Abstract
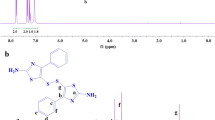
Long chain semiaromatic polyamides with high molecular weight were synthesized by the reactions of undecanediamine with various aromatic diacids, and characterized by fourier transform infrared spectra (FT-IR) and nuclear magnetic resonance (1H-NMR). The thermal behaviors were determined by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA). The solubility, dynamic mechanical, physical and mechanical properties of the polyamides were also investigated. The experimental results show that the polyamides with high molecular weights are obtained only the PH value of the polyamides salt are in the range of 7.2–7.4. The melting temperatures and the glass transition temperatures of the polyamides increase with the increase of the rigidity of aromatic diacids, and are near to those of poly(nonamethyleneterephthalamide) (PA9T).
Similar content being viewed by others
References
Sergei VL, Edward DW, Menachem L. Thermal Decomposi-tion of Aliphatic Nylons[J]. Polym. Int., 1999, 48(7): 532–557
Zhang GZ, Yoshida H, Kawai T. Miscibility of Nylon 66 and Nylon 48 Blend Evaluated by Crystallization Dynamics[J]. Thermoc. Act., 2004,416(1–2):79–85
Khanna YP, Murthy NS, Kuhn WP, et al. Pseudo Super-miscibility: Blends of Semi-crystalline Nylon Pairs Exhibiting a Single T g and a Single T m[J]. Polym. Eng. Sci., 1999, 39(11): 2 222–2 232
Persyn O, Miri V, Lefebvre JM, et al. Mechanical Behavior of Films of Miscible Polyamide 6/Polyamide 6I-6T Blends[J]. J. Polym. Sci. Part B: Polym. Phys., 2006, 44(12):1 690–1 701
Endo M, Morishima Y, Yano S, et al. Miscibility in Binary Blends of Aromatic and Alicyclic Polyamides [J]. J. Appl. Polym. Sci., 2006, 101(6): 3 971–3 978
Rao Y, Waddon AJ, Farris R. Structure-property Relation in Poly(pphenylene terephthalamide) (PPTA) Fibers[J]. Polym. 2001, 42(13): 5 937–5 940
Kudo K, Suguie J, Hirami M. Melt-polymerized Aliphatic-aromatic Copolyamides. I. Melting Points of Nylon 66 Copolymerized with Aromatic Diamines and Terephthalic Acid[J]. J. Appl. Polym. Sci., 1992, 44(9): 1 625–1 629
Ahmed JU, Ohkoshi Y, Gotoh Y. Melt Spinning and Laser-heated Drawing of a New Semiaromatic Polyamide, PA9-T fiber[J]. J. Polym. Sci. Part B: Polym. Phys., 2004, 42(3): 433–444
Morgan PW, Kwolek SL. Polymides from Phenylenediamines and Aliphatic Diacids [J]. Macromol., 1975,8(2):104–111
Jackson JB. The Effect of Absorbed Water on the Dynamic Mechanical Parameters of Some Polyamides[J]. Polym., 1969,10:159–165
Li YJ, Zhu XY, Tian GH, et al. Multiple Melting Endotherms in Melt-crystallized Nylon 10,12[J]. Polym. Int., 2001, 50(6):677–678
Ramesh C, Keller A, Eltink SJ. Studies on the Crystallization and Melting of Nylon 66:3. Melting Behaviour of Negative Spherulites by Calorimetry[J]. Polym., 1994, 35(24):5 300–5 302
Li WH, Yan DY. Synthesis and Characterization of Nylons based on Hexadecane Diacid[J]. J. Appl. Polym. Sci., 2003, 88(10):2 462–2 467
Ma JM, Song SW, Guo J. New Heat Resistance Polyamide[J]. Mod. Plast. P. Ap., 2003, 15(2):41–44
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, W., Huang, A., Liu, A. et al. Synthesis and characterization of long chain semiaromatic polyamides based on undecanediamine. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 27, 689–693 (2012). https://doi.org/10.1007/s11595-012-0530-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-012-0530-3