Abstract
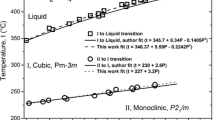
Lithium dihydrogen phosphate, LiH2PO4 (LDP), was studied for water electrolysis at elevated temperatures. It is shown that pure LDP, melting at ~ 224 °C and with correct DSC and X-ray diffraction diagram, in presence of sufficient humidity is stable up to 360 °C. Both solid and liquid LDP is structurally disordered and superprotonic conducting with conductivities reaching 0.48 S*cm−1, when determined in sealed conductivity cells. The vapor pressure above LDP, pure and mixed with LiPO3 or H2O, was determined by means of Raman spectroscopy based on N2 as internal reference gas. Pressures up to ~ 20 bar at 350 °C were found and conductivities are given for solid and liquid states. Water splitting by electrolysis, 2H2O ➔ 2H2 + O2, was demonstrated by Raman spectroscopy at ~ 225 °C and ~ 4.2 bar with Pt/W electrodes, thus showing that molten LDP has a significant potential for elevated temperature water electrolysis with high efficiency.












Similar content being viewed by others
References
Taninouchi YK, Uda T, Awakura Y, Ikeda A, Haile SM (2007) Dehydration behavior of the superprotonic conductor CsH2PO4 at moderate temperatures: 230 to 260 °C. J Mater Chem 17:3182–3189
Ulleberg Ø, Nakken T, Eté A (2010) The wind/hydrogen demonstration system at Utsira in Norway: evaluation of system performance using operational data and updated hydrogen energy system modeling tools. Int J Hydrog Energy 35(5):1841–1852
Ebbesen SD, Jensen SH, Hauch A, Mogensen MB (2014) High temperature electrolysis in alkaline cells, solid proton conducting cells, and solid oxidec. Chem Rev 114(21):10697–10734
Götz M, Lefebvre J, Mörs F, Koch AM, Graf F, Bajohr S, Reimert R, Kolb T (2016) Renewable power–to–gas: a technological and economic review. Renew Energy 85:1371–1390
Christensen E, Petrushina IM, Nikiforov AV, Berg RW, Bjerrum NJ (2020) CsH2PO4 as electrolyte for the formation of CH4 by electrochemical reduction of CO2. J Electrochem Soc 167:044511. https://doi.org/10.1149/1945-7111/ab75fa
Goñi-Urtiaga A, Presvytes D, Scott K (2012) Solid acids as electrolyte materials for proton exchange membrane (PEM) electrolysis: review. Int J Hydrog Energy 37:3358–3372
Papandrew AB, Zawodzinski TA (2014) Nickel catalysts for hydrogen evolution from CsH2PO4. J Power Sources 245:171–174
Bretzler P, Köhler K, Nikiforov AV, Christensen E, Berg RW, Bjerrum NJ (2020) Efficient water splitting electrolysis on a platinum-free tungsten carbide electrocatalyst in molten CsH2PO4 at 350–390 °C. Int J Hydr Energ S0360-3199(20):31947–31949. https://doi.org/10.1016/j.ijhydene.2020.05.145
Beltzer M (1971) Alkalimetal dihydrogen phosphate melt electrolytes, United States patent, 3615837
Baranov AI, Kopnin EM, Grebenev VV, Sin A, Zaopo A, Dubitsky Y, Caracino P (2007) Influence of humidity and thermal decomposition on the protonic conductivity of single and polycrystalline CsH2PO4. Solid State Ionics 178:657–660
Taninouchi YK, Uda T, Awakura Y (2008) Dehydration of CsH2PO4 at temperatures higher than 260 °C and the ionic conductivity of liquid product. Solid State Ionics 178:1648–1653
Nikiforov AV, Berg RW, Petrushina IM, Bjerrum NJ (2016) Specific electrical conductivity in molten potassium dihydrogen phosphate KH2PO4 – an electrolyte for water electrolysis at ∼300 °C. Appl Energy 175:545–550
Nikiforov AV, Berg RW, Bjerrum NJ (2018) Vapor pressure and specific electrical conductivity in the solid and molten H2O–CsH2PO4–CsPO3 system – a novel electrolyte for water electrolysis at ~225–400 °C. Ionics 24(9):2761–2782. https://doi.org/10.1007/s11581-017-2420-3
Osterheld RK, Audrieth LF (1952) Polymerization and depolymerization phenomena in phosphate–metaphosphate systems at higher temperatures. I. Condensation reactions involving the potassium hydrogen orthophosphates. J Phys Chem 56(1):38–42
Thilo E, Grunze H (1955) Der Entwässerungsverlauf der Dihydrogenmonophosphate des Li+, Na+, K+ und NH4+. Z Anorg Allgem Chem 281:262–273
Osterheld RK, Markowitz MM (1956) Polymerization and depolymerization phenomena in phosphate-metaphosphate systems at higher temperatures. IV. Condensation reactions of alkali metal hydrogen phosphates. J Phys Chem 60:863–867
Lavrova GV, Bulina NV, Min’kov VS, Matvienko AA (2016) Structure and thermal decomposition of Cs2HPO4·2H2O. Russian J Inorg Chem 61(3):284–290
Berg RW, Nikiforov AV, Bjerrum NJ (2020) CsH2PO4 is not stable at 260 °C unless confined. Comments to article by C. E. Botez, I. Martinez, A. Price, H. Martinez, and J.H. Leal in J. Phys. Chem. Solids 129 (2019) 324–328. J Phys Chem Solids 136: 109177
Baranov AI, Merinov BV, Tregubchenko AV, Khiznichenko VP, Shuvalov LA, Schagina NM (1989) Fast proton transport in crystals with a dynamically disordered hydrogen-bond network. Solid State Ionics 36(3–4):279–282
Lee KS (1996) Hidden nature of the high-temperature transitions in crystals of KH2PO4-type: is it a physical change? J Phys Chem Solids 57(3):333–342
Ortiz E, Vargas RA, Mellander BE (1999) On the high-temperature phase transitions of CsH2PO4: a polymorphic transition? A transition to a superprotonic conducting phase? J Chem Phys 110(10):4847–4853
Ortiz E, Vargas RA, Mellander BE (1999) On the high-temperature phase transitions of some KDP-family compounds: a structural phase transition? A transition to a bulk–high proton conducting phase? Solid State Ionics 125:177–185
Bronowska W (2001) Comment on “does the structural superionic phase transition at 231 °C in CsH2PO4 in really not exist?” [J. Chem. Phys. 110, 4847 (1999)]. J Chem Phys 114(1):611–612
Boysen DA, Haile SM, Liu H, Secco RA (2003) High-temperature behavior of CsH2PO4 under both ambient and high pressure conditions. Chem Mater 15:727–736
Boysen DA, Uda T, Chisholm CRI, Haile SM (2004) High–performance solid acid fuel cells through humidity stabilization. Science 303(5654):68–70
Park JH (2004) Possible origin of the proton conduction mechanism of CsH2PO4 crystals at high temperatures. Phys Rev B 69:054104–1–054104-6
Baranov AI, Kopnin EM, Grebenev VV, Sin A, Dubitsky Y, Caracino P (2009) Kinetics of the thermal decomposition in CsH2PO4 superprotonic crystal. Phys Status Solidi A 206(1):36–41
Otomo J, Ishigooka T, Kitano T, Takahashi H, Nagamoto H (2008) Phase transition and proton transport characteristics in CsH2PO4/SiO2 composites. Electrochim Acta 53:8186–8195
Botez CE, Carbajal D, Adiraju VAK, Tackett RJ, Chianelli RR (2010) Intermediate–temperature polymorphic phase transition in KH2PO4: a synchrotron X-ray diffraction study. J Phys Chem Solids 71:1576–1580
Chee TH, Shyuan LK, Mohamad AB, Kadhum AAH (2011) CsH2PO4: electrolyte for intermediate temperature fuel cells. Adv Mater Res 239-242(3):2492–2498
Cai W, Katrusiak A (2013) Structure of the high–pressure phase IV of KH2PO4 (KDP). Dalton Trans 42:863–866
Ettoumi H, Gao Y, Toumi M, Mhiri T (2013) Thermal analysis Raman spectroscopy and complex impedance analysis of Cu2+–doped KDP. Ionics 19(7):1067–1075
Lee K-S, Ko J-H, Moon J, Lee S, Jeon M (2008) Raman spectroscopic study of LiH2PO4. Solid State Com 145(9):487–492
Dekhili R, Kauffmann TH, Aroui H, Fontana MD (2018) Phase transformations in LiH2PO4 (LDP) revealed by Raman spectroscopy. Solid State Com 279(9):22–26. https://doi.org/10.1016/j.ssc.2018.04.016
Kweon JJ, Lee KW, Lee CE, Lee K-S (2011) Nuclear magnetic resonance study of the superprotonic conduction in LiH2PO4. Appl Phys Lett 98(26):262903–1–262903–3
Kweon JJ, Lee KW, Lee K-S, Lee CE (2013) Rotating–frame nuclear magnetic resonance study of the superprotonic conduction in LiH2PO4. Solid State Commun 171:5–7. https://doi.org/10.1016/j.ssc.2013.07.022
Lee K-S, Moon J, Lee J, Jeon M (2008) High–temperature phase transformations in LiH2PO4 and possible solid–state polymerization. Solid State Com 147(1–2):74–77. https://doi.org/10.1016/j.ssc.2008.04.011
Lee K-S, Cho J, Kim G, Jeon M (2009) LiH2PO4 crystal as a solid electrolyte. Kor J Mater Res 19(4):220–223. In Korean. Partly repeat of data in reference [37]
Kweon JJ, Lee KW, Lee CE, Lee K-S, Jo YJ (2012) Impedance spectroscopy of the superprotonic conduction in LiH2PO4. Appl Phys Lett 101(1):012905. https://doi.org/10.1063/1.4732790
Kweon JJ, Fu R, Steven E, Lee CE, Dalal NS (2014) High field MAS NMR and conductivity study of the superionic conductor LiH2PO4: critical role of physisorbed eater in its protonic conductivity. J Phys Chem C 118:13387–13393. https://doi.org/10.1021/jp501531h
Zapata VH, Castro WA, Vargas RA, Mellander B-E (2007) More studies on the PVOH–LiH2PO4 polymer system. Electrochim Acta 53:1476–1480
Zapata VH, Castro WA, Vargas RA (2013) Electrical conductivity relaxation in PVOH + LiH2PO4 + Al2O3 polymer composites. Ionics 19:83–89
Šušiƈ MV, Miniƈ DM (1981) Electric and electrochemical properties of solid LiH2PO4. Solid State Ionics 2:309–314
Huang Y, Zheng F, Zhang X, Li Y, Yin J, Li Q (2013) Tween40 surfactant effect on the formation of nano–sized LiFePO4/C powder via a solid state reaction and their cathode properties. Solid State Ionics 249–250:158–164
Rong BH, Lu YW, Chen QL, Tang K, Liu XW, Yang HZ, Wu XY, Shen F, Tang YF, Chen YF (2013) LiFePO4/C cathode materials prepared by one–step fast carbothermal method using Fe2O3 as raw materials. Advanced Mater Res (ISSN: 1662–8985) 773:709–713. doi:https://doi.org/10.4028/www.scientific.net/AMR.773.709
Zhang B, Ou X, Zheng J-C, Shen C, Ming L, Han Y-D, Wang J-l, Qin S-E (2014) Electrochemical properties of Li2FeP2O7 cathode material synthesized by using different lithium sources. Electrochim Acta 133:1–7. https://doi.org/10.1016/j.electacta.2014.03.188
Xiao ZW, Hu GR, Du K, Peng ZD (2014) A facile route for synthesis of LiFePO4/C cathode material with nano–sized primary particles. Materials and Product Engineering, Chinese J Chem Eng 22(5):590–595. https://doi.org/10.1016/S1004-9541(14)60067-7
Béléké AB, Faure C, Röder M, Hovington P, Posset U, Guerfi A, Zaghib K (2016) Chemically fabricated LiFePO4 thin film electrode for transparent batteries and electrochromic devices. Mater Sci Eng B 214:81–86. https://doi.org/10.1016/j.mseb.2016.10.001
Hou C-P, Ma Y, Zhang H, Geng W-C, Zhang Q-Y (2017) Magnetic activation of a LiFePO4@C composite cathode material. Int J Electrochem Sci 12:3221–3230. https://doi.org/10.20964/2017.04.43
Berg RW, Nikiforov AV, Petrushina IM, Bjerrum NJ (2016) Water vapor pressure over molten KH2PO4 and demonstration of water electrolysis at ∼300 °C. Appl Energy 180:269–275. https://doi.org/10.1016/j.apenergy.2016.07.123
Voronov AP, Babenko GN, Puzikov VM, Lurchenko AN (2013) Growth of LiH2PO4 single crystals from phosphate solutions. J Cryst Growth 374:49–52
Rollet AP, Lauffenburger R (1934) No 12. – Isothermes 0° et 20° du Systèm ternaire, Mémoires Presentés a la Société Chimique, P2O5 – Li2O – H2O. Bull Soc Chim Fr 5(2):146–152
Soboleva LV, Smolsky IL (1997) Growth of lithium dihydrogen phosphate (LiH2PO4) single crystals based on analysis of dissolution diagram for the Li2O–P2O5–H2O system. Kristallografiya 42:762–764 [Crystallogr Reports 42:700–702]
Rumble JR, ed., (2018) Physical constants of organic compounds, Handbook of chemistry and physics, 98th Edition (Internet Version 2018), CRC Press/Taylor & Francis, Boca Raton, FL
Krumgalz BS (1994) Mineral solubility in water at various temperatures. Israel Oceanographic and Limnological Research Ltd., Haifa
Wikipedia®. https://en.wikipedia.org/wiki/Solubility_table. Accessed 22 May 2019
Lee K-S, Oh I-H, Kweon JJ, Lee CE, Ahn S-H (2012) Crystal growth and morphology of LiH2PO4. Mater Chem Phys 136:802–808. https://doi.org/10.1016/j.matchemphys.2012.08.001
Soboleva LV, Voloshin AE (2004) Me2O–P2O5–H2O solubility phase diagrams and growth of MeH2PO4 single crystals (Me = Li, Na, K, Rb, Cs, NH4). Kristallografiya 49(4):773–777. Translation in Crystallography Reports 49(4):693–697
Catti M, Ivaldi G (1977) Crystal structure of LiH2PO4, structural topology and hydrogen bonding in the alkaline dihydrogen orthophosphates. Z Kristallogr 146:215–226
Oh IH, Lee K-S, Meven M, Heger G, Lee CE (2010) Crystal structure of LiH2PO4 studied by single–crystal neutron diffraction. J Phys Soc Japan 79(6):074606–1 − 074606-4
Lee K-S, Ko J-H, Schmidt VH (2005) Raman–scattering study of LiH2PO4. J Kor Phys Soc 46(1):104–107
Nguili N, Suñol JJ, Bulou A, Toumi M (2015) Crystal structure and spectroscopic studies of LiNH4(H2PO4)2 – a new solid acid in the LiH2PO4–NH4H2PO4 system. J Solid State Chem 230:272–278. https://doi.org/10.1016/j.jssc.2015.07.023
Rhimi T, Nguili N, Guermazi S, Bulou A, Toumi M (2016) Structural and vibrational study a new potassium lithium dihydrogenphosphate KLi(H2PO4)2. Vibr Spectrosc 86:50–60. https://doi.org/10.1016/j.vibspec.2016.05.008
Naïli H, Mhiri T, Daoud A (2000) Crystal structure, characterisation and vibrational study of a mixed compound Cs0.4Rb0.6H2PO4. Phase Trans 71:271–286
Naïli H, Vendier L, Jaud J, Mhiri T (2001) The influence of partial substitution of phosphorus by arsenic in monoclinic CsH2PO4. X-ray single crystal, vibrational and phase transitions in the mixed CsH2(PO4)0.72(AsO4)0.28. Solid State Sci 3:677–687
Benkhoucha R, Wunderlich B (1978) Crystallization during polymerization of lithium dihydrogen phosphate. I. Nucleation of the macromolecular crystal from the oligomer melt. Z Anorg Allg Chem 444:256–266
Benkhoucha R, Wunderlich B (1978) Crystallization during polymerization of lithium dihydrogen phosphate. II. Crystal growth by dimer addition. Z Anorg Allg Chem 444:267–276
Benkhoucha R, Wunderlich B (1979) On the existence of low- and high-temperature crystal forms of lithium polyphosphate. Acta Cryst B35:265–267 http://scripts.iucr.org/cgi-bin/paper?S0567740879003277
Benkhoucha R, Wunderlich CC, Wunderlich B (1979) Melting and crystallization of a polyphosphate. J Polymer Sci, Polymer Phys Ed 17:2151–2162
Goñi-Urtiaga A (2014) Thesis: Cesium dihydrogen phosphate as electrolyte for Intermediate Temperature Proton Exchange Membrane Water Electrolysis (IT-PEMWE), School Of Chemical Engineering and Advanced Materials (CEAM), Newcastle University, Newcastle upon Tyne, UK. Chapter 4.4: Physical and Chemical Stability. p. 83–89
Rodier M, Li Q, Berg RW, Bjerrum NJ (2016) Determination of water vapor pressure over corrosive chemicals versus temperature using Raman spectroscopy as exemplified with 85.5% phosphoric acid. Appl Spectrosc 70(7):1186–1194
Liu C, Berg RW (2012) Nonlinearity in intensity versus concentration dependence for the deep UV resonance Raman spectra of toluene and heptane. Appl Spectrosc Rev 48(5):425–437
Bribes JL, Gaufrès R, Monan M, Lapp M, Penney CM (1976) Raman band contours for water vapor as a function of temperature. Appl Phys Letters 28:336–337
Murphy WF (1978) The ro–vibrational Raman spectra of water vapour ν1 and ν3. Mol Phys 36:727–732
Avila G, Fernández JM, Maté B, Tejeda G, Montero S (1999) Ro–vibrational Raman cross sections of water vapor in the OH stretching region. J Mol Spectrosc 196:77–92
Berg RW, Maijó Ferré I, Cline Schäffer SJ (2006) Raman spectroscopy evidence of 1:1:1 complex formation during dissolution of WO3 in a melt of K2S2O7:K2SO4. Vib Spectrosc 42:346–352
Liu C, Berg RW (2012) Determining the spectral resolution of a charge–coupled device (CCD) Raman instrument. Appl Spectrosc 66(9):1034–1043
Berg RW, Nørbygaard T (2006) Wavenumber calibration of CCD detector Raman spectrometers controlled by a sinus arm drive. Appl Spectrosc Rev 41:165–183
Berg RW, von Winbush S, Bjerrum NJ (1980) Negative oxidation states of the chalcogenes in molten salts.1. Raman spectroscopic studies on aluminum chlorosulfides formed in chloride and chloroaluminate melts and some related solid and dissolved compounds. Inorg Chem 19:2688–2698
Lemmon EW, McLinden MO, Friend DG (1998) Thermophysical properties of fluid systems. NIST Chemistry WebBook, NIST Standard Reference Database Number 69, Eds. P.J. Linstrom and W.G. Mallard, National Institute of Standards and Technology, Gaithersburg MD, 20899, doi:https://doi.org/10.18434/T4D303, (retrieved August 16, 2017)
Jones G, Bradshaw BC (1993) The measurement of the conductance of electrolytes. V. a redetermination of the conductance of standard potassium chloride solutions in absolute units. J Am Chem Soc 55:1780–1800
Shreiner RH, Pratt KW (2004) Standard reference materials: primary standards and standard reference materials for electrolytic conductivity, NIST Special Publication 260–142, U.S. Department of Commerce, Gaitersburg MD
Hjuler HA, Berg RW, Zachariasen K, Bjerrum NJ (1985) Specific conductivity of NaCl–AlCl3 and NaCl–AlCl3–Al2S3 melts. J Chem Eng Data 30:203–208
Choi BK, Lee MN, Kim JJ (1989) Raman spectra of the NaH2PO4 crystal. J Raman Spectrosc 20(1):11–15
Romain F, Novak A (1991) Raman study of the high-temperature phase transition in CsH2PO4. J Mol Struct 263:69–74
Carr CW, Feit MD, Johnson MA, Rubenchik AM (2006) Complex morphology of laser–induced bulk damage in K2H2−xDxPO4 crystals. Appl Phys Lett 89:131901–1–131901-3
Petitpas G, Aceves SM, Matthews MJ, Smith JR (2014) Para–H2 to ortho–H2 conversion in a full–scale automotive cryogenic pressurized hydrogen storage up to 345 bar. Int J. Hydrogen Energ 39:6533–6547. https://doi.org/10.1016/j.ijhydene.2014.01.205
Stoicheff BP (1957) High resolution Raman spectroscopy of gases IX. Spectra of H2, HD and D2. Canad J. Phys 35:730–741
Edwards HGM, Farwell DW, Gorvin AC, Long DA (1986) Pure rotational and vibration–rotational Raman spectra of 1H2, 1H2H and 2H2. J Raman Spectrosc 17(1):129–131. https://doi.org/10.1002/jrs.1250170126
Jennings DE, Weber A, Brault JW (1987) FTS–Raman flame spectroscopy of high–J lines in H2 and D2. J Mol Spectrosc 126(1):19–28. https://doi.org/10.1016/0022-2852(87)90072-5
Schrötter HW, Klöckner HW (1979) Raman scattering cross sections in gas and liquids. Topics in current physics, vol. 11. In: Weber A (ed) Raman spectroscopy of gases and liquids. Springer Verlag, New York, pp 123–166
Schrötter HW (1995) Raman spectra of gasses. In: B. Schrader (ed.) infrared and Raman spectroscopy. Methods and applications. Weinheim, Germany: Wiley VCH, pp. 277–296
Schrötter HW (2001) Raman spectra of gasses. In: Lewis IR, Edwards HGM (eds) Handbook of Raman spectroscopy: from the research laboratory to the process line (practical spectroscopy). CRC Press, Boca Raton, FL, pp 307–348
Garrabos Y, Echargui MA, Marsault-Herail F (1989) Comparison between the density effects on the levels of the Raman spectra of the Fermi resonance doublet of the 12C16O2 and 13C16O2 molecules. J Chem Phys 91(10):5869–5881
Petrov DV, Matrosov II, Sedinkin DO, Zaripov AR (2018) Raman spectra of nitrogen, carbon dioxide, and hydrogen in a methane environment. Opt Spectrosc 124(1):8–12
Acknowledgments
We acknowledge general support from the DTU departments of Chemistry and Energy. We thank Larisa Seerup of DTU Energy for performing the differential scanning calorimetry and X-ray measurements. The quartz cells were made by the now-deceased glass blower Jan Patrick Scholer.
Funding
This investigation has been supported by the “ForskEL” research program under grant no. 2016–1-12449 of the Danish Energinet.dk program. We received economic support from the Climate-KIC program TC2018B_2.1.6-CADEL2_P240-1A and the Discovery Grant pool of DTU.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Berg, R.W., Nikiforov, A.V. & Bjerrum, N.J. Vapor pressure and specific electrical conductivity in the H2O–LiH2PO4–LiPO3 system—a novel electrolyte for water electrolysis at elevated temperature. Ionics 27, 703–719 (2021). https://doi.org/10.1007/s11581-020-03867-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-020-03867-0