Abstract
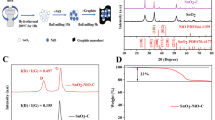
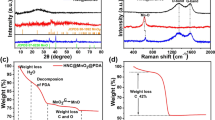
Natural spherical graphite (NSG) is a common anode material with a wide range of applications in lithium-ion batteries (LIBs). In order to improve the electrochemical performance of NSG, this paper coated NSG with MnCl2 using the molten salt method. It was demonstrated that the formation of a cladding layer with internal pores on the surface can improve the charge/discharge capacity and rate performance of NSG@MnCl2, which is higher than those of NSG mixed with MnCl2 individually. The results indicate that the capacity of NSG@MnCl2 after a 4 h reaction time at 670 °C was 493.39 mAh g−1 at a current density of 0.05 A g−1, which was 109.04 mAh g−1 higher compared to NSG. The capacity of NSG@MnCl2 was 153.38 mAh g−1 at a current density of 2A g−1, which was 128.92 mAh g−1 higher than NSG.







Similar content being viewed by others
References
Liu Y, Zhu Y, Cui Y (2019) Challenges and opportunities towards fast-charging battery materials. Nat Energy 4(7):540–550
Peled E, Menkin S (2017) Review-SEI: past, present and future. J Electrochem Soc 164(7):A1703–A1719
Endo K, Zhang HP, Fu LJ, Lee KJ, Sekine K, Takamura T, Jeong YU, Wu YP, Holze R, Wu HQ (2006) Electrochemical performance of a novel surface modified spherical graphite as anode material for lithium ion batteries. J Appl Electrochem 36(11):1307–1310
Eom JY, Cho YH, Kim SI, Han D, Sohn D (2017) Improvements in the electrochemical performance of Li4Ti5O12-coated graphite anode materials for lithium-ion batteries by simple ball-milling. J Alloy Compd 723:456–461
Kim DS, Chung DJ, Bae J, Jeong G, Kim H (2017) Surface engineering of graphite anode material with black TiO2-x for fast chargeable lithium ion battery. Electrochim Acta 258:336–342
Kim DS, Kim YE, Kim H (2019) Improved fast charging capability of graphite anodes via amorphous Al2O3 coating for high power lithium ion batteries. J Power Sources 422:18–24
Feng T, Xu Y, Zhang Z, Du X, Sun X, Xiong L, Rodriguez R, Holze R (2016) Low-cost Al2O3, low-cost Al2O3 coating layer as a preformed SEI on natural graphite powder to improve coulombic efficiency and high-rate cycling stability of lithium-ion batteries. ACS Appl Mater Interfaces 8(10):6512–6519
Yan W, Wang LY, Yi-Fan L, Zhao ZY, Yi LW, Li H, Ba YJ (2017) KCI-modified graphite as high performance anode material for lithium-ion batteries with excellent rate performance. J Phys Chem C 121(24):13052–13058
Kim K, Kim HS, Seo H (2020) Electrochemical and thermal property enhancement of natural graphite electrodes via a phosphorus and nitrogen incorporating surface treatment. Corros Sci Technol 19(01):31–36
Park MS, Lee J, Lee JW, Kim KJ, Jo YN, Woo SG, Kim YJ (2013) Tuning the surface chemistry of natural graphite anode by H3PO4 and H3BO3 treatments for improving electrochemical and thermal properties. Carbon 62:278–287
Park MS, Kim JH, Jo YN, Oh SH, Kim HS, Kim YJ (2011) Incorporation of phosphorus into the surface of natural graphite anode for lithium ion batteries. J Mater Chem 21(44):17960–17966
Wu GH, Li RY, Li ZJ, Liu JK, Gu ZG, Wang GG (2015) N-doped graphene/graphite composite as a conductive agent-free anode material for lithium ion batteries with greatly enhanced electrochemical performance. Electrochim Acta 171:156–164
Ding Y, Du J, Guo LG, Zhou HB, Yang HP, Wang F (2015) Nanoscale MnO and natural graphite hybrid materials as high-performance anode for lithium ion batteries. Electrochim Acta 170:9–15
Chae S, Choi S, Kim N, Sung J (2020) Cho J (2020) Integration of graphite and silicon anodes for the commercialization of high-energy lithium ion batteries. Angew Chem Int Ed 59(1):110–135
Liu W, Xu H, Qin H, Lv Y, Zhu G, Lei X, Lin F, Zhang Z, Wang L (2020) Rapid coating of asphalt to prepare carbon-encapsulated composites of nano-silicon and graphite for lithium battery anodes. J Mater Sci 55(10):4382–4394
Hu Y, Yu B, Qi X, Shi B, Yang J (2020) The preparation of graphite/silicon@carbon composites for lithium-ion batteries through molten salts electrolysis. J Mater Sci 55(23):10155–10167
Wang L, Zhu Y, Guo C, Zhu X, Liang J, Qian Y (2014) Ferric chloride-graphite intercalation compounds as anode materials for Li-ion batteries. Chemsuschem 7(1):87–91
Qi, X, Li, BT, Peng, SK, Wang, N, Chen, X, Yan, SJ (2020) Cobalt chloride-ferric chloride-graphite Bi-Intercalation compounds as anode materials for high-performance lithium-ion batteries[J]. Journal of Alloys and Compounds, 854
Sun YL, Han F, Zhang CZ, Zhang FQ, Zhou DW, Liu HB, Fan CL, Li XK, Liu JS (2019) FeCl3 intercalated microcrystalline graphite enables high volumetric capacity and good cycle stability for lithium-ion batteries. Energ Technol 7(1):1801091
Li Z, Zhang CZ, Han F, Wang F, Zhang FQ, Shen W, Ye C, Li XK, Liu JS (2020) Towards high-volumetric performance of Na/Li-ion batteries: a better anode material with molybdenum pentachloride -graphite intercalation compounds (MoCl5-GICs). J Mater Chem A 8(5):2430–2438
Peng PP, Wu YR, Li XZ, Zhang JH, Li YW, Cui P, Yi TF (2021) Toward superior lithium/sodium storage performance: design and construction of novel TiO2-based anode materials. Rare Met 40(11):3049–3075
Yi TF, Mei J, Peng PP, Luo SH (2019) Facile synthesis of polypyrrole-modified Li5Cr7Ti6O25 with improved rate performance as negative electrode material for Li-ion batteries. Compos B Eng 167(2019):566–572
Yi TF, Xie Y, Zhu YR, Zhu RS, Shen HY (2013) Structural and thermodynamic stability of Li4Ti5O12 anode materialfor lithium-ion battery. J Power Sources 222(2013):448–454
Augustyn V, Simon P, Dunn B (2014) Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ Sci 7(5):1597–1614
Wang SX, Chen S, Wei Q, Zhang XK, Wong SY, Sun SH, Li X (2014) Bioinspired synthesis of hierarchical porous graphitic carbon spheres with outstanding high-rate performance in lithium-ion batteries. Chem Mater 27(1):336–342
Zhu C, Wen Y, van Aken PA, Maier J, Yu Y (2015) High lithium storage performance of FeS nanodots in porous graphitic carbon nanowires. Adv Func Mater 25(15):2335–2342
Ji C, Liu Y, Xu J, Li YY, Shang YD, Su XL (2022) Enhanced microwave absorption properties of biomass-derived carbon decorated with transition metal alloy at improved graphitization degree. J Alloys Compd 890:161834
Ding F, Bolton K, Rosén A (2004) Nucleation and growth of single-walled carbon nanotubes: a molecular dynamics study. J Phys Chem B 108(45):17369–17377
Yi TF, Qiu LY, Mei J, Qi SY, Cui P, Luo SH, Zhu YR, Xie Y, He YB (2020) Porous spherical NiO@NiMoO4@PPy nanoarchitectures as advanced electrochemical pseudocapacitor materials. Sci Bull 65(2020):546–556
Wang LL, Guo C, Zhu YC, Zhou JB, Fan L, Qian YT (2014) A FeCl2-graphite sandwich composite with Cl doping in graphite layers: a new anode material for high-performance Li-ion batteries. Nanoscale 6(23):14174–14179
Liu C, Khosrozadeh A, Ren QQ, Yan LH, Goh K, Li SH, Liu J, Zhao L, Gu DM, Wang ZB (2020) Intercalation-pseudocapacitance hybrid anode for high rate and energy lithium-ion capacitors. J Energy Chem 55(2021):459–467
Funding
We received financial support from the National Natural Science Foundation of China (Grant No. 21673064, 51902072 and 22109033), Heilongjiang Touyan Team (Grant No. HITTY-20190033), Fundamental Research Funds for the Central Universities (Grant No. HIT. NSRIF. 2019040 and 2019041), Heilongjiang Province “hundred million” project science and technology major special projects (2019ZX09A02), and State Key Laboratory of Urban Water Resource and Environment (Harbin Institute of Technology) (Grant No. 2020 DX11), Heilongjiang, postdoctoral financial assistance (LBH-Z19055).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mu, J., Wu, B., Ding, F. et al. Preparation and electrochemical properties of natural spherical graphite materials coated with manganese chloride. Ionics 28, 3187–3195 (2022). https://doi.org/10.1007/s11581-022-04558-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-022-04558-8