Abstract
In fungi, little is known about connections between volatile organic compound (VOC) formation and developmental stages that are amongst others triggered by fruiting-related genes (FRGs). We analysed the volatilomes of Schizophyllum commune during different developmental stages in a variety of FRG-deletion strains and wild-type strains. The deletion strains Δtea1Δtea1, Δwc-2Δwc-2 and Δhom2Δhom2 were unable to develop fruiting bodies, and Δfst4Δfst4 formed only rudimentary fruiting body structures. Early developmental stages of these strains were dominated by esters, including methyl 2-methylbutanoate, ethyl 2-methylbutanoate, isobutyl 2-methylpropionate, and 2-methylbutyl acetate, of which the last three were not found in the headspace (HS) of the wild-type samples. Compared to the wild type, in the HS of hom2con samples, that are able to form fruiting bodies, methyl 2-methylbutanoate was the most abundant substance at early stages (68–81% of the total peak area). In contrast to fruiting body forming strains, Δtea1Δtea1, Δwc-2Δwc-2, Δhom2Δhom2 and Δfst4Δfst4 showed less sesquiterpenes in the HS. However, the sesquiterpenes found in the HS of FRG-deletion strains, namely, (E)-nerolidol, δ-cadinene, L-calamenene, α-bisabolol and β-bisabolene, were not present in hom2con or wild-type strains that mainly formed fruiting bodies and barely mycelium. Several sesquiterpenes, including α-guaiene, chamigrene and γ-gurjunene, were only found in presence of fruiting bodies. Our results show remarkable connections between FRGs, fruiting body development and VOC production in S. commune, especially counting for sesquiterpenes. Future studies are needed to reveal whether FRGs directly regulates VOC formation or indirectly by changing the phenotype.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
More than 300 distinct fungal volatile organic compounds (VOCs) have been identified so far, being in many cases aroma active (Dickschat 2017) that determine, e.g. the quality of edible fungal fruiting bodies. VOC profiles of several fungi have been analysed so far (Chen and Ho 1986; Costa et al. 2015; Dickschat 2017; Fraatz and Zorn 2011; Kleofas et al. 2015; Rühl et al. 2018; Venkateshwarlu et al. 1999). However, studies investigating volatilomes (the composition of all VOCs produced by a fungus during one time point) are often focusing on one developmental stage ignoring potential dynamics in VOC release. Studies investigating volatilomes during the fungal life cycle are mainly focusing on fruiting bodies of mushrooms (Cho et al. 2006; Combet et al. 2009; Cruz et al. 1997; Li et al. 2016; Mau et al. 1997; Tasaki et al. 2019; Zawirska-Wojtasiak et al. 2007; Zhang et al. 2008) analysing the aroma profiles in context of food quality. Prior to extraction, fruiting bodies are often grinded or otherwise damaged, which can result in unnatural VOC compositions (Combet et al. 2009). Furthermore, the focus on fruiting bodies neglects the role of the mycelium for VOC formation and possible interactions between fruiting bodies and mycelium during development. Studies analysing fungal volatilomes under more natural circumstances are rare. Recently, we established a system consisting of modified crystallizing dishes allowing a non-invasive analysis of fungal VOCs from complete cultures during different stages of the fungal life cycle using solid-phase microextraction coupled gas chromatography mass spectrometry (SPME–GC–MS) (Orban et al. 2019). Applying this system on Cyclocybe aegerita, remarkable relations between fruiting body development and VOC compositions, especially counting for the release of sesquiterpenes during sporulation, have been depicted (Orban et al. 2020). A subsequent transcriptome study of C. aegerita showed that the vegetative mycelium rather than the fruiting body is the origin of the sesquiterpenes (Orban et al. 2021).
The formation of fruiting bodies is one of the most complex developmental processes in the fungal life cycle (Sánchez-García et al. 2020). It starts with the aggregation of hyphae to form mycelial cords, developing on local spots of the mycelium into hyphal knots by intense branching. The hyphal knots develop successively into fruiting body initials (secondary hyphal knots) (Kües 2000; Kües and Liu 2000; Kües et al. 2018). Cell differentiation results in the formation of bipolar primordia already comprising the plectenchyme as characteristic tissue for mature fruiting bodies. Subsequently, differentiated primordia develop into fruiting bodies primarily due to cell elongation rather than cell differentiation (Kües 2000). In mature fruiting bodies of Basidiomycota, karyogamy and meiosis take place in the basidium, leading to the formation of basidiospores that are released during sporulation and marking the beginning of a new life cycle (Oberwinkler 1982). The fruiting process is regulated by environmental factors; e.g. light and low concentrations of CO2 have been proven to be necessary for proper fruiting body development (Kinugawa et al. 1994; Kües and Navarro-González 2015; Turner 1977; Wessels 1993). Additionally, several gene deletion studies on especially S. commune and Coprinopsis cinerea have proven that certain genes, so-called fruiting-related genes (FRGs), are essential for the fruiting process (Arima et al. 2004; Kamada et al. 2010; Knabe et al. 2013; Kuratani et al. 2010; Liu et al. 2006; Muraguchi and Kamada 1998; Muraguchi et al. 2008; Murata et al. 1998; Ohm et al. 2010, 2011, 2013; Pelkmans et al. 2017; Terashima et al. 2005). In S. commune, induction of fruiting by light is connected to the white collar (WC) complex consisting of the two proteins, WC-1 and WC-2. WC-1 contains a blue light sensing domain and WC-2 functions as a transcription factor. Deletion of wc-1 and/or wc-2 results in a blind phenotype under light conditions with symmetrical mycelial growth and being unable to form hyphal knots, primordia or fruiting bodies (Ohm et al. 2013). Inactivation of the homeodomain transcription factor gene hom2 leads to a comparable phenotype (Ohm et al. 2011). The transcription factor Tea1 is also involved in fruiting body formation, and tea1 deletion mutants show symmetrically mycelial growth with only scarce formation of flask-like fruiting bodies (Pelkmans et al. 2017). Inactivation of the zinc finger transcription factor gene fst4 results in mycelial growth comparable to the wild type but without formation of fruiting bodies (Ohm et al. 2011; Pelkmans et al. 2017). Several other FRGs have been identified in S. commune, including bri1, c2h2, fst3, gat1 and hom1, regulating different stages of fruiting (Pelkmans et al. 2017). However, connections between fungal FRGs and VOC formation have not been studied so far.
To address this gap, we studied VOC formation in the S. commune wild-type dikaryon H4-8; the dikaryotic deletion strains Δfst4Δfst4, Δtea1Δtea1, Δwc-2Δwc-2 and Δhom2Δhom2 (Ohm et al. 2010, 2011, 2013; Pelkmans et al. 2017); and the monokaryotic hom2con strain (Pelkmans et al. 2017). The latter strain, that expresses a constitutive active version of the Hom2 transcription factor, forms flask-like fruiting bodies that do not expose their spore forming gills when exposed to mushroom inducing conditions (presence of blue light and ambient CO2). We analysed the volatilomes of these S. commune strains with various phenotypes during different developmental stages using non-invasive SPME–GC–MS approaches to evaluate the effects of FRGs on volatile formation and to highlight fruiting-specific VOCs.
Material and methods
Fungal strains and growth conditions
The S. commune wild-type dikaryon H4-8; the derived dikaryotic deletion strains Δfst4Δfst4, Δtea1Δtea1, Δwc-2Δwc-2 and Δhom2Δhom2 (Ohm et al. 2010, 2011, 2013; Pelkmans et al. 2017); and the monokaryotic hom2con strain (Pelkmans et al. 2017) were routinely grown at 24 °C in the dark on Petri dishes containing minimal medium (MM) with 2.2% glucose and 1.5% agar (van Peer et al. 2009). For investigation of fungal growth and GC–MS analysis, S. commune strains were each grown in triplicates at 24 °C in the dark in modified crystallizing dishes (lower dish: 70 mm in diameter, upper dish: 80 mm in diameter; glass pipe attached to the upper dish: outer diameter 16 mm, inner diameter 14 mm) with 16 ml MM containing 2.2% glucose and 1.5% agar (van Peer et al. 2009) and sealed with Parafilm (for a picture of the setup, see Orban et al. (2019)). For S. commune wild-type strain H4-8, two different cultivation processes were applied. For the first approach (WT1), the mycelia were cultivated for 8 days after inoculation in the dark as described above. The aeration was then enabled by removing the Parafilm, and the samples were transferred to a climate chamber RUMED 3501 (Rubarth Apparate GmbH, Laatzen, Germany) (24 °C, 95% relative humidity (RH), 12/12 h day/night rhythm) and cultured on glass plates for a further 16 days. For the second approach (WT2), aeration and illumination of the cultures were already applied at day 2, and samples were cultured on glass plates for a further 22 days in the climate chamber. The S. commune strains hom2con, Δfst4Δfst4, Δwc-2Δwc-2, Δhom2Δhom2 and Δtea1Δtea1 were cultivated accordingly to the second approach with the exception that the latter was cultivated for only 14 days in the climate chamber.
Analysis of VOCs by SPME–GC–MS
VOCs were collected by SPME using a divinylbenzene-carboxen-polydimethylsiloxane (50/30 μm DVB/CAR/PDMS) fibre (Agilent Technologies, Santa Clara, CA, USA). Beginning with the transfer of the samples to the climate chamber (see above), VOCs were extracted directly in the HS of the crystallizing dishes for 1 h. This extraction was carried out every second day. For GC–MS analysis, an Agilent Technologies 7890A gas chromatograph equipped with Agilent VF-WAXms column (30 m × 0.25 mm, 0.25 μm) and connected to an Agilent 5975C MSD Triple-Axis mass spectrometer was used. Helium was used as a gas carrier, with a flow rate of 1.2 ml min−1. Mass spectra were acquired in the mass range of 33–300 m/z. Ionization was done by electron impact at 70 eV with an ion source temperature set at 230 °C. The SPME fibre was inserted into the injector of the gas chromatograph for thermal desorption in splitless mode for 1 min, with the injector temperature held at 250 °C. The GC oven temperature was programmed to ramp from 40 °C (hold for 3 min) to 240 °C (hold for 7 min) at 5 °C min−1. Only VOCs were considered that were not found in the MM agar controls. VOCs were identified initially by comparing obtained mass spectra with data from the NIST14 database (National Institute of Standards and Technology, Gaithersburg, MD, USA). Furthermore, linear retention indices of VOCs were determined using a C7–C30 alkane mix solved in hexane (100 µg mL−1 each component), of which 1 µL was injected in the GC–MS and analysed with a 1:10 split. Obtained retention indices were compared to published ones or the NIST database. In case of 3-methylfuran, dimethyl trisulfide, 2-methylpropan-1-ol, 3-methylbutan-1-ol, oct-1-en-3-ol, oct-2-en-1-ol, octan-3-one, oct-1-en-3-one, methyl 2-methylbutanoate, ethyl 2-methylbutanoate, α-pinene, β-pinene, β-myrcene, viridiflorene, β-bisabolene and (E)-nerolidol, VOC structures were additionally verified by comparison of retention time and mass spectra with authentic standards. Additionally, certain sesquiterpenes were verified using essential oils with known terpene compositions (Agger et al. 2009; Lopez-Gallego et al. 2010): cedrela woods oil (δ-cadinene, L-calamenene) and cubeb oil (α-cubebene, α-copaene, β-cubebene, germacrene D, cadina-1,4-diene). Nine VOCs were tentatively identified as sesquiterpenes, taking their masses and fragmentation patterns into account (Supplementary Fig. S1). However, considering the retention indices and the results from the comparison with the NIST database, for these VOCs, no satisfying matches were found. Hence, they were regarded as not identified sesquiterpenes (n. i. ST).
Data processing
Only VOCs were considered being found in at least two of the triplicates. For the heatmaps, the peak areas of identified substances were determined and used to calculate the relative amount of the respective VOC. Therefore, each substance of every sample was considered individually, meaning that 100% is related to the highest peak area of the compound measured in a sample during the experiment. The mean value of the relative VOC amounts of the triplicates was calculated for each substance and, afterwards, normalized to 100%. This approach enabled an appropriate overview about the connections between developmental stages and VOC patterns in the HS, whereas a direct comparison of the peak areas is generally difficult due to the lack of a reference quantity. The normalized data were used to generate heatmaps with OriginPro 2020b (OriginLab Corporation, Northampton, MA, USA). For principal component analysis (PCA), each sample was checked for the presence of a specific VOC at each sample time point, and the data was summarized showing if a certain VOC was present during any of the stages or not. This means that the PCA data for a sample is not limited to one time point but represents the volatilomes of all stages. PCA of the volatilome data was carried out with OriginPro 2020b.
Results
Mycelial growth and fruiting body development in S. commune
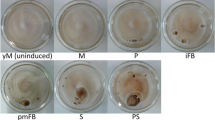
To analyse how mycelium and fruiting body development affect the volatilome of the dikaryotic S. commune wild-type strain H4-8, we chose two different cultivation processes. For the first approach (WT1), the mycelia were cultivated for eight days after inoculation in the dark sealed with Parafilm. The aeration was then enabled by removing the Parafilm, and the samples were exposed to a light day/night rhythm. For the second approach (WT2), aeration and illumination of the cultures were already applied at day 2. WT1 led to pronounced growth of mycelia (Fig. 1A). Around day 11, the mycelia developed into hyphal knots, and beginning with day 14, a further development into few but large fruiting bodies was observed (Fig. 1A). Different developmental stages were simultaneously present in the samples, including hyphal knots, primordia and young and mature fruiting bodies. In contrast, WT2 resulted in development of many small fruiting bodies (Fig. 1B) starting to appear already around day 8. Accordingly, these cultures showed less mycelium and more fruiting bodies compared to WT1 (Fig. 1A, B). Moreover, mycelia of WT2 showed a more irregular growth than that of WT1 samples. All S. commune mutant strains were analysed using the second cultivation approach to increase the chances of fructification. Strain hom2con grew more regular and slower than wild-type mycelia using the WT2 approach and started to develop bulging fruiting bodies around day 10 that only in some cases slightly began to expose their gills at late stages around day 20 (Fig. 1C). The Δfst4Δfst4 dikaryon showed, like the WT2 mycelium, irregular growth but it grew faster (Fig. 1D). In contrast to previous reports (Ohm et al. 2010; Pelkmans et al. 2017), Δfst4Δfst4 started around day 16 to develop spherical, fruiting body-like structures that elongated and branched with ongoing cultivation (Fig. 1D, Supplementary Fig. S2). The absence of fruiting body formation by Δfst4Δfst4 is likely explained by the lower light intensity that was used in the past showing that it can be important to apply different cultivation conditions to investigate the growth potential of fungal (deletion) strains. The Δtea1Δtea1 dikaryon mycelia showed a radial growth of aerial mycelia with irregular surface that grew faster than WT2 mycelia (Fig. 1E), but the few clusters of fruiting bodies that were observed previously (Pelkmans et al. 2017) did not develop. The Δwc-2Δwc-2 samples formed dense and symmetrical mycelia without formation of fruiting bodies (Fig. 1F). The Δhom2Δhom2 dikaryon showed the fastest growth of all samples covering the plates already at day 6 with thin, symmetrically growing mycelia, which formed aerial hyphae especially at the outer edge of the samples (Fig. 1G). No formation of fruiting bodies was observed for Δhom2 Δhom2 samples.
Morphology and fruiting body formation of S. commune wild-type samples and derived mutants. Samples were grown at 24 °C in the dark in modified crystallizing dishes with MM and sealed with Parafilm. Except for WT1 (A, details see below), the Parafilm was removed 2 days after inoculation, and the samples were transferred to a climate chamber (24 °C, 95% rH, 12/12 h day/night rhythm) and cultured a further 22 days. A Wild-type dikaryon H4-8 (WT1) with mycelia cultivated for eight days after inoculation in the dark sealed with Parafilm. Afterwards, aeration and light exposure were applied as described above and samples were cultivated for a further 16 days. B Wild-type dikaryon H4-8 (WT2). C Monokaryon hom2.con. D Dikaryon Δfst4 Δfst4. E Dikaryon Δtea1 Δtea1. F Dikaryon Δwc-2 Δwc-2. G Dikaryon Δhom2 Δhom2
VOCs in the HS of S. commune
By cultivating the S. commune wild type using two different approaches, one with pronounced mycelial growth as well as fruiting body development (WT1) and another resulting in early and pronounced fruiting body growth (WT2), we wanted to highlight fruiting-specific VOCs. For WT1, alcohols and esters as well as 3-methylfuran were the dominant VOCs in the HS of mycelial stages between day 8 and 12 decreasing with ongoing cultivation and with methyl 2-methylbutanoate being the most abundant substance (~ 70% of the total peak area at early stages) (Fig. 2). Beginning with the appearance of fruiting bodies at day 14, various sesquiterpenes were detected in the HS of WT1 peaking at day 18 with development of mature fruiting bodies (Fig. 2). At these late stages, α-guaiene, γ-gurjunene and δ-cadinene were the most dominant VOCs in the HS (Supplementary Fig. S3). Interestingly, some sesquiterpenes such as viridiflorene, germacrene D and cadina-1,4-diene were only detectable for a short period around day 18 whereas others, including α-guaiene, γ-gurjunene and chamigrene, remained present in the HS throughout all following sampling days (Fig. 2).
VOCs of the dikaryotic wild type strain S. commune H4-8 cultivated accordingly to the first cultivation approach (WT1). Hyphal knots appeared around day 11, at day 12 first primordia grew and first mature fruiting bodies developed about day 16. Shown is the development of the relative amounts of VOCs between days 8 and 24 after inoculation. The percent disclosure refers to each substance individually, meaning that 100% is related to the highest peak area of the compound measured during the experiment. Samples were grown in triplicates on MM at 24 °C in the dark in modified crystallizing dishes sealed with Parafilm. Eight days after inoculation, the Parafilm was removed, and the samples were transferred to a climate chamber (24 °C, 95% rH, 12/12 h day/night rhythm) and cultured for a further 16 days. VOCs were collected by SPME from day 8 onwards: volatiles were absorbed directly from the HS of the crystallizing dish for 1 h and analysed by GC–MS. n. i. ST: not identified sesquiterpene
Comparable with WT1, the HS of early developmental stages between days 4 and 8 of WT2 was dominated by alcohols and esters. At these stages, methyl 2-methylbutanoate was again the most abundant VOC (~ 68% of the total peak area) (Fig. 3A). It seems that these VOCs are associated with mycelial growth. Oct-1-en-3-ol and several sesquiterpenes appeared in the HS beginning with the development of fruiting bodies around day 8 and peaked at day 12 during maturation of fruiting bodies with γ-gurjunene and α-guaiene being the most abundant VOCs with ~ 34% and ~ 25% of the total peak area. Comparable to WT1, these sesquiterpenes remained present in the HS of the samples with ongoing cultivation. However, in contrast to WT1, remarkably less different sequiterpenes were detected in the HS. Considering the less pronounced mycelial growth of WT2, it could be that certain sesquiterpenes, such as α-guaiene, γ-gurjunene and chamigrene, are formed in fruiting bodies, whereas others, including α-cubebene, α-copaene and viridiflorene, might require in addition to mature fruiting bodies the presence of mycelium. To further investigate the connections between fungal growth and VOC formation, we analysed VOCs of S. commune deletion mutants with different phenotypes.
VOCs of fruiting body forming S. commune strains cultivated accordingly to the second cultivation approach: wild type strain H4-8 (WT2, A), monokaryotic transformant hom2con (B) and dikaryotic deletion strain ∆fst4∆fst4 (C). For WT2, at day 5, first primordia appeared developing into fruiting bodies around day 8, and first mature fruiting bodies developed about day 10. For hom2con, primordia grew around day 7, developing into mature fruiting bodies around day 18. For ∆fst4∆fst4, primordia appeared around day 14, developing into spherical, fruiting body like structures with ongoing cultivation. The heatmaps show the development of the relative amounts of VOCs between days 2 and 24 after inoculation. The per cent disclosure refers to each substance individually, meaning that 100% is related to the highest peak area of the compound measured during the experiment. Samples were grown in triplicates on MM at 24 °C in the dark in modified crystallizing dishes sealed with Parafilm. Two days after inoculation, the Parafilm was removed and the samples were transferred to a climate chamber (24 °C, 95% rH, 12/12 h day/night rhythm) and cultured for a further 22 days. VOCs were collected by SPME from day 2 onwards: volatiles were absorbed directly from the HS of the crystallizing dish for 1 h and analysed by GC–MS. n. i. ST: not identified sesquiterpene
In early stages of hom2con between days 2 and 8, alcohols and esters, including 2-methylpropan-1-ol, 3-methylbutan-1-ol and methyl 2-methylbutanoate, dominated the volatilomes, and their amounts decreased with ongoing fruiting body development (Fig. 3B). Like for WT2, sesquiterpenes, such as α-guaiene, γ-gurjunene and chamigrene, were detected beginning with the appearance of fruiting bodies at day 10 matching the assumption that these sesquiterpenes are formed in fruiting bodies (Fig. 3B). The amounts of the sesquiterpenes successively increased along with the further development of the fruiting bodies and peaked around day 22. However, despite showing pronounced mycelial growth and the presence of fruiting bodies, certain sesquiterpenes found in WT1, like α-cubebene, were not detected in hom2con. This could be related to the fact that the monokaryotic hom2con strain is able to form fruiting bodies, but these are not fully developed in contrast to the wild type.
The dikaryotic ∆fst4∆fst4, able to develop rudimentary fruiting bodies, showed remarkable differences in its VOCs profiles compared with WT1, WT2 and hom2con (Figs. 1D and 3C). Like for wild-type samples, early mycelial stages from days 4 to 8 were dominated by esters, including methyl 2-methylbutanoate (most abundant substance at these stages with ~ 70% of the total peak area), ethyl 2-methylbutanoate, isobutyl 2-methylpropionate and 2-methylbutyl acetate. However, the last three esters were not found in the HS of the wild-type samples. Additionally, the sulphur containing VOCs dimethyl disulphide and dimethyl trisulfide could be detected in the HS of ∆fst4∆fst4 samples at days 2 and 4, but were not present in the wild-type samples or hom2con. We did not detect in ∆fst4∆fst4 certain sesquiterpenes, like α-guaiene, which were associated in WT1, WT2 and hom2con with fruiting body development, which supports the assumption that the formation of certain sesquiterpenes requires the presence of fully mature fruiting bodies. Instead, three sesquiterpenes, namely, (E)-nerolidol, L-calamenene and α-bisabolol, were present in the HS of ∆fst4∆fst4 at days 14 and 16, simultaneously with the appearance of the fruiting body structures of which the last two were also detected in WT1 (Figs. 2 and 3C).
Comparable to VOC patterns of ∆fst4∆fst4, esters (mainly methyl 2-methylbutanoate, ~ 80% of the total peak area) and dimethyl trisulfide could be detected in the HS of early stages, mainly between days 2 and 6, of the S. commune deletion strains ∆wc-2∆wc-2 and ∆hom2∆hom2 (Fig. 4A, B). For ∆wc-2∆wc-2, the sesquiterpenes β-bisabolene, δ-cadinene and L-calamenene could be detected in late stages around day 22 (Fig. 4A), whereas for ∆hom2∆hom2 δ-cadinene, (E)-nerolidol and α-bisabolol were detectable around day 12 (Fig. 4B). For ∆tea1∆tea1, no sesquiterpenes could be detected in the HS of samples (Supplementary Fig. S4). However, at days 2 and 4, the monoterpenes β-myrcene and α-terpineol were present in the HS of ∆tea1∆tea1 of which the latter could not be found in wild-type samples (Supplementary Fig. S4). Taking into account that β-bisabolene, δ-cadinene, L-calamenene, (E)-nerolidol and α-bisabolol were present in the HS of S. commune strains unable to develop fruiting bodies, it seems that the formation of these sesquiterpenes, in contrast to others mentioned above, does not require the presence of fruiting bodies.
VOCs of S. commune strains unable to form fruiting bodies and cultivated accordingly to the second cultivation approach: dikaryotic deletion strain ∆wc-2∆wc-2 (A) and dikaryotic deletion strain ∆hom2∆hom2 (B). Shown is the development of the relative amounts of VOCs between days 2 and 24 after inoculation. The per cent disclosure refers to each substance individually, meaning that 100% is related to the highest peak area of the compound measured during the experiment. Samples were grown in triplicates on MM at 24 °C in the dark in modified crystallizing dishes sealed with Parafilm. Two days after inoculation, the Parafilm was removed and the samples were transferred to a climate chamber (24 °C, 95% rH, 12/12 h day/night rhythm) and cultured for a further 22 days. VOCs were collected by SPME from day 2 onwards: volatiles were absorbed directly from the HS of the crystallizing dish for 1 h and analysed by GC–MS. n. i. ST: not identified sesquiterpene
To further highlight the differences in the volatilomes, a principal component analysis (PCA) was carried out for the VOC composition of the different S. commune (Fig. 5). The first two principal components covered ~ 62% of the data’s original variation and clustered roughly into two main groups consisting of strains able to form fruiting bodies and the ones unable to do so (Fig. 5). Despite being able to develop fruiting body like structures, the ∆fst4∆fst4 dikaryon samples clustered with the ∆tea1∆tea1, ∆wc-2∆wc-2 and ∆hom2∆hom2 samples that only form mycelia, reflecting the lack of ∆fst4∆fst4 to form fruiting-related sesquiterpenes. Overall, the ability to develop fruiting bodies seems to have a tremendous effect on the volatilomes of S. commune.
Principal component analysis (PCA) for volatilome data of S. commune strains. A dot represents the data of one sample of the triplicates. Each sample was checked for the presence of a specific VOC at each sample time point, and the data was summarized showing if a certain VOC was present during any of the stages or not. This means that the PCA data for a sample is not limited to one time point but represents the volatilomes of all stages. WT1: wild-type samples cultivated accordingly to the first cultivation approach (for details see Material and Method section). WT2: wild-type samples cultivated accordingly to the second cultivation approach
Only considering VOCs present in the HS of at least two samples of the triplicates, in total 54 VOCs were found in the HS of the tested strains during all 231 GC–MS measurements (Table 1). Based on the VOC data shown above, we classified these VOCs into three categories: (1) VOCs found in strains with and without the ability to form fruiting bodies, (2) VOCs only found in strains unable to form (proper) fruiting bodies and (3) VOCs only found in strains able to form fruiting bodies (Table 1). The first category mainly harbours esters and alcohols, which are as mentioned above typical for early mycelial stages of all samples but with only 2-methylpropan-1-ol, 3-methylbutan-1-ol, methyl 2-methylpropanoate and methyl 2-methylbutanoate being detected in the HS of all samples (Table 1). Additionally, some terpenes, namely, β-myrcene, δ-cadinene, L-calamene and α-bisabolol, were found in WT1, and certain S. commune samples unable to develop fruiting bodies showing that these terpenes do not require fruiting bodies for their formation (Table 1). The same applies to the terpenes α-terpineol, β-bisabolene and (E)-nerolidol, which could only be found in strains unable to form fruiting bodies (Table 1). Also some esters like methyl butanoate belong to this second category. Interestingly, certain C8 volatiles such as oct-2-en-1-ol and sulphur containing VOCs, namely dimethyl disulphide and dimethyl trisulfide, could also be detected in strains unable to form fruiting bodies only. In contrast, nearly half of all VOCs could only be found in samples able to develop fruiting bodies being all terpenes, mainly sesquiterpenes (Table 1). The formation of these VOCs seems to require the presence of fruiting bodies. However, due to the fact that many of these were only found in WT1, it seems that the formation also requires the mycelium and/or fully developed fruiting bodies.
Discussion
We were able to show that developmental stages highly affect volatilomes of S. commune and that the presence of fruiting bodies have a remarkable influence on the VOCs emitted by the fungus. Previous studies analysed VOC formation in S. commune only at one time point. Pimenta et al. (2017) studied the VOC composition in the mycelia of three different S. commune wild-type strains grown for 6 days on PDA agar. The VOC composition of the strains slightly differed, but was comparable to our results, alcohols such as 2-methylpropan-1-ol and 3-methyl butan-1-ol, esters including methyl 2-methylbutanoate, ethyl 2-methylbutanoate and 2-methylbutyl acetate as well as sesquiterpenes such as α-bisabolol, α-curcumene and γ-himachalene were found. Interestingly, in the HS of mycelial samples of the S. commune wild-type strain 4–39, cultivated for 7 days on CYM agar, alcohols and esters but no sesquiterpenes were found (Wirth et al. 2018). However, our study shows that the VOC composition of S. commune depends on the cultivation approach and changes during different stages, which should be taken into account for the analysis of fungal VOCs especially regarding the potential of fungi to produce certain VOCs.
Numerous studies analysing fungal VOCs were carried out including research on Calocybe gambosa (Kleofas et al. 2015), Clithocybe odora, Lentinellus cochleatus, Agaricus essettei (Rapior et al. 2002), Agaricus bisporus, Pleurotus florida, Calocybe indica (Venkateshwarlu et al. 1999), Pleurotus ostreatus (Okamoto et al. 2002), Tricholoma matsutake (Cho et al. 2008), Lentinula edodes (Chen and Ho 1986), Hygrophorus chrysodon (Ouzouni et al. 2009), Clitocybe fragrans, Hebeloma crustuliniforme, Lepista nuda, Tricholoma fracticum and Tricholoma terreum (Malheiro et al. 2013). Notwithstanding the large number of studies on fungal VOCs, the connection between fungal development and VOC formation was not assessed in detail. Additionally, studies dealing with changes of volatilomes during fungal development mainly focus on the fruiting bodies of mushrooms, such as A. bisporus (Combet et al. 2009; Cruz et al. 1997; Zawirska-Wojtasiak et al. 2007), Volvariella volvacea (Mau et al. 1997; Zhang et al. 2008), Tricholoma matsutake (Cho et al. 2006; Li et al. 2016) and Pleurotus ostreatus (Tasaki et al. 2019; Zhang et al. 2008), analysing the aroma profiles in context of food quality. In contrast, studies analysing fungal volatilomes under natural circumstances considering mycelia and fruiting bodies are rare. Recently, a comprehensive study on the volatilomes of C. aegerita strains during different stages showed that the release of VOCs is connected to fungal development (Orban et al. 2020). For C. aegerita, in the HS of early mycelial stages before primordia appeared alcohols and ketones, including oct-1en-3-ol, 2-methylbutan-1-ol and cyclopentanone, were the dominant substances. Here, we showed that in S. commune wild-type samples, the alcohols 2-methylpropan-1-ol and 3-methylbutan-1-ol as well as the structural related esters methyl 2-methylpropanoate and methyl 2-methylbutanoate were the main VOCs present in the HS during early developmental stages. Interestingly, ∆fst4∆fst4, ∆tea1∆tea1, ∆wc-2∆wc-2 and ∆hom2∆hom2 samples, all being unable to develop proper fruiting bodies, produced a remarkably higher variability of esters compared to hom2con and the wild-type samples. Hence, for S. commune, these alcohols and esters are most likely produced in the mycelium rather in the fruiting bodies. This assumption is supported by the fact that in a previous study none of these VOCs was found in fruiting bodies of S. commune (Ziegenbein et al. 2006). Several fungal volatile alcohols, aldehydes and esters are derived from amino acids. In Saccharomyces cerevisiae, 2-methylpropan-1-ol is produced by the Ehrlich pathway with valine as precursor (Dickinson et al. 2003). Accordingly, S. commune seems to produce enzymes of the Ehrlich pathway as not only 2-methylpropan-1-ol was present in all analysed samples but also the related acid, 2-methylpropanoic acid, was detected in two S. commune mutant strains. Even an esterified product of 2-methylpropan-1-ol, methyl 2-methylpropanoate, was present in all analysed strains. Besides the Ehrlich pathway compounds derived from valine, products of leucine, such as 3-methylbutan-1-ol, and isoleucine, such as methyl 2-methylbutanoate, were present as well. The relation between isoleucine and methyl 2-methylbutanoate was proven by Zhang et al. (2018) in liquid cultures of L. edodes applying stable isotope labelled precursor-feeding studies. However, further research is necessary to reveal whether the formation of amino acid derived VOCs during early mycelial stages is a common feature in fungi and if these VOCs play a biological role during fructification.
In later stages around day 18, we observed that in S. commune wild-type samples, especially counting for WT1, most of the sesquiterpenes appeared in a short period during fruiting body maturation. Simultaneously with the development of fruiting bodies, sesquiterpenes were also detected in the HS of the hom2con strain. These results are in good agreement with previous observations on C. aegerita, where especially during sporulation, sesquiterpenes were the main VOCs in the HS (Orban et al. 2020). Interestingly, also for the bracket fungi Fomitopsis pinicola and Fomes fomentarius (Fäldt et al. 1999) as well as for C. cinerea (Chaisaena 2009; Thakeow 2008), an obvious increase of sesquiterpenes was observed during sporulation. Hence, sesquiterpene release closely connected to the development of fruiting bodies could be a common feature in higher fungi of the phylum Basidiomycota. Sesquiterpenes can exhibit antimicrobial activities and may therefore protect fungi during critical developmental stages against other parasitic fungi or bacteria (Ishikawa et al. 2001; Solís et al. 2004). VOCs from S. commune, especially β-bisabolol and bisabolene, inhibit mycelial growth of P. ostreatus, Ganoderma lucidum, Flammulina velutipes and Kuehneromyces mutabilis, all fungi competing with S. commune for natural resources (Wirth et al. 2021). Additionally, the composition of sesquiterpenes differs remarkably between fungal species (Chaisaena 2009; Fäldt et al. 1999; Orban et al. 2020) predestining these VOCs, along with C8 oxylipins, for a role as species-dependent infochemicals that could be involved in the communication between different fungal species (El Ariebi et al. 2016; Hiscox and Boddy 2017; Hynes et al. 2007), between fungi and animals (Drilling and Dettner 2009; Holighaus et al. 2014; Kües et al. 2018), between fungi and bacteria (Orban et al. 2023; Wirth et al. 2021) and/or fungi and plants (Ezediokpu et al. 2022).
Regardless of the function of sesquiterpenes, the question remains, which part of the fungus, mycelium or fruiting body, is responsible for the formation of these VOCs. The deletion mutants, ∆fst4∆fst4, ∆tea1∆tea1, ∆wc-2∆wc-2 and ∆hom2∆hom2, showed remarkably less sesquiterpenes in the HS than strains able to form fruiting bodies. However, the sesquiterpenes found in the HS of these samples, namely (E)-nerolidol, δ-cadinene, L-calamenene, α-bisabolol and β-bisabolene, were not present in hom2con or WT2 that mainly formed fruiting bodies and barely mycelium. Other studies confirmed the presence of α-bisabolol and β-bisabolene in mycelial samples of S. commune (Schalchli et al. 2011; Pimenta et al. 2017), which make the mycelium likely to be the main origin of these VOCs. However, (E)-nerolidol, δ-cadinene and α-bisabolol were also found in extracts of S. commune fruiting bodies obtained by hydrodestillation, showing that these sesquiterpenes can also be formed in at least mature fruiting bodies (Ziegenbein et al. 2006). Certain sesquiterpenes, including α-guaiene, chamigrene and γ-gurjunene, were only found in the presence of fruiting bodies and a previous study confirmed the occurrence of α-guaiene in fruiting bodies of S. commune (Ziegenbein et al. 2006) leading to the assumption that these sesquiterpenes originate from fruiting bodies. Many sesquiterpenes, inter alia α-cubebene, α-copaene, β-cubebene, viridiflorene, α-himachalene and germacrene D, were only detected in the HS of WT1 samples and not in WT2 or hom2con samples. The different cultivation approaches for the S. commune wild-type strain H4-8 resulted in a more pronounced mycelial growth and less but bigger fruiting bodies in case of WT1, whereas WT2 samples developed many but smaller fruiting bodies and produced less mycelium. These alternations in the phenotype are most likely the reason for the differences in the volatilomes. Recently, combined volatilomic and transcriptomic data of C. aegerita during different developmental stages showed that, despite their release during sporulation, the origin of most sesquiterpenes in C. aegerita is probably the mycelium (Orban et al. 2020, 2021). Taking the less pronounced mycelial growth of WT2 samples into account, we can speculate that the origin of sesquiterpenes in the WT1 samples mentioned above may be the mycelium rather than the fruiting body. Also in other fungi, differences in VOC composition of different morphological structures were observed. In stipes and in the pileus of the pine-mushroom Tricholoma matsutake, VOCs like hexanal, linalool and octan-3-ol contribute differently to the overall odour impression (Cho et al. 2008). Formation of sesquiterpenes or other VOCs in different parts of the fungus with changing patterns during development could be an additional aspect of VOCs functioning as infochemicals. In case of the monokaryotic hom2con strain, the absence of certain sesquiterpenes, despite the ability to grow fruiting bodies and having pronounced mycelial growth, might be connected to an incomplete development of fruiting bodies. For C. aegerita, it was shown that monokaryotic strains, even if they can form fruiting bodies, produce much less sesquiterpenes than the dikaryon during sporulation (Orban et al. 2020). Formation of sesquiterpenes in mycelium triggered by certain developmental stages of the fruiting body, like sporulation, might be a common feature of fungi and should be considered for future studies.
As discussed above, we could show that VOC biosynthesis in S. commune depends on the developmental stage and is intertwined with fruiting body formation. However, the formation processes of fungal VOCs are in many cases still barely understood (Dickschat 2017). Even for the ubiquitously present C8 VOCs, including oct-1-en-3-ol, knowledge is still missing. It is widely accepted that C8 VOCs are derived from linoleic acid. Nonetheless, a resilient connection of further steps in the formation process involving lipoxygenases (LOXs), dioxygenases (DOXs), hydroperoxide lyases (HPLs), alcohol dehydrogenases (ADHs) and ene-reductases representing the complete C8 pathways is still missing (Chen and Wu 1984; Karrer et al. 2021; Orban et al. 2021; Tasaki et al. 2019; Teshima et al. 2022; Wanner and Tressl 1998; Wurzenberger and Grosch 1984a, b, c). The biosynthesis of fungal terpenes is comparatively well understood. The large diversity of terpenes is derived from only two precursors, dimethylallyl diphosphate and isopentenyl diphosphate, which in fungi are produced from acetyl-CoA by the mevalonate pathway (Miziorko 2011). Condensation of these two isomers results in linear molecules with different chain length: C10 geranyl diphosphate, C15 farnesyl diphosphate, C20 geranylgeranyl diphosphate and C25 geranylfarnesyl diphosphate. Dephosphorylation and cyclization reactions of these hydrocarbons, catalysed by terpene synthases, are the origin of the wide range of terpenes including sesquiterpenes (Christianson 2006). Additionally, modifications of terpenes catalysed by cytochrome P450 monooxygenases, oxidoreductases and different transferases contribute also to the high diversity of terpenes in fungi (Quin et al. 2014).
In recent years, the interest in fungal sesquiterpene synthases increased remarkably (Agger et al. 2009; Engels et al. 2011; Ichinose and Kitaoka 2018; Lopez-Gallego et al. 2010; Mischko et al. 2018; Ntana et al. 2021; Zhang et al. 2020), but the regulation of the formation of fungal sesquiterpenes and other VOCs is barely understood. In S. commune, Thn1 is a regulator of G-protein signalling, and its deletion resulted in reduced formation of aerial mycelium with dikaryotic ∆thn1∆thn1 and ∆thn1 mutants unable to form fruiting bodies (Wirth et al. 2018). Sesquiterpenes, including β-bisabolol, β-bisabolene and α-bisabolol, were found in the HS of S. commune ∆thn1 samples but not in the HS of wild-type samples. The authors therefore proposed that Thn1 negatively regulates sesquiterpene synthesis in S. commune (Wirth et al. 2018). However, the study analysed VOCs and gene expression patterns only at day 7 after inoculation. Our results show that the release of sesquiterpenes in S. commune is highly connected to the cultivation conditions and developmental stage. Hence, the observed absence of sesquiterpenes in the S. commune wild-type strain could be due to the sampling process rather than negative regulation of sesquiterpene formation by Thn1. Alternatively, alternations of the VOC patterns between wild-type strain and ∆thn1 mutant may be related to the growth differences of the mycelia and, thus, are only indirectly caused by Thn1.
Here, we analysed the effect of transcription factors on volatilomes in S. commune. To the best of our knowledge, this is the first study investigating volatilomes of basidiomycete deletion strains during different developmental stages. Deletion of the FRGs wc-2 (coding for a transcription factor involved along with WC-1 in blue light sensing (Ohm et al. 2013)), the homeodomain gene hom2 (Ohm et al. 2011; Pelkmans et al. 2017) and the TEA/ATTS transcription factor gene tea1 (Pelkmans et al. 2017) led to the formation of mycelia unable to produce fruiting bodies. In case of the zinc finger transcription factor gene fst4 (Ohm et al. 2011; Pelkmans et al. 2017), inactivation resulted in the formation of only rudimentary fruiting body structures. Accordingly, ∆wc-2∆wc-2, ∆hom2∆hom2, ∆fst4∆fst4 and ∆tea1∆tea1 samples showed comparable VOC compositions in the HS (Table 1, Fig. 5). These results are in good agreement with previous transcriptome data showing similarities in the RNA profiles of ∆wc-2∆wc-2, ∆hom2∆hom2 and ∆fst4∆fst4 12 days after inoculation (Pelkmans et al. 2017). Expression of a constitutive active Hom2 protein, Hom2con (Pelkmans et al. 2017), as well as the wild type showed comparable VOCs in the HS but at different times. However, the release of VOCs, especially sesquiterpenes, was tightly connected to fruiting body development. Taking the strong connection between developmental stage and VOC release observed for all samples into account, it seems likely that WC-2, Hom2, Fst4 and Tea1 affect the volatilomes of S. commune indirectly by influencing fruiting rather than being direct regulators of sesquiterpene synthesis.
Data availability
All data are included in this article and in the supporting file.
References
Agger S, Lopez-Gallego F, Schmidt-Dannert C (2009) Diversity of sesquiterpene synthases in the basidiomycete Coprinus cinereus. Mol Microbiol 72:1181–1195. https://doi.org/10.1111/j.1365-2958.2009.06717.x
Arima T, Yamamoto M, Hirata A et al (2004) The eln3 gene involved in fruiting body morphogenesis of Coprinus cinereus encodes a putative membrane protein with a general glycosyltransferase domain. Fungal Genet Biol 41:805–812. https://doi.org/10.1016/j.fgb.2004.04.003
Chaisaena W (2009) Light effects on fruiting body development of wildtype in comparison to light-insensitive mutant strains of the basidiomycete Coprinopsis cinerea, grazing of mites (Tyrophagus putrescentiae) on the strains and production of volatile organic compounds during fruiting body development. Doctoral thesis, University Göttingen
Chen CC, Ho CT (1986) Identification of sulfurous compounds of Shiitake mushroom (Lentinus edodes Sing.). J Agric Food Chem 34:830–833. https://doi.org/10.1021/jf00071a016
Chen CC, Wu CM (1984) Studies on the enzymic reduction of 1-octen-3-one in mushroom (Agaricus bisporus). J Agric Food Chem 32:1342–1344. https://doi.org/10.1021/jf00126a030
Cho IH, Choi H-K, Kim Y-S (2006) Difference in the volatile composition of pine-mushrooms (Tricholoma matsutake Sing.) according to their grades. J Agric Food Chem 54:4820–4825. https://doi.org/10.1021/jf0601416
Cho IH, Namgung H-J, Choi H-K, Kim Y-S (2008) Volatiles and key odorants in the pileus and stipe of pine-mushroom (Tricholoma matsutake Sing.). Food Chem 106:71–76. https://doi.org/10.1016/j.foodchem.2007.05.047
Christianson DW (2006) Structural biology and chemistry of the terpenoid cyclases. Chem Rev 106:3412–3442. https://doi.org/10.1021/cr050286w
Combet E, Henderson J, Eastwood DC, Burton KS (2009) Influence of sporophore development, damage, storage, and tissue specificity on the enzymic formation of volatiles in mushrooms (Agaricus bisporus). J Agric Food Chem 57:3709–3717. https://doi.org/10.1021/jf8036209
Costa R, De Grazia S, Grasso E, Trozzi A (2015) Headspace-solid-phase microextraction-gas chromatography as analytical methodology for the determination of volatiles in wild mushrooms and evaluation of modifications occurring during storage. J Anal Methods Chem 2015. https://doi.org/10.1155/2015/951748
Cruz C, Noël-Suberville C, Montury M (1997) Fatty acid content and some flavor compound release in two strains of Agaricus bisporus, according to three stages of development. J Agric Food Chem 45:64–67. https://doi.org/10.1021/jf960300t
Dickinson JR, Salgado LEJ, Hewlins MJE (2003) The catabolism of amino acids to long chain and complex alcohols in Saccharomyces cerevisiae. J Biol Chem 278:8028–8034. https://doi.org/10.1074/jbc.M211914200
Dickschat JS (2017) Fungal volatiles – a survey from edible mushrooms to moulds. Nat Prod Rep 34:310–328. https://doi.org/10.1039/C7NP00003K
Drilling K, Dettner K (2009) Electrophysiological responses of four fungivorous coleoptera to volatiles of Trametes versicolor: implications for host selection. Chemoecology 19:109. https://doi.org/10.1007/s00049-009-0015-9
El Ariebi N, Hiscox J, Scriven SA et al (2016) Production and effects of volatile organic compounds during interspecific interactions. Fungal Ecol 20:144–154. https://doi.org/10.1016/j.funeco.2015.12.013
Engels B, Heinig U, Grothe T et al (2011) Cloning and characterization of an Armillaria gallica cDNA encoding protoilludene synthase, which catalyzes the first committed step in the synthesis of antimicrobial melleolides. J Biol Chem 286:6871–6878. https://doi.org/10.1074/jbc.M110.165845
Ezediokpu MN, Krause K, Kunert M et al (2022) Ectomycorrhizal influence on the dynamics of sesquiterpene release by Tricholoma vaccinum. J Fungi (basel) 8:555. https://doi.org/10.3390/jof8060555
Fäldt J, Jonsell M, Nordlander G, Borg-Karlson A-K (1999) Volatiles of bracket fungi Fomitopsis pinicola and Fomes fomentarius and their functions as insect attractants. J Chem Ecol 25:567–590. https://doi.org/10.1023/A:1020958005023
Fraatz MA, Zorn H (2011) Fungal Flavours. In: Hofrichter M (ed) Industrial applications. Springer Berlin Heidelberg, Berlin, Heidelberg, pp 249–268
Hiscox J, Boddy L (2017) Armed and dangerous – Chemical warfare in wood decay communities. Fungal Biol Rev 31:169–184. https://doi.org/10.1016/j.fbr.2017.07.001
Holighaus G, Weißbecker B, von Fragstein M, Schütz S (2014) Ubiquitous eight-carbon volatiles of fungi are infochemicals for a specialist fungivore. Chemoecology 24:57–66. https://doi.org/10.1007/s00049-014-0151-8
Hynes J, Müller CT, Jones TH, Boddy L (2007) Changes in volatile production during the course of fungal mycelial interactions between Hypholoma fasciculare and Resinicium bicolor. J Chem Ecol 33:43–57. https://doi.org/10.1007/s10886-006-9209-6
Ichinose H, Kitaoka T (2018) Insight into metabolic diversity of the brown-rot basidiomycete Postia placenta responsible for sesquiterpene biosynthesis: Semi-comprehensive screening of cytochrome P450 monooxygenase involved in protoilludene metabolism. Microb Biotechnol 11:952–965. https://doi.org/10.1111/1751-7915.13304
Ishikawa NK, Fukushi Y, Yamaji K et al (2001) Antimicrobial cuparene-type sesquiterpenes, enokipodins C and D, from a mycelial culture of Flammulina velutipes. J Nat Prod 64:932–934. https://doi.org/10.1021/np000593r
Kamada T, Sano H, Nakazawa T, Nakahori K (2010) Regulation of fruiting body photomorphogenesis in Coprinopsis cinerea. Fungal Genet Biol 47:917–921. https://doi.org/10.1016/j.fgb.2010.05.003
Karrer D, Gand M, Rühl M (2021) Expanding the biocatalytic toolbox with a new type of ene/yne-reductase from Cyclocybe aegerita. ChemCatChem 13:2191–2199. https://doi.org/10.1002/cctc.202002011
Kinugawa K, Suzuki A, Takamatsu Y et al (1994) Effects of concentrated carbon dioxide on the fruiting of several cultivated basidiomycetes (II). Mycoscience 35:345–352. https://doi.org/10.1007/BF02268504
Kleofas V, Popa F, Niedenthal E et al (2015) Analysis of the volatilome of Calocybe gambosa. Mycol Progress 14:93. https://doi.org/10.1007/s11557-015-1117-0
Knabe N, Jung E-M, Freihorst D et al (2013) A central role for Ras1 in morphogenesis of the basidiomycete Schizophyllum commune. Eukaryot Cell 12:941–952. https://doi.org/10.1128/EC.00355-12
Kües U (2000) Life history and developmental processes in the basidiomycete Coprinus cinereus. Microbiol Mol Biol Rev 64:316–353
Kües U, Liu Y (2000) Fruiting body production in basidiomycetes. Appl Microbiol Biotechnol 54:141–152. https://doi.org/10.1007/s002530000396
Kües U, Navarro-González M (2015) How do Agaricomycetes shape their fruiting bodies? 1. Morphological aspects of development. Fungal Biol Rev 29:63–97. https://doi.org/10.1016/j.fbr.2015.05.001
Kües U, Khonsuntia W, Subba S, Dörnte B (2018) Volatiles in communication of Agaricomycetes. In: Anke T, Schüffler A (eds) Physiology and genetics: Selected basic and applied aspects. Springer International Publishing, Cham, pp 149–212
Kuratani M, Tanaka K, Terashima K et al (2010) The dst2 gene essential for photomorphogenesis of Coprinopsis cinerea encodes a protein with a putative FAD-binding-4 domain. Fungal Genet Biol 47:152–158. https://doi.org/10.1016/j.fgb.2009.10.006
Li Q, Zhang L, Li W et al (2016) Chemical compositions and volatile compounds of Tricholoma matsutake from different geographical areas at different stages of maturity. Food Sci Biotechnol 25:71–77. https://doi.org/10.1007/s10068-016-0010-1
Liu Y, Srivilai P, Loos S et al (2006) An essential gene for fruiting body initiation in the basidiomycete Coprinopsis cinerea is homologous to bacterial cyclopropane fatty acid synthase genes. Genetics 172:873–884. https://doi.org/10.1534/genetics.105.045542
Lopez-Gallego F, Agger SA, Pella DA et al (2010) Sesquiterpene synthases Cop4 and Cop6 from Coprinus cinereus: Catalytic promiscuity and cyclization of farnesyl pyrophosphate geometrical isomers. ChemBioChem 11:1093–1106. https://doi.org/10.1002/cbic.200900671
Malheiro R, Guedes de Pinho P, Soares S et al (2013) Volatile biomarkers for wild mushrooms species discrimination. Food Res Int 54:186–194. https://doi.org/10.1016/j.foodres.2013.06.010
Mau J-L, Chyau C-C, Li J-Y, Tseng Y-H (1997) Flavor compounds in straw mushrooms Volvariella volvacea harvested at different stages of maturity. J Agric Food Chem 45:4726–4729. https://doi.org/10.1021/jf9703314
Mischko W, Hirte M, Fuchs M et al (2018) Identification of sesquiterpene synthases from the Basidiomycota Coniophora puteana for the efficient and highly selective β-copaene and cubebol production in E. coli. Microb Cell Factories 17:164. https://doi.org/10.1186/s12934-018-1010-z
Miziorko HM (2011) Enzymes of the mevalonate pathway of isoprenoid biosynthesis. Arch Biochem Biophys 505:131–143. https://doi.org/10.1016/j.abb.2010.09.028
Muraguchi H, Kamada T (1998) The ich1 gene of the mushroom Coprinus cinereus is essential for pileus formation in fruiting. Development 125:3133–3141
Muraguchi H, Fujita T, Kishibe Y et al (2008) The exp1 gene essential for pileus expansion and autolysis of the inky cap mushroom Coprinopsis cinerea (Coprinus cinereus) encodes an HMG protein. Fungal Genet Biol 45:890–896. https://doi.org/10.1016/j.fgb.2007.11.004
Murata Y, Fujii M, Zolan ME, Kamada T (1998) Molecular analysis of pcc1, a gene that leads to A-regulated sexual morphogenesis in Coprinus cinereus. Genetics 149:1753–1761
Ntana F, Bhat WW, Johnson SR et al (2021) A sesquiterpene synthase from the endophytic fungus Serendipita indica catalyzes formation of viridiflorol. Biomolecules 11:898. https://doi.org/10.3390/biom11060898
Oberwinkler F (1982) The significance of the morphology of the basidium in the phylogeny of basidiomycetes. In: Wells K, Wells EK (eds) Basidium and basidiocarp: Evolution, cytology, function, and development. Springer, New York, NY, pp 9–35
Ohm RA, de Jong JF, Lugones LG et al (2010) Genome sequence of the model mushroom Schizophyllum commune. Nat Biotech 28:957–963. https://doi.org/10.1038/nbt.1643
Ohm RA, de Jong JF, de Bekker C et al (2011) Transcription factor genes of Schizophyllum commune involved in regulation of mushroom formation. Mol Microbiol 81:1433–1445. https://doi.org/10.1111/j.1365-2958.2011.07776.x
Ohm RA, Aerts D, Wösten HAB, Lugones LG (2013) The blue light receptor complex WC-1/2 of Schizophyllum commune is involved in mushroom formation and protection against phototoxicity. Environ Microbiol 15:943–955. https://doi.org/10.1111/j.1462-2920.2012.02878.x
Okamoto K, Narayama S, Katsuo A et al (2002) Biosynthesis of p-anisaldehyde by the white-rot basidiomycete Pleurotus ostreatus. J Biosci Bioeng 93:207–210. https://doi.org/10.1016/S1389-1723(02)80015-9
Orban A, Fraatz MA, Rühl M (2019) Aroma profile analyses of filamentous fungi cultivated on solid substrates. In: Steudler S, Werner A, Cheng JJ (eds) Solid state fermentation: Research and industrial applications. Springer International Publishing, Cham, pp 85–107
Orban A, Hennicke F, Rühl M (2020) Volatilomes of Cyclocybe aegerita during different stages of monokaryotic and dikaryotic fruiting. Biol Chem 401:995–1004. https://doi.org/10.1515/hsz-2019-0392
Orban A, Weber A, Herzog R et al (2021) Transcriptome of different fruiting stages in the cultivated mushroom Cyclocybe aegerita suggests a complex regulation of fruiting and reveals enzymes putatively involved in fungal oxylipin biosynthesis. BMC Genomics 22:324. https://doi.org/10.1186/s12864-021-07648-5
Orban A, Jerschow JJ, Birk F et al (2023) Effect of bacterial volatiles on the mycelial growth of mushrooms. Microbiol Res 266:127250. https://doi.org/10.1016/j.micres.2022.127250
Ouzouni PK, Koller W-D, Badeka AV, Riganakos KA (2009) Volatile compounds from the fruiting bodies of three Hygrophorus mushroom species from Northern Greece. Int J Food Sci Technol 44:854–859. https://doi.org/10.1111/j.1365-2621.2009.01919.x
Pelkmans JF, Patil MB, Gehrmann T et al (2017) Transcription factors of Schizophyllum commune involved in mushroom formation and modulation of vegetative growth. Sci Rep 7:310. https://doi.org/10.1038/s41598-017-00483-3
Pimenta L, Ferreira MA, Pedroso MP et al (2017) Wood-associated fungi produce volatile organic compounds toxic to root-knot nematode. Scientia Agricola 74:303–310. https://doi.org/10.1590/1678-992x-2015-0472
Quin MB, Flynn CM, Schmidt-Dannert C (2014) Traversing the fungal terpenome. Nat Prod Rep 31:1449–1473. https://doi.org/10.1039/C4NP00075G
Rapior S, Breheret S, Talou T et al (2002) The anise-like odor of Clitocybe odora, Lentinellus cochleatus and Agaricus essettei. Mycologia 94:373–376. https://doi.org/10.1080/15572536.2003.11833201
Rühl V, Lotz-Winter H, Neuss A et al (2018) Comprehensive analysis of the volatilome of Scytinostroma portentosum. Mycol Progress 17:417–424. https://doi.org/10.1007/s11557-017-1367-0
Sánchez-García M, Ryberg M, Khan FK et al (2020) Fruiting body form, not nutritional mode, is the major driver of diversification in mushroom-forming fungi. PNAS. https://doi.org/10.1073/pnas.1922539117
Schalchli H, Hormazabal E, Becerra J et al (2011) Antifungal activity of volatile metabolites emitted by mycelial cultures of saprophytic fungi. Chem Ecol 27:503–513. https://doi.org/10.1080/02757540.2011.596832
Solís C, Becerra J, Flores C et al (2004) Antibacterial and antifungal terpenes from Pilgerodendron uviferum (D. Don) Florin. J Chil Chem Soc 49:157–161. https://doi.org/10.4067/S0717-97072004000200010
Tasaki Y, Kobayashi D, Sato R et al (2019) Variations in 1-octen-3-ol and lipoxygenase gene expression in the oyster mushroom Pleurotus ostreatus according to fruiting body development, tissue specificity, maturity, and postharvest storage. Mycoscience 60:170–176. https://doi.org/10.1016/j.myc.2019.02.005
Terashima K, Yuki K, Muraguchi H et al (2005) The dst1 gene involved in mushroom photomorphogenesis of Coprinus cinereus encodes a putative photoreceptor for blue light. Genetics 171:101–108. https://doi.org/10.1534/genetics.104.040048
Teshima T, Funai R, Nakazawa T, et al (2022) Coprinopsis cinerea dioxygenase is an oxygenase forming 10(S)-hydroperoxide of linoleic acid, essential for mushroom alcohol, 1-octen-3-ol synthesis. J Biol Chem 102507. https://doi.org/10.1016/j.jbc.2022.102507
Thakeow P (2008) Development of a basic biosensor system for wood degradation using volatile organic compounds. Doctoral thesis, University Gottingen
Turner EM (1977) Development of excised sporocarps of Agaricus bisporus and its control by CO2. Trans Br Mycol Soc 69:183–186. https://doi.org/10.1016/S0007-1536(77)80035-1
van Peer AF, de Bekker C, Vinck A et al (2009) Phleomycin increases transformation efficiency and promotes single integrations in Schizophyllum commune. Appl Environ Microbiol 75:1243–1247. https://doi.org/10.1128/AEM.02162-08
Venkateshwarlu G, Chandravadana MV, Tewari RP (1999) Volatile flavour components of some edible mushrooms (basidiomycetes). Flavour Fragr J 14:191–194. https://doi.org/10.1002/(SICI)1099-1026(199905/06)14:3%3c191::AID-FFJ810%3e3.0.CO;2-7
Wanner P, Tressl R (1998) Purification and characterization of two enone reductases from Saccharomyces cerevisiae. Eur J Biochem 255:271–278. https://doi.org/10.1046/j.1432-1327.1998.2550271.x
Wessels JGH (1993) Fruiting in the higher fungi. In: Rose AH (ed) Advances in Microbial Physiology. Academic Press, pp 147–202
Wirth S, Kunert M, Ahrens L-M et al (2018) The regulator of G-protein signalling Thn1 links pheromone response to volatile production in Schizophyllum commune. Environ Microbiol 20:3684–3699. https://doi.org/10.1111/1462-2920.14369
Wirth S, Krause K, Kunert M et al (2021) Function of sesquiterpenes from Schizophyllum commune in interspecific interactions. PLOS ONE 16:e0245623. https://doi.org/10.1371/journal.pone.0245623
Wurzenberger M, Grosch W (1984a) Stereochemistry of the cleavage of the 10-hydroperoxide isomer of linoleic acid to 1-octen-3-ol by a hydroperoxide lyase from mushrooms (Psalliota bispora). Biochim Biophys Acta (BBA) - Lipids Lipid Metab 795:163–165. https://doi.org/10.1016/0005-2760(84)90117-6
Wurzenberger M, Grosch W (1984b) The formation of 1-octen-3-ol from the 10-hydroperoxide isomer of linoleic acid by a hydroperoxide lyase in mushrooms (Psalliota bispora). Biochim Biophys Acta Acta (BBA) - Lipids Lipid Metab 794:25–30. https://doi.org/10.1016/0005-2760(84)90293-5
Wurzenberger M, Grosch W (1984c) Origin of the oxygen in the products of the enzymatic cleavage reaction of linoleic acid to 1-octen-3-ol and 10-oxo-trans-8-decenoic acid in mushrooms (Psalliota bispora). Biochim Biophys Acta Acta (BBA) – Lipids Lipid Metab 794:18–24. https://doi.org/10.1016/0005-2760(84)90292-3
Zawirska-Wojtasiak R, Siwulski M, Wasowicz E, Sobieralski K (2007) Volatile compounds of importance in the aroma of cultivated mushrooms Agaricus bisporus at different conditions of cultivation. Polish J Food Nutr Sci 57:367–372
Zhang Z-M, Wu W-W, Li G-K (2008) A GC—MS study of the volatile organic composition of straw and oyster mushrooms during maturity and its relation to antioxidant activity. J Chromatogr Sci 46:690–696. https://doi.org/10.1093/chromsci/46.8.690
Zhang Y, Fraatz MA, Birk F et al (2018) Enantiomeric ratios of 2-methylbutanoic acid and its methyl ester: Elucidation of novel biogenetic pathways towards (R)-methyl 2-methylbutanoate in a beverage fermented with shiitake. Food Chem 266:475–482. https://doi.org/10.1016/j.foodchem.2018.06.027
Zhang C, Chen X, Orban A et al (2020) Agrocybe aegerita serves as a gateway for identifying sesquiterpene biosynthetic enzymes in higher fungi. ACS Chem Biol 15:1268–1277. https://doi.org/10.1021/acschembio.0c00155
Ziegenbein FC, Hanssen H-P, König WA (2006) Chemical constituents of the essential oils of three wood-rotting fungi. Flavour Fragr J 21:813–816. https://doi.org/10.1002/ffj.1732
Acknowledgements
We thank both reviewers for their helpful comments and suggestions.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was financially supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – funder Id: https://doi.org/10.13039/501100001659, grant number: RU 2137/1–1.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Lisa Janaina Schwab and Axel Orban carried out experiments and analysed data. The first draft of the manuscript was written by Axel Orban and Martin Rühl. All authors commented on the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Marco Thines
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Orban, A., Schwab, L.J., Lugones, L.G. et al. Effect of fruiting-related genes on the formation of volatile organic compounds in the mushroom Schizophyllum commune. Mycol Progress 22, 67 (2023). https://doi.org/10.1007/s11557-023-01914-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-023-01914-4