Abstract
Cryptodicus muriformis and Schizoxylon gilenstamii (Stictidaceae, Ostropales, Lecanoromycetes, Ascomycota) are described as new to science from collections made in Europe (Sweden, Austria, Switzerland) and North America (Arizona), based on phylogenetic analyses of three loci. Both species show unique morphological characters that distinguish them from closely related species; Cryptodiscus muriformis, growing on decorticated or sometimes corticated branches of Picea abies and Pseudotsuga menziesii, that resembles C. foveolaris and C. tabularum in the yellowish-orange ascomatal disc, but is clearly distinguished by the muriform ascospores. Schizoxylon gilenstamii grows on dead stems of Globularia vulgaris and has a characteristic unique red-purple pigment in the epithecium and upper part of the excipulum that changes to bluish-green with KOH. We also report for the first time the anamorph of Stictis brunnescens.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The family Stictidaceae (Ostropales, Lecanoromycetes, Ascomycota) contains predominantly saprotrophic discomycetes, but also lichens, parasites, and endophytes (e.g., Gilenstam 1969; Fernández-Brime et al. 2011; Baloch et al. 2013; Aptroot et al. 2014; Jahn et al. 2017). Sherwood (1977a, 1977b) produced a detailed and still very useful worldwide taxonomic and nomenclatural revision of the family with main focus on taxa from the USA. With the advent of DNA technologies, molecular phylogenetic studies have been crucial to revise established generic limits and species concepts in the family, although these studies are still in their infancy. Wedin et al. (2004, 2005, 2006) investigated taxa living on Populus and Salix in Northern Scandinavia. They found that several species of Stictis and Schizoxylon can live either as lichens or as saprotrophs depending on the substrate they inhabit, a phenomenon they called “optional lichenization”. These results led to a widened concept of Stictis and Schizoxylon, which until then only included saprotrophic species. Wedin et al. (2006) pointed out that these two genera are potentially heterogeneous, but as molecular data was available only for a small number of species they could not pursue this further at that time. Finally, Cryptodiscus was studied in detail in Scandinavia by Baloch et al. (2009), resulting in a revision of the generic concept and the description of a new species.
Our knowledge of the Stictidaceae is obviously still very limited. The family contains small and generally drought-tolerant fungi, which are easily overlooked and rarely collected. For this reason, in recent years we have continued exploring plant debris and, as a result, we found specimens of stictidaceous fungi that did not fit any described taxa. This paper introduces two new species collected in Sweden, Austria, Switzerland and USA, and provides detailed descriptions and a three-locus phylogeny that places them in the here adopted genera within the context of the Stictidaceae.
Anamorphs have been reported from several Stictidaceae species. Pycnidial anamorphs have been found in Acarosporina microspora, Conotrema urceolatum, Cryptodiscus gloeocapsa, Cyanodermella oleoligni, and Stictis radiata (e.g., Gilenstam 1969; Sherwood 1977a; Johnston 1983; Baloch et al. 2009; Nieuwenhuijzen et al. 2016). Johnston (1983) established the connection between anamorphs and teleomorphs of several Stictis species using cultures. In all cases, the anamorphs described were pycnidia. As a result from recent field work in Sweden, we found several conidiomata, which are presumably the anamorph of Stictis brunnescens. We have also used the three-locus phylogeny to test the connection between the anamorph and teleomorph.
Materials and methods
Morphological and anatomical studies
Specimens were examined morphologically and anatomically and preserved in the herbaria GJO, S, UPS (herbarium codes according to Thiers [2017, continuously updated]) and H. O. Baral’s private herbarium (H.B.). Hand-cut sections mounted in water were used to measure margin, hymenium, asci, paraphyses, and ascospores. Freezing microtome sections, 20–30 μm thick, mounted in water or stained with lactic blue or Congo red, were produced for a more detailed study of the anatomy. Lugol’s iodine solution, before and after treatment with 10% potassium hydroxide (KOH), was used to detect amyloid structures. Thereby, “type RR” means that the ascus wall reacts red at both low and high concentration before applying KOH. For the anatomical features measurements, the extreme values are shown after rejecting 10% of the highest and the lowest values; the highest and lowest absolute values are given in parentheses.
DNA extraction, PCR amplification, and sequencing
Total DNA was extracted from a selected number of samples with the DNeasy Plant Mini Kit (Qiagen, Germany) following the manufacturer’s instructions or with the E.Z.N.A. Forensic DNA Kit following an improved protocol (Stielow et al. 2013).
The selected markers for this study were the internal transcribed spacer complete repeat (ITS), the first part (D1–D2 region, ca. 600 bp) of the large subunit (nLSU) of the nuclear ribosomal DNA, and the small subunit of the mitochondrial ribosomal DNA (mtSSU). Primer combinations for amplification of the three loci were: ITS1F (Gardes and Bruns 1993) and ITS2, ITS3, and ITS4 (White et al. 1990) for ≈ 0.6 kb ITS, LR0R and LR3 (Vilgalys and Hester 1990) for ≈ 0.6 kb nLSU, and mrSSU1 and mrSSU3R (Zoller et al. 1999) for the ≈ 0.8 kb mtSSU. Symmetric PCR amplifications were performed using IllustraTM Hot Start PCR beads, according to the manufacturer’s instructions, with the following settings for the three loci: initial denaturation 94 °C for 5 min, followed by five cycles (94 °C for 45 s, 54 °C for 30 s, and 72 °C for 60 s), 30 cycles (94 °C for 45 s, 52 °C for 30 s, and 72 °C for 60 s), and a final extension 72 °C for 8 min. After examination by gel electrophoresis, amplification products were purified using ExoSAP-IT (USB Corp., U.S.A.). Sequencing of both strands was performed with the Big Dye Terminator technology kit v3.1 (ABI PRISM, U.S.A.) using the PCR primers.
Sequence alignment and phylogenetic analyses
Sequence fragments were edited and assembled with Sequencher 4.9 (Gene Codes Corp., USA) and consensus sequences subjected to BLAST searches to rule out amplification of contaminants. The newly produced sequences were included in individual alignments for each locus, together with sequences retrieved from GenBank representing morphologically similar species from the family Stictidaceae. The taxon Karstenia rhopaloides was selected as an outgroup as it is closely related to Stictidaceae, but Baloch et al. (2009) showed that it does not belong to the family. Sequences were aligned with the online version of MAFFT v7 (http://mafft.cbrc.jp/alignment/server/) using the algorithms L-INS-I for ITS and nLSU, and E-INS-I for mtSSU. The ambiguously aligned regions were identified using the G-blocks v0.91b (http://molevol.cmima.csic.es/castresana/Gblocks_server.html) and removed before analysis. Alignments were submitted to TreeBase (http://www.treebase.org; ID number 21812).
Phylogenetic analyses were performed for each locus individually with RAxML v 8.2.0 (Stamatakis 2014), using the rapid bootstrap analysis and search for best-scoring maximum likelihood algorithm (−f a), the GTRGAMMA model and 1000 bootstrap replicates (ML-BS). Then, the individual phylogenies were compared to assess congruence, considering there to be a significant conflict when two alternative relationships corresponding to different loci were both supported with bootstrap values ≥70% (Mason-Gamer and Kellog 1996). As no significant conflicts were detected, the three alignments were concatenated into a single dataset with five partitions (ITS1, 5.8S, ITS2, nLSU and mtSSU). Phylogenetic relationships and confidence were inferred on the combined dataset using maximum likelihood (ML) and Bayesian inference (BI). For the ML analysis, the same settings used for the individual gene analyses were used in RAxML, with the GTRGAMMA model applied to each partition. The Bayesian inference of the phylogeny was carried out by a Markov chain Monte Carlo with Metropolis coupling (MCMCMC) as implemented in MrBayes v3.2.6 (Ronquist et al. 2012). The substitution models estimated using the Bayesian Information Criterion (BIC) as currently implemented in jModeltest v.0.1.1 (Posada 2008) were: GTR + I + G for ITS1 and mtSSU, K80 + I for 5.8S and SYM + G for ITS2 and nLSU. Two parallel runs with four independent chains each were conducted for 20 million generations, with trees sampled at intervals of 500 generations and a random starting tree. A burn-in sample of the first 25% of the trees was discarded for each run, and the remaining trees were used to estimate branch lengths and posterior probabilities (BI-PP). All analyses were run in the CIPRES Science Gateway.
Results
Phylogenetic analyses of combined sequence data
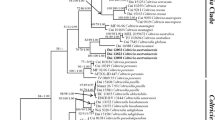
The combined dataset comprised 43 specimens, for which 41 ITS, 37 nLSU and 43 mtSSU sequences were available. The 21 newly generated sequences were submitted to GenBank and the voucher information and the accession numbers are available in Table 1. The data matrix contained 1381 aligned characters after the exclusion of 901 sites corresponding to ambiguously aligned regions, introns, and both ends of the alignments. Of the total number of analysed characters, 913 were constant and 468 were variable, of which 387 were parsimony-informative characters.
Both ML and BI analyses of the combined dataset resulted in very similar topologies; as the ML tree that resulted was slightly more resolved, we show here the best-scoring RAxML tree with support values indicated for both analyses (Fig. 1). Internodes were considered significantly supported when ML-BS ≥ 70% and/or BI-PP ≥ 0.95.
The monophyly of the putative new species Cryptodiscus muriformis and Schizoxylon gilenstamii received maximum support in both analyses (ML-BS: 100%, BI-PP: 1). Cryptodiscus muriformis appeared nested inside the Cryptodiscus clade with a highly supported (ML-BS: 99%, BI-PP: 1) sister relationship with C. tabularum. Schizoxylon gilenstamii appeared nested inside Schizoxylon and sister to the only specimen of S. berkeleyanum present in our phylogeny. The three samples belonging to a green conidiophore structure clustered unequivocally with the specimens of Stictis brunnescens (ML-BS: 100%, BI-PP: 1).
Taxonomy
Cryptodiscus muriformis Fdez.-Brime, Olariaga, Baral, Friebes, Jaklitsch, Senn-Irlet & Wedin, sp. nov. Fig. 2.
Cryptodicus muriformis a-b Habit, the mature ascomata remain mostly immersed in the substrate (holotype UPS F-647154) c Ascoma cross-section (holotype UPS F-647154) d Ascoma wall structure with textura angularis (holotype UPS F-647154) e Hymenium detail with 4-spored asci (GJO 85613) f Hymenium red reaction after applying Lugol’s solution (GJO 85613) g Ascus apex (GJO 85614) h Amyloid ascus after applying Lugol’s solution with KOH-pretreatment (GJO 85613) i Inamyloid ascus after applying Lugol’s solution with KOH-pretreatment (H.B. 6773) j-k Muriform hyaline spores (GJO 85614). Scale is indicated by bars (a: 1 mm; b: 0.5 mm; c: 100 μm; d-k: 10 μm)
MycoBank: MB 823292.
Diagnosis: similar to the phragmosporous Cryptodiscus tabularum but distinguished by the ca. 2–3× broader, ellipsoid, muriform ascospores (19–)21–30(−36) × (8–)9–13(−15) μm.
Etymology: the species name refers to the characteristic muriform ascospores.
Type: Sweden. Uppland, Ekerö par., Hagalundsskogen, 59°22′53″N 17°29′44″E, 50 m, on thin, decorticated branches of Picea abies still attached to the tree, 19.IX.2013, leg. I. Olariaga (UPS F-647154, holotype). GenBank: MG281962 ITS, MG281962 nLSU rDNA, MG281972 mtSSU rDNA.
Apothecia at first round, opening at the apex by a 0.2–0.3 mm wide pore, becoming ellipsoid when mature, 0.2–0.8 × 0.2–0.5 mm, 130–170 μm thick, erumpent, remaining semi-immersed, with the margin partly protruding beyond the surface layers of wood or bark; disc yellowish-orange, concave; margin hyaline to pale orange-brown, entire or sometimes slightly lacerate, protruding 15–90 μm beyond hymenial surface, 45–60 μm thick, consisting of small, isodiametrical to elongate, gelatinized, guttulate cells oriented parallel to the outside, periphysoids lacking, without crystals within; hymenium hyaline, subhymenium pale yellow; paraphyses 1–2 μm diam., thread-like, with high lipid content, apex generally unbranched, a few forked at the subterminal cell, smooth, apically slightly inflated and densely septate, partly moniliform, sometimes with a sparse, hyaline exudate; asci †70–110(−125) × (10–)12–18(−21) μm, cylindric-clavate, short-stalked, initially 8-spored but four spores early aborted, apex hemispherical, with a †4–7(−8) μm thick apical dome (at maturity often only †1.5–2.5 μm), lateral wall †~1–3 μm thick, entire outer wall and apical ring deeply red in Lugol (I+ hemiamyloid, type RR), KOH/ Lugol deeply blue, amyloidity of apical dome or also outer wall sometimes absent; ascospores hyaline, ellipsoid, muriform, with (4–)5–7(−8) transverse and 1–2(−3) longitudinal septa, slightly to strongly constricted at the septa, multiguttulate, smooth, */†(19–)21–30(−36) × (8–)9–13(−15) μm.
Substrate: on decorticated or rarely corticated, dead, 8–20 mm thick branches of Picea abies and once on Pseudotsuga menziesii, on wood, rarely bark; found from 0.2 to 1.8 m above the ground, still attached to living or fallen trunks. Associated species: Mellitiosporium propolidoides, Orbilia alpigena, Propolis rhodoleuca, P. hillmanniana and different lichens, in North America Claussenomyces pseudotsugae, Dactylospora sp., cf. Flavoparmelia sp., Orbilia spermoides, O. vinosa and Propolis cf. strobilina.
Known distribution: Europe: Sweden, Austria, Switzerland; North America: USA, Arizona.
Additional specimens examined: Austria. Osttirol, near Matrei, northeast of Zedlach, Zedlacher Paradies, MTB 8941/3 (grid reference), ca. 47°0′25″N 12°30′30″E, ca. 1500 m, on branch of Picea abies, 28.VIII.2000, leg. W. Jaklitsch 1536 (H.B. 6773). Steiermark, district Deutschlandsberg, Koralpe, shortly W of Grünangerhütte, at the trail to Bärentalalm, 46°48′43″N 14°59′57″E, 1660 m, on dead, decorticated branch of Picea abies, 30.V.2014, leg. G. Friebes (GJO 85613); Ibidem, 46°48′45″N 15°00′25″E, 1620 m, on decorticated branch of Picea abies, 6.V.2016, leg. G. Friebes; Ibidem, shortly NE of Grünangerhütte, at the trail to Grünangerhütte, 46°48′52″N 15°00′52″E, 1540 m, 17.IV.2017, on decorticated branch of Picea abies, leg. G. Friebes (GJO 85614); Freiländer Alm, 46°54′51″N 15°03′23″E, 1433 m alt, on decorticated branches of Picea abies, 3.V.2017, leg. G. Friebes (GJO 86305). Switzerland. Graubünden, Lenzerheide, Scharmoins, 46°44′15.4″N 9°34′14.25″E, 1840 m, on decorticated branch of Picea abies, 28.VIII.2008, leg. B. Senn-Irlet 08/151 (Z). Ticino, Airolo, Pian Taiöi, 46°30′24.35″N 8°30′12.32″E, 1630 m, on decorticated branch of Picea abies, 17 IX.2014, leg. B. Senn-Irlet 14/83 (Z). Bern, Lauterbrunnen, Grütschalp, 46°35′35.68″N 7°53′19.81″E, 1595 m, on decorticated branch of Picea abies, 11.VI.2017, leg. B. Senn-Irlet 17/54 (Z). USA. Arizona, Southern Rocky Mountains, Mogollon Rim, Hannagan Meadow, Coronado Trail, 33°34′49″N 109°21′55″W, 2820 m, on corticated branch of Pseudotsuga menziesii, on bark, 2.VI.2003, leg. G. Marson (H.B. 9576d).
Schizoxylon gilenstamii Fdez.-Brime, Baloch & Wedin, sp. nov. Fig. 3.
Schizoxylon gilenstamii a Habit, ascomata with margins covered by whitish pruina (holotype S F300891) b Habit, ascomata that have lost the pruina (S F300893 photo) c-d Ascoma wall structure (holotype S F300891); note the red-purple pigment in the epithecium and the inner marginal part of the excipulum (c) reacting green with KOH (d) e Filiform spores arranged inside the asci, mounted in water stained with lactic blue (holotype S F300891) f Filiform spores emerging from an ascus (S F300892). Scale is indicated by bars (a-b: 1 mm; c-d: 40 μm; e-f: 20 μm)
MycoBank: MB 823298.
Diagnosis: black erumpent apothecia 0.4–0.8 mm in diam., in section medullary excipulum green, which continues below the subhymenium, with a red-purple pigment in the upper part of the excipulum and the epithecium that changes to KOH+ bluish-green.
Etymology: the species is named after the Swedish lichenologist and mycologist Gunnar Gilenstam. Gunnar has had a long interest in the Stictidaceae and has contributed enormously to our understanding of the biology and diversity of these fungi in northern Scandinavia.
Type: Sweden. Öland, Gynge Alvar Nature Reserve, open gravel-covered limestone pavement area (the SCIN-site), 56°32′31.7″N 16°28′42.2″E, 42 m, on dead stems of Globularia vulgaris, 15.V.2012, leg. M. Wedin 9490 (S F300891, holotype; K, isotype). GenBank: MG281968 ITS, MG281968 nLSU rDNA, MG281977 mtSSU rDNA.
Apothecia globose, ca. (0.2–)0.4–0.8(−1.7) mm in diam., erumpent, partly covered by substrate remnants; margin black, sometimes more or less covered by a fine whitish pruina, lacking crystals within, ca. 40 μm protruding, composed of two layers: outer layer carbonised, ca. (15–)20–50(−60) μm thick, including remnants from the host substrate, inner layer brown-olivaceous, ca. (20–)30–60(−120) μm thick, the inner part of the marginal excipulum with a red-purple pigment that changes to KOH+ bluish-green, periphysoids absent; inner layer continuing below the subhymenium by forming a ca. 50 μm thick, brown-olivaceous lower ascoma wall; disc almost invisible in young fruiting bodies, only exposed by a small apical pore making young apothecia resemble perithecia, then becoming wider, flat; hymenium ca. (215–)232–320(−335) μm high, Lugol- and KOH/Lugol-; asci cylindrical, †(125–)135–230(−240) × 7–10 μm; lateral wall ca. †2–2.5 μm thick, apical thickened to ca. †3–3.5 μm, 8-spored; paraphyses hyaline, septate, apically branched, 1 μm thick, generally exceeding the length of the asci, tips forming a prominent, ca. 25–50 μm thick epithecium showing the same red-purple pigment as the margin, changing to bluish-green in KOH; ascospores ca. †(100–)105–140(−165) × 1.5–2.5 μm, more or less coiled inside the asci, filiform, constricted at the septa, easily breaking apart into small non-septate part-spores ca. †3–4 μm long after discharge (part-spores were only seen outside the asci).
Substrate: on last year’s dead herbaceous stems (flower pedicles) of Globularia vulgaris.
Known distribution: Sweden, all collections are from the island of Öland.
Additional specimens examined (all on dead stems of Globularia vulgaris): Sweden. Öland, Gårdby par., N of Ekelunda karst-area, along the track from Skarpa Alby, open gravel-covered limestone pavement area, 56°35′22.9″N 16°35′12.5″E, ca. 40 m, 18.V.2012, leg. M. Wedin 9495 (S F300893); Ibidem, leg. M. Wedin 9496 (S F300894); Öland, Gräsgårds par., ca. 3 km east of S Möckleby, alvar (limestone plain) along the road from S Möckleby and Solberga, 56°21′06″N 16°27′49.6″E, ca. 20 m, 06.VI.2006, leg. E. Baloch SW230 (S F187523); Öland, Hulterstad par., Gösslunda, S of the road, open gravel-covered limestone pavement area, 56°29′18″N 16°31′58″E, ca. 22 m, 17.V.2013, leg. M. Wedin (S F300896); Ibidem, 56°29′21.1″N 16°31′56.9″E, 17.V.2013, leg. M. Westberg (S F300897); Öland, Resmo par., alvar ground, 25.VI.1952, leg. L. Holm (UPS F-124784); Ibidem, alvar between main road and lake Möckelmossen, 5.VI.1989, Holm & Holm 5438c (UPS F-124781); Ibidem, Gynge Alvar Nature Reserve, open gravel-covered limestone pavement area (the SCIN-site), 56°32′31.7″N 16°28′42.2″E, ca. 42 m, 3.IV.2012, leg. M. Wedin 9475 (S F300889); Ibidem, 15.V.2013, leg. M. Wedin 9512 (S F300895); Ibidem, 7.IV.2014, leg. M. Wedin 9549 (S F301515); Ibidem, 15.V.2012, leg. M. Wedin 9490 (S F300891); Ibidem, 56°32′21.2″N 16°28′41.7″E, ca. 40 m, 17.V.2012, leg. M. Wedin 9493 (S F300892).
Stictis brunnescens Gilenstam, Döring & Wedin. Fig. 4.
Stictis brunnescens, anamorph state a Habit, anamorph state with dull green surface, growing intermixed with the larger teleomorph state with ochraceous glossy disc and broader whitish margin (Wedin 7057) b Habit (S F302425) c Cross-section, note the presence of crystals in the lower part of the conidioma and in the margin (arrows), which is only partly visible in this photograph (S F301516) d Conidial masses surrounded by a gelatinous sheath, mounted in water stained with Congo red. Scale is indicated by bars (a-b: 1 mm; c: 100 μm; d: 10 μm)
A detailed description of the species is provided in Wedin et al. (2006). Here, we report for the first time an asexual morph. Conidiomata similar in structure to sporodochia, not pulvinate but cupulate, discoid, ca. 0.4–0.6 mm in diam, margin white and pruinose, with many crystals in section, conidial mass dull green; conidiophores 50–65 × 1.5–2 μm, anastomosing, branched apically; conidia subspherical, 1.5–3 μm diam, which are clustered in groups inside a sheath forming subglobose balls of ca. 12–17 μm diam.
Specimens with conidiomata examined: Sweden. Hälsingland, Forsa par., ca. 4.5 km SW Forsa church, along the road towards the summit of Mt. Storberget, close to a power-line, Pinus-dominated forest, 61°53′N 16°42′E, ca. 120 m, on wood of partly decorticated standing trunk of Populus tremula, 15.VII.2004, leg. M. Wedin 7594; Färila par., South of Ed, by the farm Östigården, coniferous and mixed forest growing in an abandoned agricultural land, 61°49′49.9″N 15°43′40.8″E, 190 m, on dead branch of standing Populus tremula, 04.IX.2013, leg. S. Fernández-Brime 1100 & M. Wedin (S F301516); Ibidem, 4 km West of Ed, South of lake Breasen, coniferous and mixed forest, 61°50′45″N 15°38′36.3″E, 205 m, under bark of Populus tremula, 04.IX.2013, leg. S. Fernández-Brime 1105 & M. Wedin (S F301517); Jämtland, Rätan par., Vitvattnet, SE part of the Lake Vitvattnet, Knätten, mixed coniferous forest close to the lake, 62°21′N 14°40′E, alt. ca. 360 m, on wood of partly decorticated standing trunk of Populus tremula, 11.VII.2004, leg. M. Wedin 7579; Lycksele Lappmark, Lycksele par., river Vindelälven, ca. 3 km S of Forsholm, just NW of Solbacken, along the road close to the mire, young Pinus-dominated forest, 64°45′N 19°03′E, ca. 240 m, on dead branch of young Populus tremula, 30.IX.2005, leg. M. Wedin 7921; Ibidem, Tärna par., Lake Övre Boksjön, S of Mt. Nils-Eriksberget, old growth Picea-dominated forest S of the road, 65°39′N, 15°52′E, alt. ca. 480 m, on wood of partly decorticated standing trunk of Salix caprea, 19.X.2002, leg. M. Wedin 7057; Norrbotten, Norrfjärden par., ca. 4.5 km NW of Sjulsmark along the forest road Abborrtjärnvägen ca. 550 m NNW of the lake Abborrtjärnen, in Picea-dominated mixed forest, 65°33.31′31.4″N 21°25′44.4″E, alt. ca. 145 m, on wood under bark of standing Populus tremula, 28.VIII.2009, leg. M. Wedin 8571 (S F302425).
Discussion
We introduce the new species Cryptodiscus muriformis and Schizoxylon gilenstamii in the family Stictidaceae, based on the presence of differences in morphological characters and phylogenetic inference.
Cryptodiscus muriformis is nested within the highly supported clade representing Cryptodiscus sensu Baloch et al. (2009) (Fig. 1). Its phylogenetic placement is supported by the presence of characteristic traits of Cryptodiscus (Sherwood 1977a; Baloch et al. 2009): apothecia round to ellipsoid in outline, persistently immersed in the substrate, deeply concave disc; margin without crystals and periphysoids and without differentiation into layers; asci cylindrical to clavate, 8-spored, with amyloid ascus wall (I+ red, KOH/I+ blue) and distinct apical dome, paraphyses filiform, simple; ascospores hyaline. So far, all described species of Cryptodiscus produce narrowly cylindric-ellipsoid to ellipsoid or ellipsoid-fusoid ascospores with 1–7 transversal septa. However, C. muriformis has broadly ellipsoid, muriform ascospores. Most Cryptodiscus are saprotrophs except for three lichenicolous (Pino-Bodas et al. 2017) and three lichenized species that form a thin thallus (Baloch et al. 2009). Green patches corresponding to algae were observed on the wood substrate and around the fruiting bodies of C. muriformis (Fig. 2 a-b); similar algal patches have been observed in other Cryptodiscus species (see Fig. 3 c, Baloch et al. 2009). However, there is currently nothing that suggests that these algae are associated with the fungus as they appear scattered throughout the wood rather than forming the algal clumps observed in other loosely lichenized Stictidaceae (Wedin et al. 2004).
Ascus amyloidity was found to be variable in C. muriformis. The apical dome includes a large, distinctly amyloid ring (Fig. 2h) in the samples from Sweden, Steiermark (Bärentalalm), and Osttirol, but when re-examining the latter collection, the asci were inamyloid even regarding the entire ascus wall (Fig. 2i). The amyloid ring was also absent in the samples from Bern and Arizona. In the apothecia with inamyloid asci from Osttirol a faintly amyloid perihymenial medullary excipulum was observed. However, not every collection was thoroughly studied for amyloidity. Contrary to the European samples, the apothecia in the sample from Arizona grew on bark of a corticated branch of Pseudotsuga, but morphologically it well concurred with those from Europe.
In the resulting phylogeny, C. muriformis appears closely related to C. foveolaris and C. tabularum. All three species share a distinctly yellowish-orange disc, and C. tabularum, the sister taxon of C. muriformis, is also similar in having the hymenium Lugol + red-brown. However, C. foveolaris has one-septate ascospores and grows on wood of deciduous trees, and C. tabularum has 3-septate ascospores and it has been found so far only on decorticated wood of Pinus sylvestris.
Schizoxylon gilenstamii clearly forms a monophyletic group with the other Schizoxylon species included in the phylogeny (Fig. 1). This is supported by the combination of morphological characters that Sherwood (1977a) stated as diagnostic for the genus: cartilaginous apothecia, with a margin lacking periphysoids, long cylindrical asci with a thickened apex opened by a broad pore, and long filiform multiseptate ascospores disarticulating into part-spores. All Schizoxylon species are saprotrophs, except for S. albescens that can live either as a saprotroph or as loosely lichenized with algal clumps associated with its fruiting bodies (Wedin et al. 2006; Muggia et al. 2011). No algae were observed associated with S. gilenstamii.
The new Schizoxylon species is, to our knowledge, the first in the genus having red-purple pigments in the epithecium and the upper part of the excipulum changing K+ bluish-green. Based on Sherwood’s monographs (1977a, b), which are the most complete studies of Schizoxylon up to date, other species having 8-spored asci less than 300 μm long and ascospores disarticulating in unicellular part-spores are S. ligustri, S. nigrellum (recorded from Norway), and S. sepincola. Schizoxylon ligustri has larger fruiting bodies that are not completely erumpent, but remain partly immersed in the substrate when mature, and are never globose but have flat grey disc and plane or reflexed pruinose white margin. Schizoxylon nigrellum and S. sepincola both have black erumpent apothecia of the same size as S. gilenstamii (0.5–0.8 mm in diam.). However, apart from lacking red-purple pigments in the epithecium and excipulum, in both species the margin in cross-section is brown (with colourless crystals in S. nigrellum), not brown-olivaceous as in S. gilenstamii, and have shorter asci and ascospores. All these species have been found on various types of woody substrate, but never on herbaceous remnants. Schizoxylon berkeleyanum is similar to S. gilenstamii because it also grows on herbaceous plant debris and may sometimes have non-septate part-spores, but the apothecia in S. berkeleyanum are larger and have a thick margin covered with white to yellow pruina that reacts with KOH+ yellow, and the paraphysis tips are olivaceous brown and not red-purple as is characteristic for S. gilenstamii.
Schizoxylon gilenstamii is an ephemeral species that grows on last year’s dead flower pedicles of Globularia vulgaris. Globularia is, in Sweden, considered a relict from a warmer period, and occurs here only in the alvar limestone pavement vegetation on the Baltic islands of Öland and Gotland. Schizoxylon gilenstamii should be searched for on Gotland and in the other parts of its host’s distribution area (Spain and southern France).
References
Aptroot A, Parnmen S, Lücking R, Baloch E, Jungbluth P, Caceres ME, Lumbsch HT (2014) Molecular phylogeny resolves a taxonomic misunderstanding and places Geisleria close to Absconditella s. str. (Ostropales: Stictidaceae). Lichenologist 46:115–128
Baloch E, Gilenstam G, Wedin M (2009) Phylogeny and classification of Cryptodiscus, with a taxonomic synopsis of the Swedish species. Fungal Diver 38:51–68
Baloch E, Gilenstam G, Wedin M (2013) The relationships of Odontotrema (Odontotremataceae) and the resurrected Sphaeropezia (Stictidaceae)—new combinations and three new Sphaeropezia species. Mycologia 105:384–397
Fernández-Brime S, Llimona X, Molnar K, Stenroos S, Högnabba F, Bjork C, Lutzoni F, Gaya E (2011) Expansion of the Stictidaceae by the addition of the saxicolous lichen-forming genus Ingvariella. Mycologia 103:755–763
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes – application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gilenstam G (1969) Studies in the genus Conotrema. Ark Bot 7:149–179
Jahn L, Schafhauser T, Pan S, Weber T, Wohlleben W, Fewer D, Sivonen K, Flor L, van Pée KH, Caradec T, Jacques P, Huijbers M, van Berkel W, Ludwig-Müller J (2017) Cyanodermella asteris sp. nov. (Ostropales) from the inflorescence axis of Aster tataricus. Mycotaxon 132:107–123
Mason-Gamer RJ, Kellogg EA (1996) Testing for phylogenetic conflict among molecular data sets in the tribe Triticeae (Gramineae). Syst Biol 45:524–545
Muggia L, Baloch E, Stabentheiner E, Grube M, Wedin M (2011) Photobiont association and genetic diversity of the optionally lichenized fungus Schizoxylon albescens. FEMS Microbiol Ecol 75:255–272
van Nieuwenhuijzen EJ, Miadlikowska JM, Houbraken JA, Adan OCG, Lutzoni FM, Samson RA (2016) Wood staining fungi revealed taxonomic novelties in Pezizomycotina: new order Superstratomycetales and new species Cyanodermella oleoligni. Stud Mycol 85:107–124
Pino-Bodas R, Zhurbenko MP, Stenroos S (2017) Phylogenetic placement within Lecanoromycetes of lichenicolous fungi associated with Cladonia and some other genera. Persoonia 39:91–117
Posada D (2008) jModelTest: phylogenetic model averaging. Mol Biol Evol 25:1253–1256
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Sherwood MA (1977a) The Ostropalean fungi. Mycotaxon 5:1–277
Sherwood MA (1977b) The Ostropalean fungi. II. Schizoxylon, with notes on Stictis, Acarosporina, Coccopeziza, and Carestiella. Mycotaxon 6:215–260
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313
Stielow B, Hensel G, Strobelt D, Makonde HM, Rohde M, Dijksterhuis J, Klenk HP, Göker M (2013) Hoffmannoscypha, a novel genus of brightly coloured, cupulate Pyronemataceae closely related to Tricharina and Geopora. Mycol Progress 12:675–686
Thiers B (2017, Continuously updated). Index Herbariorum: a global directory of public herbaria and associated staff. New York botanical Garden’s virtual herbarium. Home page at: http://sweetgum.nybg.org/ih/
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, New York, pp 315–322
Wedin M, Döring H, Gilenstam G (2004) Saprotrophy and lichenization as options for the same fungal species on different substrata: environmental plasticity and fungal lifestyles in the Stictis–Conotrema complex. New Phytol 164:459–465
Wedin M, Döring H, Gilenstam G (2006) Stictis s. Lat. (Ostropales, Ascomycota) in northern Scandinavia, with a key and notes on morphological variation in relation to lifestyle. Mycol Res 110:773–789
Wedin M, Döring H, Könberg K, Gilenstam G (2005) Generic delimitations in the family Stictidaceae (Ostropales, Ascomycota): the Stictis–Conotrema problem. Lichenologist 37:67–75
Zoller S, Scheidegger S, Sperisen C (1999) PCR primers for the amplification of mitochondrial small subunit ribosomal DNA of lichen-forming ascomycetes. Lichenologist 31:511–516
Acknowledgements
The staff at the herbaria UPS and S is thanked for arranging loans of material. Bodil Cronholm from the Molecular Systematics Laboratory at the Swedish Museum of Natural History assisted in the laboratory work to sequence the specimens. Karen Hansen is thanked for letting us use her equipment for photographing. This study was supported by the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (FORMAS) BiodivERsA grant 226-2011-1795, as a part of the Soil Crust International (SCIN) project. Additional funding came from the Swedish Research Council grant VR2016-03589.
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Gerhard Rambold
This article is part of the “Special Issue on ascomycete systematics in honour of Richard P. Korf who died in August 2016”.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Fernández-Brime, S., Olariaga, I., Baral, HO. et al. Cryptodiscus muriformis and Schizoxylon gilenstamii, two new species of Stictidaceae (Ascomycota). Mycol Progress 17, 295–305 (2018). https://doi.org/10.1007/s11557-017-1363-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11557-017-1363-4