Abstract
A combined morphological and molecular genetic study of the European species within the /Rhombisporum clade of the genus Entoloma reveals a high species diversity. This group comprises typical grassland species with pronounced and well-differentiated cheilocystidia, and a wide range of spore shapes varying from rhomboid to five-angled. To fix the concept of the classical species E. rhombisporum, a neotype is designated. Nine species are described as new to science based on the result of nrDNA ITS phylogeny with additional gap coding, and morphological characterization: E. caulocystidiatum, E. lunare, E. pararhombisporum, E. pentagonale, E. perrhombisporum, E. rhombiibericum, E. rhombisporoides, E. sororpratulense, and E. subcuboideum. The ITS sequences of the holotypes of previously described species belonging to the /Rhombisporum clade, viz., E. laurisilvae and E. pratulense have also been generated and are published here for the first time. Since many of the above-mentioned species have been misidentified as E. cocles, it seemed opportune to also study this species and to designate a neotype to fix its current concept. A key including European species is presented. As most of the species are potentially important indicators for threatened grassland communities, the 130 ITS barcodes newly generated for this study may be useful as a reference in conservation and metabarcoding projects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Kühner and Boursier (1929) described Leptonia rhombispora, a new entolomatoid species from a subalpine meadow in the French Alps, with rather peculiar spores with a more or less rhomboid outline; and it was incorporated in the famous Flore analytique des Champignons supérieurs de France (Kühner and Romagnesi 1953). Since then, this species has been accepted in mycological literature (e.g., Horak 1976; Noordeloos 1980, 1987, 1992). Rhodophyllus prismatospermus, another species with more or less similar spores, was added by Romagnesi (1974), and Noordeloos & Wölfel (Noordeloos 1987) described a variety of E. rhombisporum with cystidia all over the length of the stipe as E. rhombisporum var. floccipes. The current phylogenetic study based on the nrDNA ITS region revealed that the European species with rhomboid spores cluster together in a well-supported clade but mixed with species with five or six-angled spores. All species, however, are characterized by the presence of well-differentiated cheilocystidia. The /Rhombisporum clade can therefore be characterized as follows: habit mycenoid or collybioid, with a conical-campanulate to convex pileus, often with a truncate-slightly depressed centre, hygrophanous or not hygrophanous, glabrous or radially fibrillose to rugulose pileus; adnate-emarginate lamellae, often with a distinct decurrent tooth (becoming sometimes arcuate-decurrent in expanded pilei); and a polished, pruinose or fibrillose stipe. Spores (irregularly) rhomboid or 5 (–6) angular in side-view; cheilocystidia always present, typically lageniform, sometimes partly clavate-mucronate to fusiform, often thin-walled and easily collapsing, sometimes, however, rather large and conspicuous, and with thickened walls. The pigment in the pileipellis is exclusively intracellular. Brilliant granules are absent. Caulocystidia are sometimes present in form of fusiform to lageniform cystidia; otherwise, no caulocystidia could be found or just some undifferentiated, subcylindrical terminal elements of the hyphae of the stipe surface. Clamp-connections are present or absent.
According to Pegler and Young (1979), the spores of E. rhombisporum are 7-faceted, and not cuboid, as they are sometimes incorrectly called. The /Rhombisporum clade, however, is phylogenetically distant from the true cuboid-spored /Cubospora clade (Karstedt et al. 2019), as well as from the Entoloma conferendum group in the /Nolanea clade with cruciform spores. The name E. cocles very often has been applied to specimens of the /Rhombisporum clade, and for that reason it has been decided to fix this species here with a neotype and give a full description. Nine new species are described in the present study which reveals the remarkable diversity in the /Rhombisporum clade. Many species treated here are characteristic for old, semi-natural grasslands, a critically endangered vegetation type all over Europe (e.g., Nitare 1988; Griffith et al. 2013), and therefore we discuss some ecological and conservational aspects.
Materials and methods
Morphological study
The base of this study is formed by material collected in the field by the authors, and the sequenced exsiccates from various institutions. Freshly collected specimens were photographed in the field, where also much attention was paid to characterize the surrounding vegetation and ecology. The material was described straight after collecting to fix the ephemeral macroscopic characters, then dried and stored. The state of the sequenced exsiccates varied, some were well-annotated, others not. All the available material was studied microscopically, either by the collectors, or by the first author (MEN) during visits to the botanic garden and herbarium in Oslo. The second author (GMJ) studied a large set of exsiccates with a Leica DMLS microscope, using a drawing tube and a Touptek Phototronics camera and a Zeiss Axiophot microscope with DC controlled Cree XP-G3 R3 CRI 90+ LED illumination, Plan Neofluar objectives 40×/1.30 Oil, 100×/1.30 Oil, differential interference contrast (DIC) and 12MP Touptek video camera with SONY Exmor IMX226 CMOS sensor, Toupview advanced video & image processing application. Spores, basidia and cystidia were observed in squash preparations of small parts of the lamellae in 5 % KOH or 1 % Congo Red in concentrated NH4OH. The pileipellis was studied by examining a radial section or a scalp in water, 5% KOH or ammonia. Basidiospore dimensions are based on observing 20 spores, and cystidia and basidia dimensions on observing at least 10 structures per collection. Basidia were measured without sterigmata, and the spores without hilum. Spore length to width ratios are reported as Q.
Molecular genetic study and phylogenetic reconstruction
DNA extraction, PCR amplifications and sequencing were performed in the Norwegian Barcode of Life (NorBOL) or followed Larsson and Jacobsson (2004), Larsson et al. (2018), Alvarado et al. (2015), Dima et al. (2016), Morozova et al. (2018), Reschke et al. (2018), and Voitk et al. (2020a). The primer pairs ITS1F/ITS4, ITS1F/ITS4B, ITS1F/ITS2 and ITS3/ITS4 (White et al. 1990; Gardes and Bruns 1993) were used to amplify and sequence the complete or partial nrDNA ITS barcoding region. Chromatograms were checked and edited with the CodonCode Aligner package (CodonCode Corp., Centerville, Massachusetts, USA). Sequence comparison with public and own databases followed Noordeloos et al. (2017). Our dataset is composed of 141 nrDNA ITS sequences, carefully selected after an initial analysis using published and all our unpublished ITS sequences (data not shown). Newly generated sequences were submitted to GenBank (Table 1). The dataset was aligned in MAFFT online v7 (http://mafft.cbrc.jp/alignment/server) choosing the E-INS-I method (Katoh and Standley 2013) with setting the scoring matrix for nucleotide sequences to 1PAM / k=2, gap opening penalty to 1.0 and offset value to 0.123. The alignment was checked and edited in SeaView 4 (Gouy et al. 2010). Based on the work of Nagy et al. (2012), phylogenetically informative indels were coded as presence/absence data with FastGap 1.2 (Borchsenius 2009) following the simple indel coding algorithm (Simmons et al. 2001). Maximum Likelihood (ML) phylogenetic reconstruction was performed in raxmlGUI (Silvestro and Michalak 2012) using rapid bootstrap analysis with 1,000 replicates. Three nucleotide partitions (ITS1, 5.8S, ITS2) were set to the GTRGAMMA substitution model in addition to one binary partition (indel characters) that was set to default.
MrBayes 3.1.2 (Ronquist and Huelsenbeck 2003) was used to perform Bayesian Inference (BI) phylogeny. The alignment was divided into four partitions (ITS1, 5.8S, ITS2, and indels): the GTR + Γ substitution model was applied to the nucleotide characters, while the two-parameter Markov model was set for the indels. Two independent runs of four Markov Chain Monte Carlo (MCMC) were performed each for 5 000 000 generations, sampling every 1000th generation. The first 35% of the trees was discarded as burn-in. For the remaining trees, a 50% majority rule consensus phylogram with posterior probabilities as nodal supports was computed. The phylogenetic trees were edited in MEGA 7 (Kumar et al. 2016) and Adobe Illustrator CS4.
Results
Phylogeny
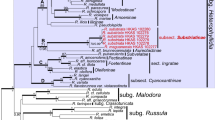
A total of 141 sequences, 130 of which belong to the /Rhombisporum clade, were included in the analyses (Table 1). The ITS alignment comprises 737 characters. After gap coding, a binary set of 103 characters was added to the nucleotide alignment. The final data matrix including nucleotide and binary data was composed of 840 characters (Supplementary Material). The /Undulatosporum clade was selected as the outgroup. Phylogenetic trees from ML and BI analyses showed congruent topologies. The best scoring ML tree is shown in Figs. 1 and 2.
Maximum likelihood tree showing phylogenetic relationships in the /Rhombisporum clade based on nrDNA ITS sequence analyses using RAxML and MrBayes. Species level clades are compressed. New species are marked in green and boldface, other European species of the clade are marked in black and boldface. Rhomboid (in blue) and pentagonal (in red) spore shapes are indicated after the species. Species with clampless hyphae are marked with crossed circles. Scale bar indicates 0.01 expected change per site per branch
Maximum Likelihood tree showing the phylogenetic relationship in the /Rhombisporum clade based on nrDNA ITS sequence analyses using RAxML and MrBayes. Sequences are labelled with their voucher numbers and geographical origin. Species treated in this study are marked in boldface. ML bootstrap values >70% as well as Bayesian posterior probabilities >0.90 are indicated above branches. Scale bar indicates 0.01 expected change per site per branch
The /Rhombisporum clade received strong to high support (MLBS=89 / BPP=1) in our analyses with Entoloma cocles placed basally. The /Rhombisporum clade consists of 16 lineages of which most of them can be regarded as species supported by phylogenetic and additional morphological evidence. The exception is E. aff. caulocystidiatum represented by a single sequence which only slightly differs from E. caulocystidiatum and needs further study. Two single branches represent North American species yet without a name. The remaining thirteen lineages represent European species of which nine are described here as new to science. All species are monophyletic and gained high statistical supports (Figs. 1 and 2), except E. caulocystidiatum which was supported only moderately by the ML analysis (MLBS=70). Entoloma rhombiibericum and E. cf. prismatospermum are represented by a single sequence each to date. The species belonging to the /Rhombisporum clade treated in this study as well as E. cocles have little intraspecific sequence variation in the nrDNA ITS region that is 0–3 substitution and/or indel differences (< 0.5%). The interspecific sequence dissimilarity towards a sister species is at least 2–7% (11–43 substitution and/or indel differences). We sequenced type specimens of three taxa (E. laurisilvae, E. pratulense, E. rhombisporum var. floccipes) and provide neotypes for E. rhombisporum and E. cocles. Altogether 130 ITS barcode sequences were newly generated for this study (Table 1).
Characters used for species delimitation
The macromorphology of the species in the clade is rather uniform: most species share a more or less conico-truncate pileus, usually with a slight umbo, a slightly to distinctly hygrophanous pileus which is indistinctly or distinctly translucently striate, and not entirely smooth, but covered with innate fibrils, that sometimes give the surface a lustrous to slightly fluffy appearance; the adnate-decurrent, arcuate lamellae, and a stipe that is often much paler than the pileus, with either a hyaline, polished or innate silvery fibrillose surface. The spores differ in size and shape, ranging from either rhomboid, 4 to 5(–6)-angled in side view, more or less isodiametrical to heterodiametrical. Cheilocystidia are always present, and generally in majority lageniform. They can be very thin-walled, slender and rather inconspicuous and easily collapsing, or robust, with relatively thick walls, and with a rather broad basal part, very obvious and protruding from the hymenium. Rarely there are also pleurocystidia, similar to cheilocystidia or substantially smaller, but usually only near the lamella edge. Some species lack clamp-connections, in other species either only the basidia have clamp-connections, or they are found in several tissues. In a search for new diagnostic characters, a very distinct character was discovered during our study: in E. pararhombisporum the stipe covering has very abundant and distinct congophilous plaques in the hyphal wall, a seemingly constant and rather striking character that was not encountered in any of other species within this clade, nor is it known from other Entoloma species or other agarics. It is therefore considered of potential diagnostic value, and supports the phylogenetically distinct position of that species.
Taxonomy
Entoloma rhombisporum (Kühner & Boursier) E. Horak, Sydowia 28(1–6): 228 (1976) [1975–1976] Fig. 3
MycoBank: MB 313808
≡ Leptonia rhombispora Kühner & Boursier, Bull. trimest. Soc. mycol. Fr. 45: 276 (1929), Rhodophyllus rhombisporus (Kühner & Boursier) Romagn., Bull. trimest. Soc. mycol. Fr. 48(3–4): 319 (1933)
Holotype: not indicated. No original material available.
Neotype (designated here): Sweden, Västernorrlands län, Kullen mountain, sheep pasture, 26 Aug 2018, K. Reschke, KaiR1207 (L-0607527). MycoBank MBT 10004483. GenBank No. OL853794 (ITS).
Etymology: rhomboid (Lat.), diamond shaped, referring to the shape of the spores.
Original description (translated from French): Pileus 15–30 mm, hemispherical to convex, with blunt, not or weakly umbilicate centre (sometimes truncate-umbilicate), hygrophanous, translucently striate when moist, sordid yellow-brown or yellowish red-brown, absolutely glabrous or even shiny, finely fibrillose with sometimes rugulose at centre. Context very thin. Smell weakly spermatic or reminiscent of onion. Lamellae, L = 19–23, l = 3, moderately distant, pinkish flesh-colour (not brown), not decurrent, ventricose, sinuate, adnate but not broadly so. Stipe 35–50 × 1.5–3.5 mm, equal, with white mycelium at base, whitish, hyaline, glabrous or slightly white pruinose at apex, almost stuffed and very fragile. Spores short, 9.2–11.5 × 8–10, average 10 × 8.7 μm, more or less cuboid or 5-angled. Basidia 4-spored. Some inconspicuous, not differentiated hairs along the lamella edge.
Description: Pileus 10–30 mm diam., convex, expanding with age, often slightly umbilicate or vaguely umbonate, slightly hygrophanous, dark yellow-brown to dark reddish brown, distinctly translucently striate at margin, sometimes up to half the radius, innately radially fibrillose, centre sometimes rugulose or fluffy. Lamellae, L = 20–36, l = 3–5, distant, variably inserted, adnate-emarginate with decurrent tooth, or arcuate-decurrent in expanded specimens, whitish then brownish pink with entire, concolorous edge. Stipe 20–60 × 1–2(–3) mm, cylindrical, very pale, almost white to pale yellow-brown, often appearing hyaline, glabrous, polished. Smell and taste not known. Basidiospores 9.0–12.0 × 7.0–10(–11) μm, av. 9.7–10.4 × 8.0–8.8 μm, Q=1.0–1.4, Qav=1.18–1.22, 4-(rarely 5–6-) angled in side-view. Basidia 36–57 × 11.5–16 μm, clavate, 4-spored, with clamp-connections. Hymenophoral trama made up of cylindrical elements, 50–280 × 5–12 μm, slightly constricted at the septa, hyaline, no clamp-connections seen. Lamella edge heterogeneous with basidia and rather thin-walled, easily collapsing cheilocystidia, not or hardly protruding from the hymenium, 35–80 × 10–25 (base) × 3–9 (apex) μm, fusiform, lageniform with a long neck (with constrictions) rarely furcate or tibiiform, with clamp-connections. Pileipellis a cutis of cylindrical hyphae, 3–10(–20) μm wide, with brown intracellular and globular pigment. Pileitrama regular, made up of 5–40 μm wide, hyaline hyphae. Stipitipellis a cutis of cylindrical hyphae, 6–10(–18) μm wide, with some loose, subcylindrical terminal elements at apex; differentiated caulocystidia absent. Clamp-connections frequent in the hymenium, elsewhere not observed.
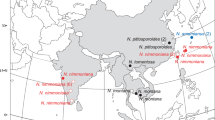
Habitat and distribution: In small groups in semi-natural grasslands and in the margin of subalpine, calcareous bogs, in boreal and subalpine regions of Europe. Distribution poorly known, due to the unexpected diversity in this group. Sequenced material is known from Norway, Sweden and Russia.
Comments: We have interpreted Entoloma rhombisporum here as closest to the original description as possible, i.e., as a species with predominantly rhomboid (4-sided) spores, and thin-walled, easily collapsing, scattered to fairly abundant lageniform cheilocystidia. In his comments on the species, Kühner in Kühner and Romagnesi (1953) added some more information that helped us to interpret this species correctly: spores 8.5–12.2 × 7–9.7 μm, seemingly 4-sided in profile and side-view (rhomboid sensu lato). Cheilocystidia are inconspicuous, but rather frequent, hidden among the basidia and not protruding from the hymenium, ventricose-fusiform or clavate-mucronate with rather blunt and short appendix, thin-walled. Stipe finely powdered at apex, downwards grey-brown, horn brown or almost white; pileus rather sordid greyish brown, sometimes with slight yellow tinge, distinctly translucently striate, blunt, subumbilicate; pigment brown, intracellular. Clamp-connections present, but not at all septa. Lamellae pallid, without grey tinge. Smell spermatic. Taste distinctly farinaceous. Praz de St Bon, Savoie, France. No type has been indicated, nor is original material preserved.
The following characters are therefore considered distinctive for E. rhombisporum: a translucently striate pileus, almost white, hyaline stipe, spores 4-angled in profile and side-view, and inconspicuous cheilocystidia. As such, the description above fits best with the original diagnosis. The interpretation of E. rhombisporum by Horak (1976), based on a collection by Favre, and accepted by Noordeloos (1980) refers to another species in the clade with a non-translucently striate pileus, yellow to brown fibrillose stipe, slightly smaller spores and conspicuous cheilocystidia, and is described below as a new species (see E. perrhombisporum). In the ITS region, E. rhombisporum differs from E. rhombiibericum and E. subcuboideum by 28 and 29 substitution and indel positions, respectively, with a similarity of 95%.
Additional specimens examined: Norway, Nordland, Grane, Holmvassdalen, on shallow soil over limestone at the edge of a rich fen, 3 Sept 2010, S. E. Eidissen, O-F-242475; Grane, Holmvassdalen, calcareous spruce forest, in moist soil (dried out pond) surrounded by low herb meadow, 29 Aug 2016, J. Lorås, JL105-16; Møre og Romsdal, Hustadvika, Fræna, Farstad, W of Storholmen, semi-natural pasture near the sea shore, 24 Sept 2004, J. B. Jordal, O-F-177991; Aure, Ertvågsdalen, calcareous semi-natural snowbed-grassland grazed by sheep, 14 Sept 2004, F. Oldervik, O-F-187955. Russia, Tver region, Zubtsov district, 2 km to SE from Mozgovo village, open place in calcareous pine forest, right bank of the Derzha river, 11 Sept 2015, O. Morozova, 963-24TV15 (LE 312533). Sweden, Västernorrlands län, Kullen mountain, sheep pasture, 26 Aug 2018, K. Reschke, KaiR1208 (L-0607528).
Entoloma perrhombisporum Noordel., Dima, J.B. Jordal & O.V. Morozova, sp. nov. Fig. 4
MycoBank: MB 842124
Holotype: Norway, Nordland, Alstahaug, Skålvær, Buøya, 17 Sept 2004, rich, semi-natural sheep pasture, D. Pettersen, A. B. Stærnes, J. B. Jordal, A. Knutsen, P. Fadnes, O-F-67220. GenBank No. OL853762 (ITS).
Etymology: Refers to the resemblance to Entoloma rhombisporum
Diagnosis: Entoloma perrhombisporum is characterized by the rather regularly shaped, mostly quadrangular spores in side-view and the rather pronounced, thick-walled cheilocystidia.
Description: Pileus 10–20 mm diam., conical slightly expanding to convex with age, with straight margin, slightly hygrophanous, not translucently striate, when moist warm yellow-brown to fulvous, darker and more grey-tinged brown at centre, pallescent from centre along radial streaks, innately finely fibrillose, slightly aeriferous. Lamellae, L = 24–32, l = 3–5, moderately distant, adnate-emarginate with long decurrent tooth, pale cream, then with pink tinge, with an entire or eroded, concolorous edge. Stipe 30–45 × 1–2 mm, cylindrical, pallid, creamy-white, almost smooth and polished, but initially with some silvery, longitudinal fibrils, apex slightly white pruinose. Smell and taste not noted. Basidiospores 8.5–11.0 × 7.0–9.8 μm, av. 9.8 × 8.4 μm, Q=1.0–1.3, Qav=1.12–1.18, rather regularly quadrangular in side-view. Basidia 40–57 × 12–15 μm, clavate, 4-spored, with clamp-connections. Cheilocystidia 40–81 × 9.5–24 (base) × 3–8 μm (apex), fusiform, clavate, lageniform, thick-walled, with clamp-connections. Hymenophoral trama 5–15 μm wide cylindrical hyphae, no clamp-connections seen. Pileipellis a cutis of thin hyphae, 3–10 μm wide with pale brown, intracellular pigment. Pileitrama regular, made up of cylindrical hyphae, 6–18 μm wide, no clamp-connections seen. Stipitipellis a cutis of 4–17 μm wide cylindrical hyphae; at apex with some subcylindrical terminal ends, no differentiated caulocystidia present. Clamp-connections present in the hymenium, elsewhere not observed.
Habitat and distribution: Terrestrial in extensively grazed meadows on calcareous soil. Sequenced collections are from the county of Nordland in Norway, and Jämtland in Sweden, in hot-spot sites with many calciphilous Entoloma species.
Comments: Entoloma perrhombisporum fits very well with the interpretation of E. rhombisporum by Horak (1976) and Noordeloos (1980), but, as discussed above, this does not agree with the protologue of that species. Entoloma perrhombisporum differs from its closest species E. caulocystidiatum by 36 substitution and indel positions in the ITS region, with a similarity of 94%.
Additional specimens examined: Norway, Nordland, Vega, Stora Buøya, rich pasture/Calluna heath, 18 Sept 2004, T. Solem, M. Gjestland, B. H. Larsen, E. Håve, E. W. Hanssen, E. Johannesen, O-F-67323. Sweden, Jämtland, Krokom, Trångsviken, Alsens-Ede, garden/semi-natural calcareous grassland, 27 Aug 2016, O. Morozova, K. & E. Bendiksen, KB&EB 21/16 (O-F-76644, LE 312541); Alsens-Ede, 27 Aug 2016, T.E. Brandrud, B. Dima TEB323-16 (O); Krokom, Jale, Trång, calcareous, dry semi-natural grassland, 27 Aug 2016, T.E. Brandrud, TEB320-16 (O).
Entoloma subcuboideum Noordel., J.B. Jordal, Vila & Dima, sp. nov. Fig. 5
MycoBank: MB 842125
Holotype: Norway, Trøndelag, Oppdal, Medlisetrene, calcareous sheep pasture, 24 Aug 2005, J. B. Jordal, JBJ-3106 (O-F-158286). GenBank No. OL853819 (ITS).
Etymology: Refers to the spores appearing partly almost cuboid.
Diagnosis: Entoloma subcuboideum is mainly characterized by its sordid greyish brown pileus, somewhat fibrillose stipe, small, rhomboid to 5-angled spores, and conspicuous cheilocystidia.
Description: Pileus 10–20 mm diam., conical, only slightly expanding to conico-convex or convex, with straight, rarely crenulate margin, slightly hygrophanous, indistinctly translucently striate, moderately to dark grey-brown, paler to the margin, radially fibrillose, rugulose at centre, sometimes with micaceous patches. Lamellae, L = 20–38, l = 3–7, moderately distant to fairly crowded, adnate-emarginate with decurrent tooth, almost free, narrowly ventricose, greyish white/pale cream, then greyish pink with subentire, concolorous edge. Stipe 20–40 × 2–3 mm, cylindrical to somewhat flattened, greyish brown, longitudinally fibrillose-striate, pruinose at apex, glabrous with age. Smell and taste indistinct. Basidiospores 7.5–11 × 7.0–10.5 μm, av. 9.0–10.5 × 7.5–8.5 μm, Q=1.0–1.4, Qav=1.15–1.2, rhomboid, 4–5 angled in side-view. Basidia 32–57 × 11–15 μm, clavate, 4-spored, with clamp-connections. Lamellae edge heterogeneous with abundant cheilocystidia, 55–90 × 10–17(–20) (base) × 3.5–6 μm (apex), lageniform, top often with constrictions, relatively thick-walled and not collapsing easily, with clamp-connections. Pileipellis a cutis of cylindrical hyphae, 3.0–11.5 μm wide, subcutis made up of 5–20 μm wide, cylindrical hyphae, constricted at septa. Pileitrama regular, made up of 4–10 μm wide, cylindrical, hyaline hyphae. Pigment pale brown, intracellular. Stipitipellis a cutis of cylindrical hyphae, 3.5–17 μm wide, without clamp-connections, with pale brown intracellular pigment. Caulocystidia absent. Clamp-connections present except in stipitipellis.
Habitat and distribution: In small groups in various types of habitats, most records are from semi-natural grasslands (pasture grazed by sheep, slightly to distinctly calcareous), but also found in rich, open and swampy fir (Abies alba) forest vegetation, calcareous herb rich forest and rich fen margins. Except for a coastal, lowland find in SW Norway and Teletskoye Lake shore (Altai Republic), the species was up to now only found at higher altitudes, in C and S Europe in montane-subalpine zones, and in N Europe mainly in northern boreal (and low alpine) zone(s). Widespread species, known from Austria, Germany, Norway, Spain (Pyrenees), Sweden and the Altai Republic in Asian Russia.
Comments: This is apparently the most frequent species within the /Rhombisporum clade, with a wide distribution and a wide habitat spectrum. The species can be distinguished by its well-developed, large cystidia, and small rhomboid to 5-angled spores, normally <10 μm long. Entoloma subcuboideum differs from its closest species E. rhombiibericum by 24 substitution and indel positions in the ITS region, with a similarity of 96%.
Additional specimens examined: Austria, Kärnten, Malta, Schlatzing, Schlatzingerau, poorly manured, subalpine meadow on calcareous soil, 27 Aug 2008, P. Karasch, WU28769. Germany, Taunus, Oberursel, near Hühnerbergwiesen, in semi-natural grassland, 3 Nov 2017, K. Reschke, KaiR1060. Norway, Hedmark, Tolga, Vingelen, Langsetrane, weakly calcareous semi-natural grassland (pasture), 20 Aug 2010, J. B. Jordal, JBJ10-E01 (O-F-252012); Hordaland, Bømlo, Vestra Vika, in calcareous semi-natural sheep pasture, 9 Oct 2008, J. B. Jordal, A. Knutsen, O-F-287889; Møre og Romsdal, Sunndal, Grøvudalen, Myrasetra, in rich semi-natural sheep pasture, 25 Aug 2003, J. B. Jordal, O-F-288056; Geitådalen, Hafsåssetra, semi-natural grassland (pasture), 10 Aug 2015, J. B. Jordal, JB15-E11 (O-F-251994); Aure, Ertvågsdalen, calcareous snowbed/sheep pasture, 30 Aug 2003, F. Oldervik 422.03 (O-F-223820); Møre og Romsdal, Herøy, Runde, Goksøyr, pasture grazed by sheep, 23 Sept 2019, J. B. Jordal, JBJ19-061; Nordland, Grane, Mellingsdalen ved Stillelva, SE of Storlistua, middle boreal, open, shallow, moist and calcareous edge of rich fen, 27 Aug 2005, E. Bendiksen, T. E. Brandrud, TEB 88-05 (O-F-166783); Nordland, Alstahaug, Tjøtta, Storvollhalsen, calcareous seminatural grassland grazed by sheep, 31 Aug 2018, J.B. Jordal, JBJ18-020 (O-F-256747); Trøndelag, Levanger, Hellum, calcareous semi-natural pasture, 29 Aug 2009, J. B. Jordal, H. Bratli, O-F-291191; Oppdal, Vinstradalen, 19 Aug 2005, J. B. Jordal, O-F-158290; Rogaland, Kvitsøy, Austre Buøya, 2 Oct 2008, J. I. Johnsen, J. B. Jordal, O-F-287721; Nordland, Saltdal, Junkerdalsura, 12 Sept 2019, calcareous, open herb rich birch forest, G. Gaarder, JBJ19-027. Russia, Altai Republic, Altai Nature Reserve, cost of the Teletskoye Lake, calcareous pasture on delta, 28 Aug 2018, O. Morozova, 1244-11AL18 (LE 312542). Spain, Catalonia, Espot (Plaça dels Arbres, Lleida), in an open area of a Abies alba and Betula pendula forest, 25 Aug 2014, J. Vila, O. Morozova, X. Llimona, JVG 1140825-8 (L-0607557, LE 312543). Sweden, Jämtlands län, Östersund, Frösön, Summarhagen, meadow, 29 Aug 2018, K. Reschke, KaiR1283 (L-0607526).
Entoloma pararhombisporum Noordel., G.M. Jansen, O.V. Morozova, Reschke & Dima, sp. nov. Fig. 6
MycoBank: MB 842126
Holotype: Sweden, Jämtland, near Angsta, Storvålen, horse pasture, 27 Aug 2018, K. Reschke, Kai1235 (L-0607556), GenBank No. OL853742 (ITS).
Misapplied name: Entoloma rhombisporum var. floccipes s. Morozova et al. (2016).
Etymology: Refers to the resemblance with Entoloma rhombisporum
Diagnosis: Entoloma pararhombisporum is differentiated from E. rhombisporum and E. subcuboideum particularly by the combination of a more greyish tinged pileus, fibrillose stipe, the voluminous and easily collapsing cheilocystidia, and the congophilous plaques in the stipitipellis.
Description: Pileus 10–25 mm diam., convex, with blunt or faintly depressed centre, with straight margin, not distinctly hygrophanous, obscurely to distinctly translucently striate, pale to dark yellow-brown often with grey tinge, frequently with distinctly darker centre, finely fibrillose to auriferous–silky shiny when fresh, glabrescent with age, or becoming somewhat rimose-subsquamulose at centre with age. Lamellae L = 20–25, l = 1–5, moderately distant, adnate-emarginate with decurrent tooth, arcuate, white then pale pink, with concolorous, entire edge. Stipe 30–50 × 1–3 mm, cylindrical, pale brown to almost white, much paler than the pileus, initially silvery striate lengthwise and finely pruinose when fresh, glabrescent with age and appearing polished. Smell and taste farinaceous, sometimes indistinctly so, or nitrous. Basidiospores 8–11 × 6.5–9 μm, av. 8.8–10 × 7.4–8.4 μm, Q=1.0–1.5, Qav=1.20–1.25, rhomboid, 4-sided in side-view. Basidia 40–48 × 11.5–15 μm, 4-spored, with clamp-connections. Lamella edge heterogeneous. Cheilocystidia abundant, lageniform, with or without constricted neck, rather fragile and easily collapsing, 42–67 × 10–30 (base) × 3.5–7 (apex) μm. Pileipellis a cutis of 4–10 μm wide hyphae, with some wider, subclavate terminal elements up to 22 μm wide. Pigment brown, intracellular. Stipitipellis a cutis of cylindrical hyphae, 5–12 μm wide, with light brown intracellular pigment, hyphae walls with abundant congophilous plaques. Differentiated caulocystidia usually absent, but present and abundant in one collection (KR1224), then 25–70 × 8–19 μm, clavate, clavate-mucronate or lageniform. Clamp-connections abundant.
Habitat and distribution: In semi-natural grasslands, roadsides and lawns on calcareous soil, and low herb and tall herb vegetation in calcareous forests. So far verified from mainly northern regions in Norway (many sequences), Austria, Russia, and Sweden.
Comments: A particular feature observed in some collections of this species is the presence of minute congophilous plaques (red spots) in the hyphal walls of the stipitipellis, a phenomenon not known to exist in other species of this clade. One collection (KaiR1224) resembles E. caulocystidiatum, because of the abundant caulocystidia, but these are present only at apex, and differently shaped and less voluminous than in that species. Apart from the congophilous plaques seen in some collections, E. pararhombisporum is very similar to E. subcuboideum. Entoloma pararhombisporum differs from its closest species Entoloma sp. from North America (GenBank: MK580835) by 25 substitution and indel positions in the ITS region, with a similarity of 95%.
Additional specimens examined: Austria, Tirol, Galtür, Jamtal, Alpele-Schnapfenalpe, 29 Aug 2004, A. Hausknecht, WU25061. Norway, Nordland, Alstahaug, Blomsøya, Buøya, calcareous semi-natural sheep pasture, 18 Sept 2004, G. Gaarder, R. Haugan, B. Pedersen, P. Larsen, O-F-67299; Nordland, Alstahaug, Tjøtta, Storvollhalsen, calcareous semi-natural grassland (sheep pasture), 29 Aug 2020, J.B. Jordal, JB20-E24 (O-F-76646). Nordland, Grane, Holmvassdalen, low herb meadow in old calcareous spruce forest, 5 Sept 2013, J. Lorås, JL17-13; ibid., among mosses in open, subalpine calcareous birch forest, 5 Sept 2013 J. Lorås, JL7-12; Nordland, Grane, Danielåsen nature reserve, calcareous tall herb spruce forest, 9 Aug 2016, M. Eidissen, J. Erlandsen, JL1-16; ibid. 24 Aug 2016, J. Lorås, JL31-16; Nordland, Hattfjelldal, Gryteselv, calcareous semi-natural grassland, grazed by cattle, 10 Aug 2018, J. Lorås, JL41-18; Nordland, Vefsn, Forsheim, semi-natural grassland (mown), 19 Sept 2004, G. Gaarder, B. H. Larsen, O-F-67382; Trøndelag, Bjugn, Valsøya, Plassen, calcareous semi-natural grassland, grazed by cattle, 3 Oct 2019, J. B. Jordal, K.A. Mandal, JBJ19-083; Trøndelag, Ørland, Tollefsvika, Annebakken, calcareous, grassy road verge regularly mown, 6 July 2019, K.A. Mandal, KAM 3-20 (TRH-F-18048); Trøndelag, Oppdal, Slettvoll, calcareous semi-natural pasture, 24 Aug 2009, J. B. Jordal, O-F-291330; Trøndelag, Oppdal, Engan, calcareous seminatural grassland, 1 Sept 2018, J.B. Jordal, JBJ18-026 (O-F-256752); Trøndelag, Oppdal, Øvre Gorset, calcareous semi-natural grassland, 30 Aug 2019, G. Gaarder, GG1-19, GG2-19; Møre og Romsdal, Aure: Ertvågsdalen, uppermost part, calcareous (semi-natural) snowbed-pasture grazed by sheep, 18 Sept 2004, F. Oldervik 506.04 (O-F-187951); Møre og Romsdal, Hustadvika, Fræna: Rishaug, calcareous lawn, 23 Sept 2003, G. Gaarder 3804 (O-F-223278); Møre og Romsdal, Herøy, Nerlandsøy, Mulevika, calcareous pasture grazed by sheep, 27 Sept 1994, G. Gaarder, J. B. Jordal, JBJ94-596 (O-F-254392); Hordaland, Bømlo, Vestra Hiltahuset, calcareous lawn, 29 Sept 2009, A. Knutsen, J. B. Jordal, O-F-291271. Russia, Pskov region, buffer zone of the Polistovsky Nature Reserve, Nesvino, grassland with Calamagrostis epigeios, 7 Aug 2020, L. Kalinina, LE F-331983 (as E. cf. rhombisporum (Kalinina 2021)); Tver region, Zubtsov district, 2 km to SE from Mozgovo village, right bank of the Derzha river, in grassland, 11 Sept 2015, O. Morozova, 960-34TV15 (LE 311836), as E. rhombisporum var. floccipes (Morozova et al. 2016). Sweden, Västernorrlands län, Kullen mountain, grassy stripe between gravel path and sheep pasture, 26 Aug 2018, K. Reschke, KaiR1223 (L-0607555), KaiR1224 (L-0607560), KaiR1226 (L-0607559); KaiR1232 (L-0607558), Jämtlands län, Äre, Jale in Trång, grassland in the camping, 28 Aug 2016, B. Dima, O. Morozova, 1121-OM103 (LE 312544).
Entoloma cf. prismatospermum (Romagn.) E. Horak, Sydowia 28(1–6): 216 (1976) [1975–1976] Fig. 7
MycoBank: MB 313794
Holotype: France, Neuville-en-Hez, dept. Oise, 5 Sept 1956, H. Romagnesi, 56.146 (PC).
≡ Rhodophyllus prismatospermus Romagn. Bull. mens. Soc. linn. Lyon 43(9): 386 (1974)
Etymology: Refers to the almost “prismatic” shape of the spores.
Original description (translated from Latin): Habit of a Nolanea. Pileus (in young specimens) 5–9 mm broad, hygrophanous, slightly translucently striate, conical then convex, papillate, mouse grey, pallescent on drying, silky shining. Stipe (10)20–30 × 1–2 (base 3.5) mm, weakly bulbous, pale grey then dark grey, at apex subtly pruinose. Context thin, smell and taste farinaceous-rancid. Lamellae rather distant, 1.5–2 mm broad, adnate, then slightly sinuate or uncinate, pale greyish then mouse grey. Spores bluntly prismatic sometimes almost triangular, 7.5–9 × (6)7–8.5 μm. Basidia with clamp-connections, 50–60 × 12–14 μm, cheilocystidia and caulocystidia long, fusiform, with narrow neck, 60–75 × 9–12 μm. Cutis filamentous, hyphae without clamp-connections; pigment not manifest; hyphae not clamped. In moist places under deciduous trees.
A Norwegian collection, studied by us, comes microscopically close to the original description of E. prismatospermum, particularly on account of the small, but never really triangular spores. However, apart from a photograph of a dried specimen, we have no information on the macroscopical characters. The microscopical characters (Fig. 7) are as follows: Basidiospores 6.7–9.9 × 6.0–8.3 μm, in average 8.3 × 7.1 μm; Q=1.0–1.3, Qav=1.2, rhomboid in side view. Basidia 4-spored, with clamp-connections. Lamella edge sterile, appearing brownish in 5% KOH. Cheilocystidia 60–75 × 9–12 μm, fusiform to lageniform. Hymenophoral trama made up of cylindrical elements, 50–120 × 5–7 μm, clamped. Pileipellis cutis with repent terminal cells 30–80 × 5.5–11 μm; cylindrical with rounded apex, pale brown intracellular pigment, no clamps seen. Stipitipellis composed of cylindrical hyphae, 2.5–8.5 μm wide, hyaline, no caulocystidia seen, clamped. Clamp-connections present in the hymenium.
Habitat and distribution: in a calcareous grassland, Norway.
Comments: Entoloma cf. prismatospermum has by far the smallest spores of the /Rhombisporum clade, which tend to be more or less rhomboid (and sometimes more or less triangular in the holotype). The holotype has been studied morphologically (Noordeloos 1980), but DNA extraction and PCR amplification of the ITS region have so far failed. We think, however, that the Norwegian collection fits well with the original concept of the species. We refer to this collection as E. cf. prismatospermum. It differs from its closest species E. rhombisporoides by 28 substitution and indel positions in the ITS region, with a similarity of 95%.
Collection examined: Norway, Telemark, Porsgrunn, Mulevarde, 59.0995°N, 9.7004°E (± 1 m), dry, calcareous grassland, 14 Oct 2012, A. Molia & T. Læssøe, AM340n2012 (O-F-245768).
Entoloma rhombisporoides Noordel., O.V. Morozova & Dima, sp. nov. Fig. 8
MycoBank: MB 842127
Holotypus: Russia, Tver region, Zubtsov district, 2 km to SE from Mozgovo village, right bank of the Derzha river, grassland, 11 Sept 2015, O. Morozova 964-35TV15 (LE 312545), GenBank No. OL853787 (ITS).
Etymology: Refers to the similarity to E. rhombisporum.
Diagnosis: Entoloma rhombisporoides is characterized by the presence of caulocystidia at apex of stipe, relatively small spores, and rather pronouncedly fibrillose stipe surface.
Description: Pileus 5–20 mm diam., conico-convex to convex, finally plano-convex, with blunt or slightly umbilicate centre, slightly hygrophanous, when moist translucently striate up to centre, yellow-brown, particularly in marginal zone, with a brown-grey central part, glabrous or minutely rugulose-fluffy at centre. Lamellae, L = 24–28, l = 2–3, rather distant, adnate-emarginate, often with slightly decurrent tooth, creamy white/greyish white, reddish pink when mature, with an eroded to subserrulate, concolorous edge. Stipe 20–40 × 1–2(–3) mm, slender, cylindrical, pale brown, yellow-brown, glabrous, initially with pruinose-hairy apex and, rather pronouncedly fibrillose-striate lengthwise, basal part with white tomentum. Smell and taste unknown. Basidiospores 7.5–11.0(–12.5) × 7.0–9.5 μm, av. 8.0–9.5 × 7.0–8.2 μm, Q=1.0–1.5, Qav=1.2–1.3, mostly 4-angled, rarely also 5-angled in side-view. Basidia 22–45 × 6–14 μm, 4- and 2-spored, with clamp-connections. Lamella edge heterogeneous, cheilocystidia scattered among basidia, not very pronounced, 35–80 × 9–18 μm, slenderly lageniform or utriform, often with a long stalk, up to half the length, neck sometimes septate, very thin-walled. Pileipellis a cutis made up of 6–17 μm wide hyphae with brown intracellular pigment. Caulocystidia scattered at apex only, 40–90 × 7.0–14 μm, lageniform, narrowly clavate, Clamp-connections present in the hymenium, sometimes sparse, elsewhere lacking.
Habitat and distribution: In open, dry, calcareous grasslands, often grazed or mown, often in vicinity of sea-shores or along riverbanks. Known from Germany, Norway, Russia and the Netherlands (from environmental DNA sample).
Comments: Entoloma rhombisporoides differs from its closest species E. caulocystidiatum by the presence of caulocystidia at apex only, and a rather polished stipe surface, and at least 13 substitution and indel positions in the ITS region, with a similarity of 97%.
Additional specimens examined: Estonia, Saaremaa, Lääne-Saare vald, Mäebe, Kihelkonna, in dry, calcareous grassland, 18 Sept 2015, V. Liiv, TUF120252 (UNITE: UDB024646). Germany, Bayern, Alttötting, Alzgern, in wet, north-faced slope among mosses, 3 Oct 2019, W. Sprengler (L-0607768); Sachsen, Zittau, NSG Eichgrabener Feuchtgebiet, Kaiserfelder Wiese am Lottersteg, sheep pasture, 2 Nov 2016, A. Karich (GLM-F128995). Norway, Oslo, Ulvøya, Sørstranda, open calcareous shallow soil grassland, 14 Sept 2017, E. Bendiksen, EB 381/17 (O-F-254547); Oppland, Gran, Søsterkirkene, churchyard, dry, herb-rich lawn (under Betula), 17 Aug 2017, T. E. Brandrud, TEB 139-17 (O); Telemark, Bamble, Langøya, west side, open calcareous shallow soil grassland, 15 Oct 2013, A. Molia, T. Læssøe, AM-266j-2013 (O-F-22052); Hordaland, Bømlo, Spyssøya, Myra, 10 m alt., calcareous semi-natural grassland (sheep pasture), 2 Oct 2009, A. Knudsen, J. B. Jordal, O-F-291299; Vestfold, Larvik, Løvallåsen, dry, calcareous grassland, 9 Oct 2013, A. Molia, T. Læssøe, O-F-21896.
Entoloma caulocystidiatum Noordel. & Dima, sp. nov. Fig. 9
MycoBank: MB 842131
= Entoloma rhombisporum var. floccipes Noordel. in Beih. Nova Hedwigia 91: 77 (1987), nom. inval., Art. 40.7 (Shenzhen).
Holotypus: Germany, Thuringia, Warza, 13 Oct 1984, F. Gröger, 211/1984 (L). GenBank No. OL853707 (ITS).
Etymology: The name refers to the cystidiate surface of the stipe.
Diagnosis: The species is characterized by the flocculose stipe (apex) with well-developed caulocystidia.
Description: Pileus 12–32 mm diam., campanulate or conical, expanding with age to convex with small umbo, with straight margin, hygrophanous, when moist translucently striate up to half the radius or not striate, yellow-brown, ochre or milky coffee or with grey tinge, smooth, glabrous, becoming radially fibrillose or rimose with age. Lamellae, L = 20–25, l = 1–3, distant, adnate, deeply emarginate, often with decurrent tooth, broadly ventricose, up to 9 mm broad, pale whitish then pink, often with ochre tinge, with concolorous, entire edge. Stipe 23–65 × 2.0–4.5 mm, cylindrical, sometimes slightly broadened at base, pale grey-brown or olivaceous brown, more or less concolorous with pileus, or distinctly paler, especially at apex, at apex pruinose to hairy, downwards initially fibrillose to flocculose or fibrillose striate with loose fibrils, later often more glabrous, white tomentum at base. Context thin, fibrillose, at centre of pileus grey-brown, elsewhere pallid. Smell none or slightly spermatic to almost rancid. Taste not distinctive. Basidiospores 8.5–12.0 × (6.0–)7.0–11.0 μm, Q=1.0–1.55, rhomboid, 4-angled in side view. Basidia 4-spored, with clamp-connections. Lamella edge sterile. Cheilocystidia 45–125 × 10–30 (basal part) × 2–12 (apex) μm, lageniform, often with broad base and long narrow neck. Pleurocystidia present, similar to cheilocystidia, close to lamella edge only. Pileipellis a cutis of narrow, cylindrical hyphae, 2–8 μm wide. Pigment intracellular in pileipellis, in addition sometimes distinctly membranous, but never encrusting. Stipitipellis a cutis with numerous caulocystidia all over the length, 45–80 × 5–20 μm, versiform, subcylindrical to lageniform. Clamp-connections present in all tissues.
Habitat and distribution: Gregarious in pastures, in road sides, the type in xerophytic grassland on calcareous soil. Known to occur in Austria, Estonia, Germany, Norway and the Netherlands (from environmental DNA sample).
Comments: Entoloma caulocystidiatum resembles E. rhombisporum, but differs by the floccose stipe with abundant caulocystidia, darker colours, abundant cheilocystidia, and slightly larger spores. The ITS sequence of holotype of Entoloma rhombisporum var. floccipes belongs to this well-separated species, but the name cannot be used as a basionym for a combination to species level, since two herbaria were indicated in the protologue (Noordeloos 1987) and therefore, based on ICN Art 40.7 (Turland et al. 2018), the name of this variety is invalid. A full description and line drawings can be found in Noordeloos (1987, 1992). Entoloma caulocystidiatum differs by six substitution and indel positions from its sister lineage, tentatively named as E. aff. caulocystidiatum, represented by a single sequence. Whether this latter belongs to E. caulocystidiatum needs further detailed study. Otherwise, the closest species of E. caulocystidiatum is E. rhombisporoides differing from it by 11 substitution and indel positions in the ITS region, with a similarity of 98%.
Additional specimens examined: Estonia, Lääne, Lääne-Nigula, Rannaküla, Lääne-Nigula, 15 Oct 2019, T. Ploompuu, TUF135515. Norway, Østfold, Marker, Lerbråten, open place with various plants, strongly calcareous with visible remnants of sea shells in the ground, 12 Sept 2019, Ø. Weholt, E26-19. As E. aff. caulocystidiatum: Norway, Nordland, Hattfjelldal, Salomontjørna, calcareous fen margin, 30 Aug 2005, G. Gaarder, A. Heggland, O-F-281299.
Entoloma rhombiibericum Vila, Dima & Noordel., sp. nov. Fig. 10
MycoBank: MB 842132
Holotype: Spain, Catalonia, Barcelona, Osona, Vidrà, near Turó del Pla de la Creu, 20 Sept 2008, J. Vila, F. Caballero, JVG 1080920-6 (L-0607540). GenBank No. OL853779 (ITS).
Misapplied name: Entoloma rhombisporum var. floccipes s. Vila and Caballero (2009).
Etymology: Refers to the placement in the /Rhombisporum clade and the fact that it is the only species originally described from Southern Europe, specifically from the Iberian Peninsula.
Diagnosis: Entoloma rhombiibericum differs from E. caulocystidiatum by the darker, greyish brown colour of the pileus, which is only indistinctly translucently striate, and the considerably shorter cheilo- and caulocystidia, which are present at apex of stipe only. Its phylogenetic position is rather distant from E. caulocystidiatum.
Description: Pileus 20–45 mm diam., flattened to concave, often with a small obtuse umbo; brown, darker at the centre; hygrophanous, weakly translucently striate at the margin of young specimens, glabrous or finely fibrillose when well hydrated, fibrils more visible upon drying; with straight or recurved margin. Lamellae unequal with abundant lamellulae, adnate to almost free, dense, thick, ventricose, concave, whitish when young, then pale pink, with darker pink spots from the spores; with concolorous, entire or somewhat irregular edge in well-developed basidiomata. Stipe 40–80 × 3–5 mm, cylindrical, straight, whitish, slightly to densely fibrillose-tomentose; apex pruinose, base white tomentose. Context thin, greyish brown in the pileus, stained with yellow in the stipe. Smell fungoid or somewhat spermatic. Taste not recorded. Basidiospores 8.5–11.2 × 8.3–10.7 μm, av. 9.8 × 9.1 μm, Q=1–1.2, Qav = 1.08, isodiametrical, quadrangular to rhomboid in side-view. Basidia up to 48 × 18 μm, narrowly clavate to subcylindrical, 4-spored, with clamp-connections. Lamella edge heterogeneous, with cystidia mixed with basidia. Cheilocystidia 37–54 × 11–14.5 (base) × 2.5–5.5 (apex) μm, lageniform or clavate-mucronate. Pileipellis a cutis of cylindrical hyphae, 5.5–11 μm wide, with differentiated sublageniform to fusiform terminal elements. Pigment abundant, brown, intracellular. Caulocystidia in dense clusters, at apex of stipe, similar to cheilocystidia. Clamp-connections present in all tissues, although scarce in the pileipellis.
Habitat and distribution: Terrestrial in montane (1340 m asl.), subatlantic shrub vegetation with Buxus sempervirens, Rubus caesius, and Sorbus aria, on calcareous soil. So far only known from NE Spain.
Comments: This species is well-supported phylogenetically and has some distinct micromorphological features, and is thus here described as new, although it is so far only known from the type locality with box (Buxus) shrubs. Entoloma rhombiibericum differs from its closest species E. caulocystidiatum by at least 22 substitution and indel positions in the ITS region, with a similarity of 96%.
Entoloma pentagonale J.B. Jordal, Noordel. & Dima, sp. nov. Fig. 11
MycoBank: MB 842133
Holotype: Norway, Møre og Romsdal, Giske, Molnes, 10 m alt., calcareous semi-natural grassland (cow pasture), 5 Oct 2015, J.B. Jordal, JB15-E10 (O-F-251993). GenBank No. OL853749 (ITS).
Etymology: Refers to the predominantly 5-angled spores.
Diagnosis: Entoloma pentagonale is a distinctive species within the /Rhombisporum clade with its often yellow-tinged, pallescent, polished stipe, predominantly 5-angled, small spores and clampless hyphae without clamp-connections.
Description: Pileus 10–25 mm, conical expanding to conico-convex with straight margin, slightly hygrophanous, translucently striate up to half the radius, pale to rather dark brown, strongly pallescent on drying, silky-shiny, glabrous, innately radially fibrillose. Lamellae, L = 20–28, l = 3–5, distant, adnate-emarginate with long decurrent tooth, pale greyish cream-coloured, later with pink tinge, with entire, concolorous edge. Stipe 25–40 × 1–2 cm, cylindrical, pallid, creamy grey or with yellowish tinge, glabrous, almost polished, with white basal mycelium. Smell and taste not noted. Basidiospores 8.0–11.5 × 6.5–9.0 μm, Q=1.0–1.4, average spore 9.4–10 × 7.7–8.2 μm, Qav=1.2–1.25, predominantly 5-angled, some subrhomboid 4-angled with rounded walls. Basidia 24–46 × 10–15.5 μm, narrowly clavate, almost cylindrical sometimes, clamp-connections absent. Lamella edge heterogeneous. Cheilocystidia 37–80 × 11–21 μm, fusiform to lageniform, some septate, hyaline, very scarce and inconspicuous in some collections. Hymenophoral trama made up of cylindrical hyphae, 3.5–12.5 μm wide. Pileipellis a cutis of cylindrical hyphae, 2–5 μm wide, with pale brown, intracellular pigment, particularly in the subpellis. Pileitrama made up of cylindrical hyphae, 5–10 μm, subpellis hyphae inflated, up to 20 μm wide. Stipitipellis a cutis of cylindrical, 3.5–16 μm wide hyphae with pale brown, intracellular pigment. Differentiated caulocystidia absent. Clamp-connections absent.
Habitat and distribution: In open, natural to semi-natural, coastal grasslands on calcareous soil, and calcareous forests. Widespread in Norway, also recorded from Germany.
Comments: Entoloma pentagonale is distinguished from other species in the /Rhombisporum clade mainly on micromorphological features, basically the predominantly five-angled, small spores and the hyphae without clamp-connections. Most verified collections are from South Norway, where it is found repeatedly in strictly calcareous, coastal, dry grasslands between the sea and adjacent calcareous pine forests. These semi-arid, steppe-like habitats may be typical of the species, also further south in Europe. Entoloma pentagonale differs from its closest species E. lunare by 22 substitution and indel positions in the ITS region, with a similarity of 96%.
Additional specimens examined: Germany, Rheinland-Pfalz, Simmertal, Horbach, poor, dry grassland on Devonian greenschist, K. Reschke, KaiR1046 (L-0607539). Norway, Nordland, Grane, Holmvassdalen, calcareous, moist, tall-herb spruce forest, 31 Aug 2010, M. E. Noordeloos, MEN201064 (O-F-370773); ibid., low herb vegetation in strongly calcareous spruce forest, 1 Sept 2015, J. Lorås JL 27-12; Telemark, Bamble, Langøya west, open, calcareous shallow soil grassland, 10 Oct 2012, T. Læssøe, A. Molia AM-327h-2012 (O-F-245720); Vestfold, Larvik, Fugløya, open, calcareous shallow soil grassland, 11 Oct 2012, T. Læssøe, A. Molia AM-334k-2012 (O-F-245736); Oslo, Gressholmen, open, calcareous shallow soil grassland, 7 Oct 2013, T. Læssøe, A. Molia AM-243o-2013 (O-F-21867); Telemark, Porsgrunn, Mulevarde, open, calcareous shallow soil grassland, 5 Sept 2012, T. Læssøe, A. Molia AM-232s-2012 (O-F-245763). Vestfold, Larvik, Fugløya, calcareous, dry grassland, 11 Oct 2013, A. Molia, T. Læssøe, O-F-245743, 12 Oct 2013, A. Molia, T. Læssøe, O-F-21971; Løvallåsen, calcareous, dry grassland, 9 Oct 2013, A. Molia, T. Læssøe, O-F-21887; Møre og Romsdal, Herøy, Mulevika, semi-natural, coastal grassland grazed by sheep, 13 Sept 2004, M. E. Noordeloos, G. Gulden, J. B. Jordal, O-F-178042; Trøndelag, Bjugn, Tarva, Været, semi-natural grassland grazed by sheep, 4 Oct 2019, J. B. Jordal, K. A. Mandal, JBJ19-091; Vestland, Stord, Hovaneset, calcareous semi-natural grassland (grazed by sheep), 14 Sept 2016, P. Fadnes, PF1-16.
Entoloma lunare J.B. Jordal, Dima, G.M. Jansen & Noordel., sp. nov. Fig. 12
MycoBank: MB 842134
Holotype: Norway, Nordland, Alstahaug, Tjøtta, Storvollhalsen, calcareous semi-natural grassland (sheep pasture), 31 Aug 2018, J. B. Jordal, JB18-017 (O-F-256744). GenBank No. OL853719 (ITS).
Etymology: lunare (Lat.) = pertaining to the moon, referring to the relatively pale yellow colour of the basidiomata.
Diagnosis: Entoloma lunare fits well in the /Rhombisporum clade, differing by 22 base-pairs from E. pentagonale. The basidiomata of E. lunare are paler, but microscopically both species are rather similar, except for the occurrence of caulocystidia in E. lunare.
Description: Pileus 10–15 mm, conical, expanding to plano-convex with slight umbo, with straight margin, hygrophanous, deeply translucently striate when moist, pallid cream-coloured with pale greyish brown centre, glabrous with slightly fibrillose centre. Lamellae L = 20–24, l = 3–5, distant, adnate-emarginate with decurrent tooth, creamy white, then pink with slightly irregular, concolorous edge. Stipe 30–50 × 1–3 mm, cylindrical, cream-coloured like pileus and lamellae, smooth, polished. Smell and taste not noted. Basidiospores 7.9–10.6 × 6.2–8.3 μm, av. 9.2 × 7.2 μm; Q=1.1–1.5, Qav=1.25, predominantly 5-angled, occasionally 4- or 6-angled. Basidia 35–41 × 11.5–13.5 μm, clavate, 4-spored, clamp-connections absent. Cheilocystidia 45–67 × 11–14 μm, scarce, lageniform, clamp-connections absent. Hymenophoral trama composed of cylindrical and inflated hyphae, 5–15 μm wide. Pileipellis a cutis of narrow, cylindrical hyphae, with scattered semi-erect, cylindrical to clavate terminal elements, up to 15 μm wide with pale, intracellular pigment. Stipitipellis a cutis of cylindrical hyphae, 4–9 μm wide, some clavate terminal cells up to 15 μm wide. Caulocystidia present in the upper part of the stipe, similar to the cheilocystidia. Clamp-connections absent.
Habitat and distribution: terrestrial in extensively grazed semi-natural grassland on calcareous soil. Only known from two collections at the type-locality in a coastal site in northwestern Norway.
Comments: Entoloma lunare fits well in the /Rhombisporum-clade. The basidiomata seem to be paler than those of E. pentagonale. Microscopically both species are rather similar, except for the occurrence of caulocystidia in E. lunare. Entoloma lunare differs from its closest species E. pentagonale by 22 substitution and indel positions in the ITS region, with a similarity of 96%.
Additional specimens examined: Norway, Nordland, Alstahaug, Tjøtta, Storvollhalsen (near the type locality), calcareous semi-natural grassland (sheep pasture), 29 Aug 2020, J. B. Jordal, JBJ20-E17 (O-F-76645).
Entoloma pratulense Noordel. in Beih. Nova Hedwigia 91: 89–90. 1987. Fig. 13
MycoBank: MB 133101
Holotype: Sweden, Medelpad, Finnsjö Torp, semi-natural grassland, Aug 1985, M. E. Noordeloos, MEN85040 (L). GenBank No. OL853777 (ITS).
Description: Pileus 8–14 mm diam., conical usually with small papilla, with slightly deflexed margin, then expanding to convex with or without papilla, hygrophanous, when moist deeply translucently striate, dark brown or red-brown, slightly paler at margin, pallescent along radial streaks on drying, smooth, innately radially fibrillose, centre sometimes slightly tomentose. Lamellae distant, L = 15–20, l = 1–5, adnate, emarginate, segmentiform to ventricose, white then pink with concolorous, fimbriate edge. Stipe 20–40 × 1–2 mm, cylindrical, pale brown, pruinose at apex, downwards smooth, polished. Context thin, concolorous with surface. Smell none or farinaceous. Taste not tried. Basidiospores (7.0–)8.0–11.0 × 6.0–9.0 μm, Q=1.0–1.4, 4–6 angled in side-view, probably with basal facet. Basidia 25–50 × 10–14 μm, 4-, rarely also 2-spored, with clamp-connections. Lamella edge sterile. Cheilocystidia 55–80 × 8–20 × 1.5–6 μm, slenderly lageniform or tibiiform, sometimes with moniliform neck. Pileipellis a cutis of narrow, cylindrical hyphae, 4–12(–16) μm wide, at centre sometimes with semi-erect, narrowly clavate terminal elements. Pigment intracellular in pileipellis. Caulocystidia absent. Clamp-connections present in the hymenium and covering layers.
Habitat and distribution: In semi-natural and natural grasslands, hayfields and meadows, preferably on calcareous soil, but also found once in a calcareous forest, fruiting from August to October. Rare, but widespread, in subalpine (northern boreal) and alpine regions, rarely in the lowlands, apparently with a transatlantic distribution. Sequenced records are from Norway, Sweden (type) and Canada, non-sequenced records are from Austria, Italy, Great Britain, and Sweden.
Comments: Entoloma pratulense differs in habitat preferences from the other members of the group, being concentrated to alpine and subalpine regions. Entoloma pratulense and E. sororpratulense make up a sister clade/lineage to the /Rhombisporum clade in the strict sense. Although genetically close, the pratulense-sororpratulense lineage is distinguished from the rest of the /Rhombisporum clade by the normally non-rhomboid spores (usually 5–6 angled). Furthermore, the two are characterized by very long cheilocystidia. For distinguishing between E. pratulense and E. sororpratulense, see comments on the latter. Entoloma pratulense differs from its sister species E. sororpratulense by 20 substitution and indel positions in the ITS region, with a similarity of 96%.
Additional specimens examined: Canada, Quebec, Radisson, 1 Sept 2018, R. Lebeuf, J. Landry, QFB30896. Norway, Nordland, Grane, Holmvassdalen, calcareous tall herb spruce forest, J. Lorås, JL 41A-10; Trøndelag, Trondheim, Bratsberg, Røstadkleiva, semi-natural grassland (pasture), 27 Sept 2016, T. Solem, M. Gjestland, T. Gajik TJ7-16 (TRH-F-24921); Trøndelag, Steinkjer, Kvamsfjellet, Hals, in semi-natural grassland, intermediately rich, 21 Aug 2016, J. B. Jordal, M. E. Noordeloos, O. Morozova, O-F-304563; Møre og Romsdal, Sunndal, Vangan, in semi-natural grassland, weakly calcareous, 27 Aug 1993, J. B. Jordal (O-F-240811); Møre og Romsdal, Vestnes, Løvika, Kriken, in semi-natural grassland, intermediately rich, 30 Sept 2014, J. B. Jordal (O-F-247997); Finnmark, Porsanger, Vest for Kolvik calcareous north facing semi-natural pasture, 25 Aug 2011, J. B. Jordal, (O-F-252376); Karlsoy, Reinøya, Nordeidet, calcareous semi-natural grassland, 8 Sept 1998, V. Ravolainen, TROM-F-610204; Trøndelag, Oppdal, Vinstradalen, Bjørkåssetra, semi-natural, calcareous pasture, 26 Aug 2020, J. B. Jordal, JBJ20-E03, JBJ20-E04. Russia, Khanty-Mansiyskiy Autonomous region, Beresovskiy district, vicinities of the Neroyka village, mountain road side, 9 Aug 2020, E. Zvyagina, LE 315885.
Entoloma sororpratulense J.B. Jordal, Karich, Dima & Noordel., sp. nov. Fig. 13
MycoBank: MB 842136
Holotype: Germany, Eichgraben, in marshy grassland (old firing range), 14 Oct 2019, A. Karich, GLM-F128992. GenBank No. OL853796 (ITS).
Etymology: soror (Lat.) = sister, i.e., sister of E. pratulense.
Diagnosis: A sister species to E. pratulense, differing mainly by the more distinctly 5-angled, heterodiametrical spores, quadrangular spores less frequent, and heterogeneous lamella edge with more irregularly shaped cystidia.
Description: Pileus about 10–20 mm broad, convex then expanding, usually with a slightly depressed centre, rarely with a faint umbo, with straight to slightly crenulated margin, when young deep grey-brown, reddish or yellowish brown, paler, more yellowish brown with greyish tone when mature, then with darker central spot, hygrophanous, hardly translucently striate when young, becoming deeply striate with age, drying along radial streaks, subglabrous to distinctly radially fibrillose with loose fibrils, particularly at the centre. Lamellae L = 24–40, l = 1–3, broadly adnate-emarginate or with pronounced, decurrent tooth, pallid whitish cream when young, later tinged pink, with subentire, concolorous edge. Stipe 20–60 × 1–3 mm, cylindrical, pallid, much paler than pileus, yellowish greyish, smooth and polished or with distinct loose longitudinal fibrils; base often distinctly whitely tomentose. Smell and taste not noted. Basidiospores (7.0–)7.5–10.0(–11) × 6.0–8.5 μm, av. 7.3–9.5 × 7.0–7.6 μm, Q=1.0–1.4, Qav.=1.2, irregularly 5(–6) angled, rarely appearing rectangular in side-view. Basidia 31–40 × 6.0–12 μm, clavate, 4-spored, with clamp-connections. Lamella edge heterogeneous. Cheilocystidia (40–)50–80 × 8.0–13.5 × 2.5–5.5 × 8.0–30 μm, narrowly lageniform, narrowly fusiform, clavate, long apical part, with clamp-connections. Hymenophoral trama made up of cylindrical hyphae, with elements > 150 × 2.5–17 μm, hyaline, with clamp-connections. Pileipellis a cutis of thin cylindrical hyphae, 3.5–7 μm wide, with brown intracellular pigment, often as granules, no incrustations seen, subpellis of up to 20 μm wide hyphae, brown. Pileitrama hyphae hyaline. Caulocystidia absent. Clamp-connections present.
Habitat and distribution: In groups in grasslands and occasionally in calcareous forests. The type was found on an old firing range. Most finds are from old cattle- or sheep-grazed pastures on calcareous soil, but it has also been recorded from extensively grazed young spruce (Picea) forest on moderately rich ground, and in an old quarry in spruce forest (Lunner). In Norway mainly found along the Atlantic coast, but also recorded from Jämtland and Västra Götaland in Sweden, and in Germany.
Comments: Morphologically, E. sororpratulense can apparently be distinguished by the heterogeneous lamellae edge (not completely sterile), and the more wavy-irregular cheilocystidia. Apart from the small differences in morphology, E. sororpratulense could be distinguished from its sister E. pratulense on habitat-preferences. Entoloma sororpratulense is widely distributed in the coastal and lowland regions of Norway, and lower altitude habitats in Sweden and Germany, whereas E. pratulense is a predominantly boreal/subalpine/alpine species. Entoloma sororpratulense has formerly been treated as part of E. pratulense s.l., but is well-separated genetically, differing from E. pratulense by 20 substitution and indel positions in the ITS region, with a similarity of 96%.
Additional specimens examined: Germany, Geyer, Hermannsdorfer Wiesen, marshy grassland, 2 Oct 2020 A. Karich, R. Ullrich, E. Tomschke, GLM-F128993. Norway, Oppland, Lunner, Jørstadløkka E, middle boreal, oligotrophic spruce forest (submesic, poor); young Picea plantation (after clearcutting 1968), 30 Sept 1980, E. Bendiksen, EB 1065/80 (O-F-169678); Oppland, Grua, Skjerpemyr, ridge, abandoned quarry in calcareous spruce forest, 28 Sept 1998, E. Bendiksen, T. E. Brandrud, EB 514/98 (O-F-167907); Trøndelag, Inderøy, Mosvik, Lia, calcareous semi-natural grassland (pasture), 25 Aug 2016, G. Gaarder, O-F-304541; Hordaland, Bømlo, Vestre Vika, calcareous semi-natural grassland, 9 Oct 2008, A. Knutsen & J. B. Jordal, O-F-178536; ibid., O-F-287890; Hordaland, Bømlo, Otterøya, calcareous seminatural grassland, 10 Oct 2004, A. Knutsen, J. B. Jordal, O-F-178529; Spyssøya, Myra, 2 Oct 2009, A. Knutsen & J. B. Jordal, O-F-291294; Træet ved Vikafjorden, calcareous seminatural grassland, 3 Oct 2009, A. Knutsen, J. B. Jordal, O-F-291252; Møre og Romsdal, Fræna, Farstad, W of Storholmen, rich pasture (grazed by sheep), 25 Sept 2004, J. B. Jordal, O-F-177990; Rogaland, Sola, Vigdelstranda, calcareous semi-natural grassland, 23 Sept 2010, J. B. Jordal, O-F-294507; Troms, Karlsøy, Reinøya, Nordeidet, calcareous semi-natural grassland (grazed by sheep), 12 Sept 1998, V. Ravolainen, TROM-F-610234; Hordaland, Bømlo, Træet ved Vikafjorden, calcareous semi-natural grassland, 3 Oct 2009, A. Knutsen, J. B. Jordal, O-F-291250; Vestre Vika, calcareous semi-natural grassland, 9 Oct 2008, A. Knutsen, J. B. Jordal, O-F-287888; Spyssøya, Myra, calcareous semi-natural grassland, 2 Oct 2009, A. Knutsen, J. B. Jordal, O-F-291296; Trøndelag, Bjugn, Tarva, Været, calcareous semi-natural grassland, 4 Oct 2019, J.B. Jordal, K. A. Mandal, JBJ19-085. Trøndelag, Trondheim, Skivevollen, in semi-natural grassland (mown), 16 Sept 2016, T. Gajik, TJ8-16, TJ20-16. Sweden, Västergötland, Älvsborgs län, Trollhättan, Åkerströms naturreservat, in meadow among grass, 24 Oct 1976, L. Stridvall, LAS 76/101, as E. cocles (GB-0062664, LE 302127); Älvsborgs län, Vänersborg, Frändefors, Dereviken, in meadow among grass, 13 Oct 1990, L. & A. Stridvall, LAS 90/134, as E. cocles (GB-0062665, LE 302126).
Entoloma laurisilvae Hauskn. & Noordel. In Öst. Z. Pilzk. 15: 127 (2006) Fig. 14
MycoBank: MB 521949
Holotype: Spain, Islas Canarias, La Palma, Los Tilos, biosphere reserve “Laurisilva” (laurel forest), 11 Jan 2005, M. Geier, WU23919. GenBank No. OL853718 (ITS).
Etymology: Refers to the occurrence in the laurisilva-type of vegetation (laurel forest) on the Canary Islands (Spain).
Description: Pileus 7–10 mm broad, 4–5 mm high, bluntly conico-convex, faintly umbonate, not umbilicate, often irregularly shaped, with slightly eccentric stipe, mustard brown (5E7) with paler margin, brownish grey (5C3) towards margin with slight reddish tinge, hygrophanous, when moist translucently striate up to half the radius, fading to pale brown with age, silvery grey; glabrous, somewhat fissurate at margin. Lamellae distant, L = 15–20, l = 1–3, emarginate to adnate with decurrent tooth, ventricose, pale greyish when young, later tinged pink, with entire, concolorous edge. Stipe 20–25 × 0.5–1 mm, cylindrical, hardly broadened at base, translucently greyish, when bruised or with age turning pale olivaceous yellow, fibrillose striate, white tomentose at base, glabrous. Context membranous, brittle, without special taste or smell. Basidiospores 7.0–10.5 × 6.0–9.0 μm, average 8.3–9.2 × 6.8-8.4 μm, Q=1.0–1.3, rhomboid in side-view with broad, blunt angles. Basidia 35–45 × 11–15 μm, 4-spored, with clamp-connections. Lamella edge heterogeneous. Cheilocystidia 25–70 × 9–17 μm, lageniform to fusiform, mixed with basidia. Pleurocystidia absent. Pileipellis a cutis of repent, cylindrical, 2.0–6.0 μm wide hyphae with weak parietal pigment. No differentiated caulocystidia observed. Clamp-connections seen only in the hymenium, elsewhere absent.
Habitat and distribution: in humus, along roadside in evergreen laurel forest, and in poor, semi-arid grassland. Known so far from the type locality in Spain and from one location with two collections in Germany.
Comments: Entoloma laurisilvae resembles E. prismatospermum, which has smaller spores, different cheilocystidia, darker colours, and a farinaceous-rancid smell and taste. Entoloma laurisilvae seems also to be a more southern, thermophilous species which is genetically is distant from all studied species in the /Rhombisporum clade, differing from its closest species E. perrhombisporum by 43 substitution and indel positions in the ITS region, with a similarity of 93%.
Additional specimens examined: Germany, Rheinland-Pfalz, Simmertal, Horbach, poor, semi-arid grassland on Devonian greenschist, 28 Oct 2017, K. Reschke, KaiR1041 (L-0607536), KaiR1043.
Entoloma cocles (Fr.) Noordel., Persoonia 11(2): 149 (1981) Fig. 15
MycoBank: MB 112037
≡ Agaricus cocles Fr., Epicr. syst. mycol. (Upsaliae): 158 (1838) – Rhodophyllus cocles (Fr.) Quél., Enchir. fung. (Paris): 65 (1886) – Nolanea cocles (Fr.) Gillet, Hyménomycètes (Alençon): 422 (1876) [1878].
Misapplied name: Entoloma insolitum s. Schmidt et al. (2013).
Neotype (designated here): Germany, Geyer, Hermannsdorfer Wiesen, in a flooded, peaty meadow, amongst Sphagnum sp., 2 Oct 2020, A. Karich, R. Ullrich, E. Tomschke, GLM-F128994. MycoBank MBT 10004484. GenBank No. OL853711 (ITS).
Etymology: cocles (Lat.) = one-eyed (as the cyclops), referring to the dark central spot at the centre of the pileus.
Original diagnosis: (translated from Latin): Pileus thin-fleshed, campanulate, radially grooved-sulcate, glabrous, with darker, squamulose centre, then expanding; stipe thin, glabrous, lead grey; lamellae adnate, broad, distant, white then reddish. In the expanded fungus the lamellae appear free: cfr. Secr. ll. 553. In non fertile hills, very distinct, but rare, easily overlooked. Pileus shiny, umber brown, with blackish brown grooves.
Description: Pileus 10–25 mm broad, truncated conical or subglobose to convex then expanding, with slightly depressed to umbilicate centre, sometimes weakly umbonate, with inflexed then straight, often crenulated margin, hygrophanous, when moist translucently striate up to middle or up to centre, very dark sepia brown to grey-brown with an almost black centre or central spot, slightly paler towards margin and between striae, minutely innately fibrillose or almost glabrous in marginal zone, at centre rugulose to granulose, or subsquamulose, slightly pallescent on drying. Lamellae L = 15–30, l = 0–3, moderately distant to distant, often somewhat thick, adnate, adnexed, or adnate-emarginate, ventricose, often broadly so and extending under pileus, pale grey then greyish pink, finally brownish pink, paler towards the minutely fimbriate, concolorous edge. Stipe 20–45 × 1–3 mm, slender cylindrical, sometimes broadened at base, yellow-brown or cream, olivaceous brown, or grey-brown, concolorous with or paler than pileus, sometimes darker towards base, smooth, glabrous, polished or innately silky-fibrillose, solid then fistulose. Context very thin, concolorous with surface or paler. Smell faint, herbaceous or farinaceous-spermatic, but sometimes strong when bruised. Taste mild or subfarinaceous. Basidiospores (9.0–)10.0–14.5(–18) × 7.5–10.5 μm, average 11.5–13.7 × 8.8–9.6 μm, Q=1.1–1.7, Qav.=1.25–1.5, (4–) 5–9 angled in side-view, sometimes more or less 4-angled, almost rhomboid. Basidia 29–55 × 10–15 μm, 4-spored, with clamp-connections. Lamella edge heterogeneous. Cheilocystidia 35–90(–100) × 7.5–22 × 2.5–8 μm, lageniform to lecythiform, rarely irregularly tibiiform, with acute or capitate apex, thin-walled, numerous, but always mixed with basidia. Pleurocystidia present or absent, similar to the cheilocystidia, but slightly smaller, 60–90 × 12.5–20 μm, thin-walled, and easily collapsing and often hard to find in dried material. Hymenophoral trama, regular, made up of long, cylindrical to fusiform hyphal elements, up to 230 × 6–11 μm, with brown or hyaline walls. Pileipellis a cutis with transitions to a trichoderm, made up of inflated hyphae, up to 20 μm wide. Pigment brown, intracellular in pileipellis. Stipitipellis a thin cutis of narrow, cylindrical hyphae, 5.5–9.5 μm wide. No differentiated caulocystidia found. Clamp-connections present in the hymenium, absent elsewhere.
Habitat and distribution: In poorly managed, semi-natural grassland, heaths, at the margin of Sphagnum bogs etc., on acid, peaty soil and calcareous loam, as well as in deciduous forest on loamy soil, from August to October. Sequenced records known from Germany, the Netherlands, and Norway, but probably widely distributed in northwestern Europe.
Comments: Entoloma cocles is placed basal to the /Rhombisporum clade based on our phylogenetic analysis (Figs. 1, 2), and in many cases the name E. cocles was applied to several species in the /Rhombisporum clade (cf. phylogenetic tree). For this reason, it seemed relevant to include E. cocles in this paper, and to fix the current concept with a neotype. The concept of Entoloma cocles goes back to the interpretation of Ricken (1915), Kühner and Romagnesi (1953), Einhellinger (1976), and Noordeloos (1987, 1992) which is not conflicting with the protologue. The current description is based primarily on sequenced material. Characteristic for this species are the dark colours and large spores, much larger, and normally distinctly heterodiametrical in contrast to the rhomboid or subisodiametrical spores of the /Rhombisporum clade. Phylogenetically, Entoloma cocles is distant from all members of the genus Entoloma possessing at least 12% ITS sequence dissimilarity from other species.
Additional specimens examined: Germany, Brandenburg, Bärenklau, NSG Kleinsee, in damp place with Alnus and Quercus, 23 Sept 2012, A. Karich, R. Ullrich, GLM-F128991; Bayern, Landkreis Garmisch-Partenkirchen, Gemeinde Krün, Klais, Geiflschodel, calcareous semi-natural grassland, 13 Oct 2018, M. Dondl, L-0607513; Norway, Troms, Karlsøy, Reinøya, Nordeidet, calcareous semi-natural grassland (sheep pasture), 6 Sept 1999, V. Ravolainen, TROM-F-610294; Møre og Romsdal, Tingvoll, Holmeide: east of the pasture of Fredrik, in lawn, 26 Aug 2005, G. Gaarder, GGA4464 (O-F-281107). The Netherlands, Utrecht, Zeist, Groot Heidestein, in Calluna heath on poor, acid soil, 9 Oct 2019, M. Veerkamp, L-0607620.
Key to the European species
1 Basidiospores more or less isodiametrical to subheterodiametrical, either rhomboid or pentagonal, Qav=1.0–1.3, small to medium large (never >12 μm long) (/Rhombisporum clade)...........................................................2
1* Basidiospores heterodiametrical (rarely rhomboid), (9.0–)10.0–14.5(–18) × 7.5–10.5 μm Qav = 1.25–1.45, irregularly 5–7 angled........................................................E. cocles
2 Basidiospores small, average length < 8.5 μm....................3
2* Basidiospores larger, average length > 8.5 μm..................4
3 Cheilocystidia with clamp-connections...........................................................E. cf. prismatospermum
3* Cheilocystidia without clamp-connections....E. laurisilvae
4 Differentiated caulocystidia present, at least at the apex of stipe.........................................................................................5
4* Differentiated caulocystidia absent ....................................8
5 Clamp-connections present, basidiospores rhomboid, more or less 4-sided in side view; pileus medium to dark brown .....6
5* Clamp-connections absent; Basidiospores pentagonal, isodiametrical; basidiomata pale yellowish brown...................................................................................E. lunare
6 Caulocystidia present along the entire stipe, length >100 μm, pleurocystidia present near lamella edge..........................................................E. caulocystidiatum
6* Caulocystidia at apex only, considerably shorter than 100 μm, pleurocystidia absent .......................................................7
7 Stipe polished or with scattered fibrils; basidiopores 7.5–11.0(–12.5) x 7.3-9.2 μm, average 8.0–9.5 × 7.0–8.2 μm, Q=1.0–1.3, Qav = 1.15.............................E. rhombisporoides
7* Stipe slightly to densely fibrillose-tomentose; basidiospores 8.5–11.2 × 8.3–10.7 μm, in average 9.8 × 9.1 μm, Q=1–1.2, Qav = 1.08.................................E. rhombiibericum
8 Basidiospores rhomboid ......................................................9
8* Basidiospores pentagonal or mixed rhomboid/pentagonal..10
9 Basidiospores with well-defined 4 angles in side-view; cheilocystidia inconspicuous, thin-walled and easily collapsing, often not much larger than basidioles, and often hard to find in dried material....................................E. rhombisporum
9* Basidiospores with obtuse angles; cheilocystidia conspicuous, thick-walled, often larger than basidia and protruding from hymenium......................................E. perrhombisporum
10 Basidiospores mixed rhomboid to pentagonal, 4–5-sided in side view................................................................................11
10* Basidiospores pentagonal in majority............................12
11 Basidiospores 8–11 × 6.5–9 μm, Q=1.0–1.5, average 9.2–10 × 7.5–8 μm, congophilous plaques abundant in stipitipellis; cheilocystidia rather fragile and easily collapsing ..............................................................E. pararhombisporum
11* Basidiospores 7.5–11 × 7.0–10.5 μm, Q=1.0–1.4, average 9.0–10.5 × 7.5–8.5 μm, Qav = 1.15–1.2, without congophilous plaques in stipitipellis; cheilocystidia not easily collapsing.....................................................E. subcuboideum
12 Clamp-connections absent..........................E. pentagonale
12* Clamp-connections present............................................13
13 Lamella edge heterogeneous; basidiospores in majority 5-angled; species widely distributed in lowland and colline habitats........................................................E. sororpratulense
13* Lamella edge sterile; basidiospores 4–6-angled with a fair number of 5-angled ones; (sub-) alpine or subarctic species.................................................................E. pratulense
Ecological aspects of the /Rhombisporum clade in relation to conservation issues
Many fungal species are characteristic for semi-natural grasslands, i.e., traditionally managed sites being grazed or mown, not or poorly manured (e.g., Nitare 1988). Typical bioindicators species for these habitats are many species of Clavariaceae, Hygrocybe sensu lato, Entoloma from e.g., subgenera Cyanula and Nolanea, Geoglossaceae/Microglossum, sometimes referred to with the acronym “CHEG-species”; or even “CHEGD-species” by including Dermoloma (e.g., Nitare 1988; Noordeloos 1992; Griffith et al. 2013). Semi-natural grasslands are among the most threatened habitat types in Europe; several grassland types are present on the EU red list of habitats (Janssen et al. 2016). In western Europe, a habitat loss of approximately 90% the last 75 years is estimated in semi-natural grasslands (Griffith et al. 2013), and many European “CHEG species” are currently present on the global red list (IUCN 2021).
Most species of the /Rhombisporum clade treated here seem to be more or less bound to semi-natural grasslands. In Norway, habitat information on 124 “CHEG-species” was analysed by Jordal et al. (2016). They found that 155 of 178 records with habitat information on Entoloma rhombisporum sensu lato (not sequenced), were from semi-natural grasslands, 8 from other grasslands like lawns and churchyards, and 15 from rich forests. Among 110 collections labelled E. pratulense, 103 were from semi-natural grassland and 7 from rich forests (Jordal et al. 2016). In our sequenced material we can see the same pattern. According to present, sequence-verified data, Entoloma perrhombisporum, E. subcuboideum, E. pararhombisporum, E. rhombisporoides, E. caulocystidiatum, E. pentagonale, E. lunare, and E. pratulense are mostly or exclusively found in calcareous semi-natural grasslands, some also in similar open herb-rich types like rich fen margins, churchyards, lawns or xerothermophilous, calcareous natural grasslands. Entoloma pentagonale is the species with most records from xerothermophilous, natural grasslands on coastal limestone outcrops, also a strongly threatened habitat type which was recently given high priority in the nature management of Norway. Entoloma rhombisporum, E. cf. prismatospermum, E. sororpratulense, E. laurisilvae and E. cocles are also found in semi-natural grasslands but have been recorded in (rich/calcareous) forests. Entoloma rhombiibericum is only known from the type locality in montane Buxus vegetation on basic-calcareous soils in Spain.
Only one species is found outside Europe: E. pratulense with one sequence from Quebec, Canada. Very few Entoloma species are transatlantic and this is more likely to occur in northern species (Voitk et al. 2020b), like E. pratulense.
At present, E. rhombisporum sensu lato is red listed as VU (vulnerable) in Norway, The Netherlands and Austria, and as EN (strongly threatened) in Denmark (see http://www.eccf.eu/) As this group is now split into many species, each of them will be even more rare and possibly qualify to a higher category by later revisions of the red lists, and as more information becomes available. Entoloma pratulense sensu lato is red-listed as VU in Norway, DD (data deficient) in Sweden, and CR (critically endangered) in Austria and Denmark (see http://www.eccf.eu/). The species treated here thus deserve high attention in conservation biology and habitat management.
Data availability
MycoBank, GenBank.
References
Alvarado P, Moreno G, Vizzini A, Consiglio G, Manjón JL, Setti L (2015) Atractosporocybe, Leucocybe and Rhizocybe: three new clitocyboid genera in the Tricholomatoid clade (Agaricales) with notes on Clitocybe and Lepista. Mycologia 107(1):123–136
Borchsenius F (2009) FastGap 1.2. Department of Bio-sciences, Aarhus University, Denmark. Published online at: https://www.aubot.dk/FastGap_home.htm Accessed 20 Aug 2021
Dima B, Lindström H, Liimatainen K, Olson A, Soop K, Kytövuori I, Dahlberg A, Niskanen T (2016) Typification of Friesian names in Cortinarius sections Anomali, Spilomei, and Bolares, and description of two new species from northern Europe. Mycol Prog 15:903–919
Einhellinger (1976) Die Pilze in primären und sekundären Pflanzengesellschaften Oberbayerischen Moore. Ber. Bayer. Bot. Ges. 47:75–149
Gardes M, Bruns TD (1993) ITS primers with enhanced specifity for basidiomycetes – application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gouy M, Guindon S, Gascuel O (2010) SeaView version 4: a multiplatform graphical user interface for sequence alignment and phylogenetic tree building. Mol Biol Evol 27:221–224
Griffith GW, Gamarra JGP, Holden EM, Mitchel D, Graham A, Evans DA, Evans SE, Aron C, Noordeloos ME, Kirk PM, Smith SLN, Woods RG, Hale AD, Easton GL, Ratkowsky DA, Stevens DP, Halbwachs H (2013) The international conservation importance of Welsh ‘waxcap’ grasslands. Mycosphere 4:969–984
Horak E (1976) On cuboid-spored species of Entoloma. Sydowia 28:171–236
IUCN (2021) The IUCN Red List of Threatened Species. Version 2021–3. https://www.iucnredlist.org
Janssen JAM, Rodwell JS, García Criado M, Gubbay S, Haynes T, Nieto A, Sanders N, Landucci F, Loidi J, Ssymank A, Tahvanainen T, Valderrabano M, Acosta A, Aronsson M, Arts G, Attorre F, Bergmeier E, Bijlsma R-J, Bioret F, Biţă-Nicolae C, Biurrun I, Calix M, Capelo J, Čarni A, Chytrý M, Dengler J, Dimopoulos P, Essl F, Gardfjell H, Gigante D, Giusso del Galdo G, Hájek M, Jansen F, Jansen J, Kapfer J, Mickolajczak A, Molina JA, Molnár Z, Paternoster D, Piernik A, Poulin B, Renaux B, Schaminée JHJ, Šumberová K, Toivonen H, Tonteri T, Tsiripidis I, Tzonev R, Valachovič M (2016) European Red List of Habitats: Part 2. Terrestrial and freshwater habitats. European Union. https://doi.org/10.2779/091372
Jordal JB, Evju M, Gaarder G (2016) Habitat specificity of selected grassland fungi in Norway. Agarica 37:5–32
Kalinina LB (2021) Agaricoid fungi of the Polistovsky reserve (annotated list of species). KMK Scientific Press, Moscow [Flora and Fauna of Reserves. Issue 143]
Karstedt F, Capelari M, Baroni TJ, Largent DJ, Bergeman S (2019) Phylogenetic and morphological analyses of species of the Entolomataceae (Agaricales, Basidiomycota) with cuboid basidiospores. Phytotaxa 391(1):1–27
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780
Kühner R, Boursier J (1929) La forme des spores chez les agarics rhodogoniosporés (genre Rhodophyllus Quél.). Bull trimest Soc Mycol Fr 45:264–277
Kühner R, Romagnesi H (1953) Flore analytique des champignons supérieurs (Agarics, Bolets, Chanterelles). Masson & co., Paris, 557 pp
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol Biol Evol 33(7):1870–1874
Larsson E, Jacobsson S (2004) The controversy over Hygrophorus cossus settled using ITS sequence data from 200-year-old type material. Mycol Res 108:781–786
Larsson E, Vauras J, Cripps C (2018) Inocybe praetervisa group: A clade of four closely related species with partly different geographical distribution ranges in Europe. Mycoscience 59:277–287
Morozova OV, Alexandrova AV, Popov ES, Malysheva EF (2016) New data on the agaricoid basidiomycetes of the Tver Region. Novosti Sist Nizsh Rast 50:174–186
Morozova OV, Noordeloos ME, Popov ES, Alexandrova AV (2018) Three new species within the genus Entoloma (Basidiomycota, Agaricales) with clamped basidia and a serrulatum-type lamellae edge, and their phylogenetic position. Mycol Prog 17:381–392
Nagy LG, Kocsubé S, Csanádi Z, Kovács GM, Petkovits T, Cs V, Papp T (2012) Re-mind the gap! Insertion-deletion data reveal neglected phylogenetic potential of the nuclear ribosomal internal transcribed spacer (ITS) of fungi. PLoS One 7:e49794. https://doi.org/10.1371/journal.pone.0049794
Nitare J (1988) Jordtungor, en svampgrupp på tillbakagång i naturliga fodermarker [Earth tongues, a declining group of fungi in semi-natural grasslands]. Svensk Botanisk Tidskrift 82:341–368
Noordeloos ME (1980) Entoloma subgenus Nolanea in the Netherlands and adjacent regions with a reconnaissance of its remaining taxa in Europe. Persoonia 10(4):427–534
Noordeloos ME (1987) Entoloma (Agaricales) in Europe. Synopsis and keys to all species and a monograph of the subgenera Trichopilus, Inocephalus, Alboleptonia, Leptonia, Paraleptonia, and Omphaliopsis. Beih Nova Hedw 91:1–419
Noordeloos ME (1992) Entoloma s.l. Fungi Europaei, vol. 5. Libreria editrice Giovanna Biella, Saronno, Italy, 960 pp
Noordeloos ME, Dima B, Weholt Ø, Eidissen SE, Lorås J, Brandrud TE (2017) Entoloma chamaemori (Entolomataceae, Basidiomycota) – a new boreal species, with isolated phylogenetic position. Phytotaxa 298(3):289–295
Pegler DN, Young TWK (1979) Spore form and phylogeny of Entolomataceae. Beih Sydowia 8:290–303
Reschke K, Popa F, Yang ZL, Kost G (2018) Diversity and taxonomy of Tricholoma species from Yunnan, China, and notes on species from Europe and North America. Mycologia 110:1081–1109
Ricken A (1915) Die Blätterpilze (Agaricaceae) Deutschlands und der angenzenden Länder besonders Österreichs und der Schweiz. I. Band. Text, 480 pp, II. Band. Abbildungen, VII + 122 pp, Leipzig
Romagnesi H (1974) Étude de quelques Rhodophylles. Travaux mycol. dédiés à R. Kühner. Bull Soc Linn Lyon 43:365–387
Ronquist F, Huelsenbeck JP (2003) MRBAYES 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Schmidt M, Karich A, Kellner H, Kleine J, Ullrich R (2013) Zu Entoloma-Funden mit Pleurozystiden (Entoloma insolitum agg.). Boletus 34(2):73–84
Silvestro D, Michalak I (2012) RaxmlGUI: a graphical front-end for RAxML. Org Divers Evol 12:335–337
Simmons MP, Ochoterena H, Carr TG (2001) Incorporation, relative homoplasy, and effect of gap characters in sequence-based phylogenetic analysis. Syst Biol 50(3):454–462
Turland NJ, Wiersema JH, Barrie FR, Greuter W, Hawksworth DL, Herendeen PS, Knapp S, Kusber W-H, Li D-Z, Marhold K, May TW, McNeill J, Monro AM, Prado J, Price MJ, Smith GF (eds) (2018) International Code of Nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Vegetabile 159. Koeltz Botanical Books, Glashütten
Vila J, Caballero F (2009) Entoloma nuevos o interesantes de la Península Ibérica (2). Fungi non Delineati. Pars XLV. Edizione Candusso, Italy
Voitk A, Saar I, Lücking R, Moreau P-A, Corriol G, Krisai-Greilhuber I, Thorn RG, Hay CRJ, Moncada B, Gulden G (2020a) Surprising morphological, ecological and ITS sequence diversity in the Arrhenia acerosa complex (Basidiomycota: Agaricales: Hygrophoraceae). Sydowia 73:133–162
Voitk A, Saar I, Lodge DJ, Boertmann D, Berch SM, Larsson E (2020b) New species and reports of Cuphophyllus from northern North America compared with related Eurasian species. Mycologia 112:438–452
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: A guide to the methods and applications. Academic Press, New York, pp 315–322
Acknowledgements
The Foundation Rijksherbariumfonds Dr. E. Kits van Waveren provided funds for herbarium visits of MEN and part of the molecular work. Pablo Alvarado (ALVALAB, Santander, Spain), Gunnhild Marthinsen and Katriina Bendiksen (Natural History Museum, University of Oslo, Norway) from the NorBOL team, and Ellen Larsson (University of Gothenburg, Sweden) are acknowledged for performing a large part of the molecular work. The research of BD was supported by the New National Excellence Program (ÚNKP-20-4) of the Hungarian Ministry for Innovation and Technology and by the ELTE Thematic Excellence Programme 2020 (TKP2020-IKA-05), both supported by the National Research, Development and Innovation Office. KR thanks Jan-Olof Tedebrand and participants of the Russulales Congress 2018 in Borgsjö for support. The Willkomm-Stiftung, Frankfurt, provided a travelling grant for KR to a congress including fieldwork in Sweden. The Norwegian Environment Agency funded parts of the field work for JBJ. The study of O.V. Morozova was carried out within the framework of a research project of the Komarov Botanical Institute RAS (AAAA-A-A19-119020890079-6) using equipment of its Core Facility Centre “Cell and Molecular Technologies in Plant Science”. The Austrian Ministry of Science supported sequencing within the Austrian Barcoding Project ABOL. We are greatful to Jacques Landry for providing us the photo of Entoloma pratulense.
Code availability
Not applicable.
Funding
Open access funding provided by Eötvös Loránd University. ELTE Thematic Excellence Programme 2020 (TKP2020-IKA-05) supported by the National Research, Development and Innovation Office, New National Excellence Program (ÚNKP-20-4) of the Hungarian Ministry of Human Capacities; research project of the Komarov Botanical Institute RAS (AAAA-A-A19-119020890079-6) using equipment of its Core Facility Centre “Cell and Molecular Technologies in Plant Science”; foundation Rijksherbariumfonds Dr. E. Kits van Waveren.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design as well as to material preparation, data collection and discussions. Molecular lab work was performed by B. Dima, O. Morozova, K. Reschke, and I. Krisai-Greilhuber. Phylogenetic analyses were performed by B. Dima. Macroscopic analyses were done by M. E. Noordeloos, J. B. Jordal, O. Morozova, Ø. Weholt, T. E. Brandrud, J. Vila, A. Karich, and K. Reschke. Microscopic analyses were done by G. M. Jansen, M. E. Noordeloos, and J. Vila. Ecological and nature conservation aspects were summarized by J.B. Jordal and T.E. Brandrud. Initial identification key based on microcharacters was provided by G. M. Jansen. The manuscript was written by M. E. Noordeloos and B. Dima. All authors commented on the manuscript as well as reading and approving the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Zhu-Liang Yang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(FAS 124 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Noordeloos, M.E., Jansen, G.M., Jordal, J.B. et al. Molecular and morphological diversity in the /Rhombisporum clade of the genus Entoloma with a note on E. cocles. Mycol Progress 21, 48 (2022). https://doi.org/10.1007/s11557-022-01779-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-022-01779-z