Pauli JR, Prada PO, et al. Statin modulates insulin signaling and insulin resistance in liver and muscle of rats fed a high-fat diet. Metabolism, 2008,57(1): 57–65
Xu XJ, Pories WJ, Dohm LG, et al. What distinguishes adipose tissue of severely obese humans who are insulin sensitive and resistant? Curr Opin Lipidol, 2013,24(1): 49–56
Soumaya K. Molecular mechanisms of insulin resistance in diabetes. Adv Exp Med Biol, 2012,771:240–251
Ramalingam L, Oh E, Thurmond DC. Novel roles for insulin receptor (IR) in adipocytes and skeletal muscle cells via new and unexpected substrates. Cell Mol Life Sci, 2013,70(16):2815–2834
Sarfstein R, Werner H. Minireview: nuclear insulin and insulin-like growth factor-1 receptors: a novel paradigm in signal transduction. Endocrinology, 2013,154(5): 1672–1679
Luan B, Zhao J, Wu H, et al. Deficiency of a beta-arrestin-2 signal complex contributes to insulin resistance. Nature, 2009,457(7233):1146–1149
Boller S, Joblin BA, Xu L, et al. From signal transduction to signal interpretation: an alternative model for the molecular function of insulin receptor substrates. Arch Physiol Biochem, 2012,118(3):148–155
Shaw LM. The insulin receptor substrate (IRS) proteins: at the intersection of metabolism and cancer. Cell Cycle, 2011,10(11):1750–1756
Morgan BJ, Chai SY, Albiston AL. GLUT4 associated proteins as targets for diabetes. Recent Pat Endocr Metab Immune Drug Discov, 2011,5(1):25–32
Mioni R, Mozzanega B, Granzotto M, et al. Insulin receptor and glucose transporters mRNA expression throughout the menstrual cycle in human endometrium: a physiological and cyclical condition of tissue insulin resistance. Gynecol Endocrinol, 2012,28(12):1014–1018
Chen G, Lu FE, Xu L, et al. The anti-diabetic effects and pharmacokinetic profiles of berberine in mice treated with Jiao-Tai-Wan and its compatibility. Phytomedicine, 2013,20(10):780–786
Zhang W, Chen Z. Mussel inspired polydopamine functionalized poly (ether ether ketone) tube for online solid-phase microextraction-high performance liquid chromatography and its application in analysis of protoberberine alkaloids in rat plasma. J Chromatogr A, 2013,1278:29–36
Lee HM, Lee OH, Kim KJ, et al. Ginsenoside Rg1 promotes glucose uptake through activated AMPK pathway in insulin-resistant muscle cells. Phytother Res, 2012,26(7):1017–1022
Yuan PF, Shang MY, Cai SQ. Study on fingerprints of chemical constituents of cinnamomi ramulus and cinnamomi cortex. Zhongguo Zhong Yao Za Zhi (Chinese), 2012,37(19):2917–2921
Zhanguo C, Jiuru L. Simultaneous and direct determination of oxalic acid, tartaric acid, malic acid, vitamin C, citric acid, and succinicacid in Fructus mume by reversed-phase high-performance liquid chromatography. J Chromatogr Sci, 2002,40(1):35–39
Li K, Liu MY, Wei XD. Study of Xixin, Duzhong and mixture to the antioxidant system of D-galactose-induced senile mice. Zhong Yao Cai (Chinese), 2000,23:161–163
Li X, Wu X, Huang L. Correlation between antioxidant activities and phenolic contents of radix Angelicae sinensis (Danggui). Molecules, 2009,14(12):5349–5361
Zhang YS, Gong ZN, Shi XP. Antioxidant effect of total flavonoids from zanthoxylum bungeanum seeds. Food Science (Chinese), 2011,32(15):70–73
Tan Y, Li J, Liu X. Deciphering the differential toxic responses of Radix aconite lateralis praeparata in healthy and hydrocortisone-pretreated rats based on serum metabolic profiles. J Proteome Res, 2013,12(1):513–524
Yu HY, Wang SJ, Teng JL, et al. Effects of Radix aconiti lateralis preparata and Rhizoma zingiberis on energy metabolism and expression of the mRNAs related to metabolism in rats. Chin J Integr Med, 2012,18(1):23–29
Author information
Authors and Affiliations
Corresponding authors
Additional information
This project was supported by grants from the National Natural Science Foundation of China (No. 81273683) and the Foundation of Hubei Provincial Health Department (No. 2012Z-Y04).
Rights and permissions
About this article
Cite this article
Li, Jb., Xu, Lj., Dong, H. et al. Effects of Chinese Fructus Mume formula and its separated prescription extract on insulin resistance in type 2 diabetic rats. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 33, 877–885 (2013). https://doi.org/10.1007/s11596-013-1215-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-013-1215-7