Abstract
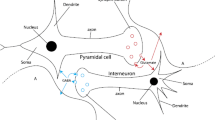
Glutamate (Glu) is a predominant excitatory neurotransmitter that acts on glutamate receptors to transfer signals in the central nervous system. Abnormally elevated extracellular glutamate levels is closely related to the generation and transition of epileptic seizures. However, there lacks of investigation regarding the role of extracellular glutamate homeostasis dysregulated by astrocyte in neuronal epileptic discharges. According to this, we propose a novel neuron-astrocyte computational model (NAG) by incorporating extracellular Glu concentration dynamics from three aspects of regulatory mechanisms: (1) the Glu uptake through astrocyte EAAT2; (2) the binding and release Glu via activating astrocyte mGluRs; and (3) the Glu free diffusion in the extracellular space. Then the proposed model NAG is analyzed theoretically and numerically to verify the effect of extracellular Glu homeostasis dysregulated by such three regulatory mechanisms on neuronal epileptic discharges. Our results demonstrate that the neuronal epileptic discharges can be aggravated by the downregulation expression of EAAT2, the aberrant activation of mGluRs, and the elevated Glu levels in extracellular micro-environment; as well as various discharge states (including bursting, mixed-mode spiking, and tonic firing) can be transited by their combination. Furthermore, we find that such factors can also alter the bifurcation threshold for the generation and transition of epileptic discharges. The results in this paper can be helpful for researchers to understand the astrocyte role in modulating extracellular Glu homeostasis, and provide theoretical basis for future related experimental studies.









Similar content being viewed by others
References
Albrecht J, Zielińska M (2017) Mechanisms of excessive extracellular glutamate accumulation in temporal lobe epilepsy. Neurochem Res 42:1724–1734. https://doi.org/10.1007/s11064-016-2105-8
Alcoreza OB, Patel DC, Tewari BP, Sontheimer H (2021) Dysregulation of ambient glutamate and glutamate receptors in epilepsy: an astrocytic perspective. Front Neurol 12:652159. https://doi.org/10.3389/fneur.2021.652159
Allam SL, Ghaderi VS, Bouteiller JMC, Legendre A, Ambert N, Greget R, Bischoff S, Baudry M, Berger TW (2012) A computational model to investigate astrocytic glutamate uptake influence on synaptic transmission and neuronal spiking. Front Comput Neurosci 6:70
Amiri M, Montaseri G, Bahrami F (2013) A phase plane analysis of neuron-astrocyte interactions. Neural Netw 44:157–165
Attwell D, Laughlin SB (2001) An energy budget for signaling in the grey matter of the brain. J Cereb Blood Flow Metab 21(10):1133–1145
Beghi E (2020) The epidemiology of epilepsy. Neuroepidemiology 54(2):185–191. https://doi.org/10.1159/000503831
Cressman JR, Ullah G, Ziburkus J, Schiff SJ, Barreto E (2009) The influence of sodium and potassium dynamics on excitability, seizures, and the stability of persistent states: I. Single neuron dynamics. J Comput Neurosci 26:159–170. https://doi.org/10.1007/s10827-008-0132-4
De Pittà M, Brunel N (2016) Modulation of synaptic plasticity by glutamatergic gliotransmission: a modeling study. Neural Plast 2016:7607924. https://doi.org/10.1155/2016/7607924
Depannemaecker D, Ivanov A, Lillo D, Spek L, Bernard C, Jirsa V (2022) A unified physiological framework of transitions between seizures, sustained ictal activity and depolarization block at the single neuron level. J Comput Neurosci 50:33–49
Destexhe A, Mainen ZF, Sejnowski TJ (1994) Synthesis of models for excitable membranes, synaptic transmission and neuromodulation using a common kinetic formalism. J Comput Neurosci 1:195–230
Destexhe A, Mainen ZF, Sejnowski TJ et al (1998) Kinetic models of synaptic transmission. Methods Neuronal Model 2:1–25
Dingledine R, Borges K, Bowie D, Traynelis SF (1999) The glutamate receptor ion channels. Pharmacol Rev 51(1):7–62
During MJ, Spencer DD (1993) Extracellular hippocampal glutamate and spontaneous seizure in the conscious human brain. Lancet 341(8861):1607–1610
Eid T, Gruenbaum SE, Dhaher R, Lee TSW, Zhou Y, Danbolt NC (2016) The glutamate-glutamine cycle in epilepsy. Adv Neurobiol 13:351–400
Fan D, Wang Q, Perc M (2015) Disinhibition-induced transitions between absence and tonic-clonic epileptic seizures. Sci Rep 5(1):1–12
Flanagan B, McDaid L, Wade J, Wong-Lin K, Harkin J (2018) A computational study of astrocytic glutamate influence on post-synaptic neuronal excitability. PLoS Comput Biol 14(4):e1006040
Gosak M, Milojević M, Duh M, Skok K, Perc M (2022) Networks behind the morphology and structural design of living systems. Phys Life Rev 41:1–21
Green JL, Dos Santos WF, Fontana ACK (2021) Role of glutamate excitotoxicity and glutamate transporter EAAT2 in epilepsy: opportunities for novel therapeutics development. Biochem Pharmacol 193:114786. https://doi.org/10.1016/j.bcp.2021.114786
Hanada T (2020) Ionotropic glutamate receptors in epilepsy: a review focusing on AMPA and NMDA receptors. Biomolecules 10(3):464. https://doi.org/10.3390/biom10030464
Hemami M, Parand K, Rad JA (2019) Numerical simulation of reaction-diffusion neural dynamics models and their synchronization/desynchronization: application to epileptic seizures. Comput Math with Appl 78(11):3644–3677
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117(4):500–544
Homer LD, Shelton JB, Williams TJ (1983) Diffusion of oxygen in slices of rat brain. Am J Physiol 244(1):R15–R22
Hotz AL, Jamali A, Rieser NN, Niklaus S, Aydin E, Myren-Svelstad S, Lalla L, Jurisch-Yaksi N, Yaksi E, Neuhauss SC (2022) Loss of glutamate transporter eaat2a leads to aberrant neuronal excitability, recurrent epileptic seizures, and basal hypoactivity. Glia 70(1):196–214. https://doi.org/10.1002/glia.24106
Hübel N, Hosseini-Zare MS, Žiburkus J, Ullah G (2017) The role of glutamate in neuronal ion homeostasis: a case study of spreading depolarization. PLoS Comput Biol 13(10):e1005804
Izhikevich EM (2007) Dynamical systems in neuroscience: the geometry of excitability and bursting. MIT Press, New York
Ji Q, Qie X, Ye M (2023) Dynamical analysis of astrocyte-induced neuronal hyper-excitation. Nonlinear Dyn 111(8):7713–7728
Kofuji P, Araque A (2021) G-protein-coupled receptors in astrocyte-neuron communication. Neuroscience 456:71–84. https://doi.org/10.1016/j.neuroscience.2020.03.025
Kong Q, Takahashi K, Schulte D, Stouffer N, Lin Y, Lin CLG (2012) Increased glial glutamate transporter EAAT2 expression reduces epileptogenic processes following pilocarpine-induced status epilepticus. Neurobiol Dis 47(2):145–154
Krishnan GP, Bazhenov M (2011) Ionic dynamics mediate spontaneous termination of seizures and postictal depression state. J Neurosci 31(24):8870–8882. https://doi.org/10.1523/JNEUROSCI.6200-10.2011
Levy LM, Warr O, Attwell D (1998) Stoichiometry of the glial glutamate transporter GLT-1 expressed inducibly in a Chinese hamster ovary cell line selected for low endogenous \({\text{Na}}^+\)-dependent glutamate uptake. J Neurosci 18(23):9620–9628
Li YX, Rinzel J (1994) Equations for InsP3 receptor-mediated \([{\text{Ca}}^{2+}]_{\text{i}}\) oscillations derived from a detailed kinetic model: a Hodgkin–Huxley like formalism. J Theor Biol 166(4):461–473
Li J, Tang J, Ma J, Du M, Wang R, Wu Y (2016a) Dynamic transition of neuronal firing induced by abnormal astrocytic glutamate oscillation. Sci Rep 6(1):32343
Li J, Wang R, Du M, Tang J, Wu Y (2016b) Dynamic transition on the seizure-like neuronal activity by astrocytic calcium channel block. Chaos, Solitons Fractals 91:702–708
Mahmoud S, Gharagozloo M, Simard C, Gris D (2019) Astrocytes maintain glutamate homeostasis in the CNS by controlling the balance between glutamate uptake and release. Cells 8(2):184. https://doi.org/10.3390/cells8020184
Manninen T, Havela R, Linne ML (2018) Computational models for calcium-mediated astrocyte functions. Front Comput Neurosci 12:14. https://doi.org/10.3389/fncom.2018.00014
Nadkarni S, Jung P (2004) Dressed neurons: modeling neural-glial interactions. Phys Biol 1(1):35
Oschmann F, Mergenthaler K, Jungnickel E, Obermayer K (2017) Spatial separation of two different pathways accounting for the generation of calcium signals in astrocytes. PLoS Comput Biol 13(2):e1005377
Pál B (2018) Involvement of extrasynaptic glutamate in physiological and pathophysiological changes of neuronal excitability. Cell Mol Life Sci 75:2917–2949. https://doi.org/10.1007/s00018-018-2837-5
Patel DC, Tewari BP, Chaunsali L, Sontheimer H (2019) Neuron–glia interactions in the pathophysiology of epilepsy. Nat Rev Neurosci 20(5):282–297. https://doi.org/10.1038/s41583-019-0126-4
Peterson AR, Binder DK (2020) Astrocyte glutamate uptake and signaling as novel targets for antiepileptogenic therapy. Front Neurol 11:1006
Petrushanko IY, Bogdanov NB, Lapina N, Boldyrev AA, Gassmann M, Bogdanova AY (2007) Oxygen-induced regulation of Na/K ATPase in cerebellar granule cells. J Gen Physiol 130(4):389–398
Pfisterer U, Petukhov V, Demharter S, Meichsner J, Thompson JJ, Batiuk MY, Asenjo-Martinez A, Vasistha NA, Thakur A, Mikkelsen J et al (2020) Identification of epilepsy-associated neuronal subtypes and gene expression underlying epileptogenesis. Nat Commun 11(1):5038. https://doi.org/10.1038/s41467-020-18752-7
Rangel GC, Prezioso J, Gerardo-Giorda L, Somersalo E, Calvetti D (2019) Brain energetics plays a key role in the coordination of electrophysiology, metabolism and hemodynamics: evidence from an integrated computational model. J Theor Biol 478:26–39
Reato D, Cammarota M, Parra LC, Carmignoto G (2012) Computational model of neuron-astrocyte interactions during focal seizure generation. Front Comput Neurosci 6:81
Rho JM, Boison D (2022) The metabolic basis of epilepsy. Nat Rev Neurol 18(6):333–347. https://doi.org/10.1038/s41582-022-00651-8
Robinson MB, Lee ML, DaSilva S (2020) Glutamate transporters and mitochondria: signaling, co-compartmentalization, functional coupling, and future directions. Neurochem Res 45:526–540
Rusakov DA, Kullmann DM (1998) Extrasynaptic glutamate diffusion in the hippocampus: ultrastructural constraints, uptake, and receptor activation. J Neurosci 18(9):3158–3170
Sætra MJ, Einevoll GT, Halnes G (2020) An electrodiffusive, ion conserving Pinsky–Rinzel model with homeostatic mechanisms. PLoS Comput Biol 16(4):e1007661
Seifert G, Carmignoto G, Steinhäuser C (2010) Astrocyte dysfunction in epilepsy. Brain Res Rev 63(1–2):212–221. https://doi.org/10.1016/j.brainresrev.2009.10.004
Silchenko AN, Tass PA (2008) Computational modeling of paroxysmal depolarization shifts in neurons induced by the glutamate release from astrocytes. Biol Cybern 98:61–74
Spampinato SF, Copani A, Nicoletti F, Sortino MA, Caraci F (2018) Metabotropic glutamate receptors in glial cells: a new potential target for neuroprotection? Front Mol Neurosci 11:414
Steriade M, Amzica F, Neckelmann D, Timofeev I (1998) Spike-wave complexes and fast components of cortically generated seizures. II. extra-and intracellular patterns. J Neurophysiol 80(3):1456–1479
Tanaka K, Watase K, Manabe T, Yamada K, Watanabe M, Takahashi K, Iwama H, Nishikawa T, Ichihara N, Kikuchi T et al (1997) Epilepsy and exacerbation of brain injury in mice lacking the glutamate transporter GLT-1. Science 276(5319):1699–1702
Thijs RD, Surges R, O’Brien TJ, Sander JW (2019) Epilepsy in adults. Lancet 393(10172):689–701
Thompson JK, Peterson MR, Freeman RD (2003) Single-neuron activity and tissue oxygenation in the cerebral cortex. Science 299(5609):1070–1072
Tian G, Azmi H, Takano T, Xu Q, Peng W, Lin J, Oberheim N, Lou N, Wang X, Zielke HR et al (2005) An astrocytic basis of epilepsy. Nat Med 11(9):973–981
Toglia P, Ullah G (2019) Mitochondrial dysfunction and role in spreading depolarization and seizure. J Comput Neurosci 47:91–108
Traub RD, Jefferys J, Miles R, Whittington MA, Tóth K (1994) A branching dendritic model of a rodent CA3 pyramidal neurone. J Physiol 481(1):79–95
Tuttle A, Riera Diaz J, Mori Y (2019) A computational study on the role of glutamate and NMDA receptors on cortical spreading depression using a multidomain electrodiffusion model. PLoS Comput Biol 15(12):e1007455
Ullah G, Schiff SJ (2010) Assimilating seizure dynamics. PLoS Comput Biol 6(5):e1000776. https://doi.org/10.1371/journal.pcbi.1000776
Umpierre AD, West PJ, White JA, Wilcox KS (2019) Conditional knock-out of mGluR5 from astrocytes during epilepsy development impairs high-frequency glutamate uptake. J Neurosci 39(4):727–742. https://doi.org/10.1523/JNEUROSCI.1148-18.2018
V-Ghaffari B, Kouhnavard M, Elbasiouny SM (2017) Mixed-mode oscillations in pyramidal neurons under antiepileptic drug conditions. PLoS One 12(6):e0178244
Wei Y, Ullah G, Ingram J, Schiff SJ (2014) Oxygen and seizure dynamics: II. Computational modeling. J Neurophysiol 112(2):213–223. https://doi.org/10.1152/jn.00541.2013
Wei Y, Ullah G, Schiff SJ (2014) Unification of neuronal spikes, seizures, and spreading depression. J Neurosci 34(35):11733–11743. https://doi.org/10.1523/JNEUROSCI.0516-14.2014
Wendling F, Benquet P, Bartolomei F, Jirsa V (2016) Computational models of epileptiform activity. J Neurosci Methods 260:233–251
Yang Z, Wang J, Yu C, Xu P, Zhang J, Peng Y, Luo Z, Huang H, Zeng J, Xu Z (2018) Inhibition of p38 MAPK signaling regulates the expression of EAAT2 in the brains of epileptic rats. Front Neurol 9:925
Yao C, He Z, Nakano T, Shuai J (2018) Spiking patterns of a neuron model to stimulus: Rich dynamics and oxygen’s role. Chaos 28(8):083112
Zhao J, Fan D, Wang Q, Wang Q (2021) Dynamical transitions of the coupled Class I (II) neurons regulated by an astrocyte. Nonlinear Dyn 103:913–924
Acknowledgements
This work was supported by the National Natural Science Foundation of China under Grant Nos. 12071369 and 62006189, the Key Industry Innovation Chain (Group) of Shaanxi Province under Grant No. 2019ZDLSF02-09-02, the China Postdoctoral Science Foundation under Grant No. 2022M722580, and the Natural Science Foundation of Shaanxi Province under Grant No. 2023-JC-QN-0028.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, D., Li, S., Pan, M. et al. The role of extracellular glutamate homeostasis dysregulated by astrocyte in epileptic discharges: a model evidence. Cogn Neurodyn 18, 485–502 (2024). https://doi.org/10.1007/s11571-023-10001-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11571-023-10001-z