Abstract
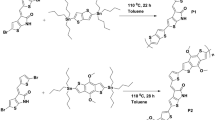
4,4′-dibromo-2-nitro-biphenyl and 4,4′-dibromo-2,3′-dinitro-biphenyl have been synthesized via nitration reaction with 4,4′-dibromobiphenyl as the raw material. Three novel thiophene derivatives, 4, 4′-di(4-hexyl-thiophen-2-yl)biphenyl, 4,4′-di(4-hexyl-thiophen-2-yl)-2-nitro-biphenyl and 4,4′-di(4-hexyl-thiophen-2-yl)-2,3′-dinitrobiphenyl were synthesized through Stille coupling reaction, followed by polymerization in the presence of FeCl3, respectively. UV-vis absorption spectra, fluorescence spectra, photoluminescence spectra and electrochemical properties of the polymers were investigated. And the band-gap (E g), HOMO orbital energy (E HOMO), and LUMO orbital energy (E LUMO) of the polymers were calculated. Among the polymers, polymer PBTN and PBTD show lower band-gap (2.67 and 2.63 eV), lower HOMO energy level (−5.38 and −5.4 eV) and broader wavelength (432 and 438 nm) than that of polymer PBTB (2.69 eV, -5.36 eV and 424 nm) with incorporation of one nitro group or two nitro groups in the main chain, respectively.
Similar content being viewed by others
References
Service R F. Outlook Brightens for Plastic Solar Cells[J]. Science, 2011, 332: 293–294
Chu T Y, Lu J P, Beaupré S, et al. Bulk Heterojunction Solar Cells Using Thieno[3,4-c]pyrrole-4,6-dione and Dithieno[3,2-b:2′,3′-d]silole Copolymer with a Power Conversion Efficiency of 7.3%3%[J]. J. Am. Chem. Soc., 2011, 133: 4 250–4 25
Park S H, Roy A, Beaupre S, et al. Bulk Heterojunction Solar Cells with Internal Quantum Efficiency Approaching 100%[J]. Nat. Photonics, 2009, 3: 297–302
Cai W, Gong X, Cao Y. Effects of Substrate Temperature on the Structural and Electrical Properties of Cu(In,Ga)Se2 Thin Films[J]. Solar Energy Materials & Solar Cells, 2010, 94: 114–118
Liang Y, Wu Y, Feng D, et al. Development of New Semiconducting Polymers for High Performance Solar Cells[J]. J. Am. Chem. Soc., 2008, 131: 56–57
Huang Y, Wang Y, Sang G Y, et al. Polythiophene Derivative with the Simplest Conjugated-Side-Chain of Alkenyl: Synthesis and Applications in Polymer Solar Cells and Field-Effect Transistors[J]. J. Phys. Chem. B, 2008, 112: 13 476–13 482
Mierloo S V, Hadipour A, Spijkman M J, et al. Improved Photovoltaic Performance of a Semicrystalline Narrow Bandgap Copolymer Based on 4H -Cyclopenta[2,1-b:3,4-b’]_dithiophene Donor and Thiazolo [5,4-d] thiazole Acceptor Units[J]. Chem. Mater., 2012, 24: 587–593
Wang M, Hu X W, Liu P, et al. Donor-Acceptor Conjugated Polymer Based on Naphtho[1,2-c:5,6-c]bis[1,2,5]thiadiazole for High-Performance Polymer Solar Cells[J]. J. Am. Chem. Soc., 2011, 133:9 638–9 641
Yang T B, Wang M, Duan C H, et al. Inverted Polymer Solar Cells with 8.4% Efficiency by Conjugated Polyelectrolyte[J]. Energy Environ. Sci., 2012, 5: 8 208–8 214
Scharber M C, Mühlbacher D, Koppe M, et al. Design Rules for Donors in Bulk-Heterojunction Solar Cells-Towards 10% Energy-Conversion Efficiency[J]. Adv. Mater., 2006, 18: 789–794
Jiang J M, Yang P A, Chen H C, et al. Synthesis, Characterization, and Photovoltaic Properties of a Low-bandgap Copolymer Based on 2,1,3-benzooxadiazole[J]. Chem. Commun., 2011, 47: 8 877–8 879
Francesco B, Gianluca M F, Francesco N, et al. Fluorinated Organic Materials for Electronic and Optoelectronic Applications: the Role of the Fluorine Atom[J]. Chem. Commun., 2007, 10: 1 003–1 022
Cheng Y J, Yang S H, Su C H. Synthesis of Conjugated Polymers for Organic Solar Cell Applications[J]. Chem. Rev., 2009, 109: 5 868–5 923
Wang E, Wang M, Wang L, et al. Donor Polymers Containing Benzothiadiazole and Four Thiophene Rings in Their Repeating Units with Improved Photovoltaic Performance[J]. Macromolecules, 2009, 42: 4 410–4 415
Roncali J. Synthetic Principles for Bandgap Control in Linear π-Conjugated Systems[J]. Chem.Rev., 1997, 97: 173–206
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No.21176193)
Rights and permissions
About this article
Cite this article
Zhou, Y., Liu, F., Wu, H. et al. Synthesis and characterization of polythiophenes bearing diphenyl groups in the conjugated chain. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 30, 568–573 (2015). https://doi.org/10.1007/s11595-015-1190-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-015-1190-x