Abstract
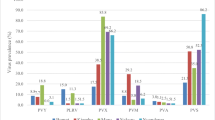
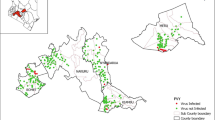
Northwestern Pakistan is the leading seed potato-producing area of the country. Potato cultivars grown commercially in the region were screened against Potato mop-top virus (PMTV) under screenhouse conditions and in naturally infested fields during 2010–2014. The potato cultivars, Asterix, Cardinal, Desiree, Diamant, Kuroda, Paramount, Raja, Rocco, Sarpomira, and Ultimas, were grown in viruliferous soil in a bait test and were all found to be susceptible to PMTV (two isolates from Pakistan-Hazara (PMTV-PkH) and Pakistan-Malakand (PMTV-PkM)) in screenhouse experiments. The virus was detected in all cultivars as tested by double antibody sandwich enzyme-linked immunosorbent assay (DAS-ELISA) and reverse transcription-polymerase chain reaction (RT-PCR) in leaf, root, and tuber samples, and by back inoculation of sap from roots of bait plants on indicator plants. The ELISA values (A405nm) were lower in leaf, root, and tuber samples in cultivar Desiree compared to the other cultivars, whereas in the infectivity assay, the virus was recovered in all cultivars and exhibited characteristic symptoms on diagnostic hosts. All 10 potato cultivars were further tested against PMTV in previously known infested fields at Baffa, Hazara division, and Kalam, Malakand division, in northwestern Pakistan. The cultivars tested at both sites were found susceptible to PMTV. The virus was detected by DAS-ELISA and RT-PCR in randomly selected potato tubers of each cultivar during 3 years at both sites. We conclude that commercially grown potato cultivars in northwestern Pakistan are susceptible (moderately sensitive to sensitive) to PMTV, requiring a comprehensive potato germplasm screening program for the evaluation of sources of resistance against the virus.


Similar content being viewed by others
References
Ahmad I, Iftikhar S, Soomro MH, Merz U (1996) First report of Spongospora subterranea f. sp. subterranea on potato in Pakistan. Plant Dis 80:959
Arif M, Torrance L, Reavy B (1994) Improved efficiency of detection of potato mop-top furovirus in potato tubers and in the roots and leaves of soil-bait plants. Potato Res 37:373–381
Arif M, Torrance L, Reavy B (1995) Acquisition and transmission of potato mop-top furovirus by a culture of Spongospora subterranea f. sp. subterranea derived from single cystosorus. Ann Appl Biol 126:493–503
Arif M, Ali M, Rehman A, Fahim M (2013) Occurrence of potato mop-top virus in Northwest of Pakistan. Eur J Plant Pathol 137:787–796
Arif M, Ali M, Rehman A, Fahim M (2014) Detection of Potato mop-top virus in soils and potato tubers using bait-plant bioassay, ELISA and RT-PCR. J Virol Methods 195:221–227
Barker H, Dale MFB (2006) Resistance to viruses in potato. In: Loebenstein G, Car JP (eds) Natural resistance mechanisms of plants to viruses. Springer, Dordrecht, pp 341–366
Barker H, Reavy B, McGeachy KD, Dawson S (1996) Transformation of Nicotiana benthamiana with potato mop-top virus coat protein gene produces a novel resistance phenotype mediated by the coat protein. Mol Plant-Microbe Interact 11:626–633
Beuch U, Berlin S, Akerblom J, Nicolaisen M, Nielsen SL, Crosslin JM, Hamm PB, Santala J, Valkonen JPT, Kvarnheden A (2015) Diversity and evolution of potato mop-top virus. Arch Virol 160:1345–1351
Budziszewska M, Wieczorek P, Nowaczyk K, Borodynko N, Pospieszny H, Obrepalska-Steplowska A (2010) First report of Potato mop-top virus on potato in Poland. Plant Dis 94:920
Calvert EL, Harrison BD (1966) Potato mop-top, a soil-borne virus. Plant Pathol 15:134–139
Carnegie SF, Saddler GS, Davey T, Peters J, Budge G (2009) Testing potato varieties for susceptibility to potato mop-top virus (PMTV). Final project report: Agriculture and horticulture development board (AHDB)/ R and D department. Potato Research Council Ltd, Kenilworth
Clark MF, Adams AN (1977) Characteristics of the microplate method of enzyme-linked immunosorbent assay for detection of plant viruses. J Gen Virol 34:475–483
Crosslin JM (2011) First report of Potato mop-top virus on potatoes in Washington State. Plant Dis 95:1483
David N, Mallik I, Crosslin JM, Gudmestad NC (2010) First report of Potato mop-top virus in North Dakota. Plant Dis 94:1506
Domfeh O, Bittara FG, Gudmestad NC (2015) Sensitivity of Potato mop-top virus-induced tuber necrosis. Plant Dis 99:788–796
Germundson A, Sandgren M, Barker H, Savenkov EI, Volkonen JPT (2002) Initial infection of roots and leaves reveals different resistance phynotypes associated with coat protein gene-mediated resistance to Potato mop-top virus. J Gen Virol 83:1201–1209
Halterman D, Charkowski A, Verchot J (2012) Potato, viruses, and seed certification in the USA to provide healthy propagated tubers. Pest Technol 6:1–14
Harrison BD, Jones RAC (1970) Host range and properties of potato mop-top virus. Ann Appl Biol 65:393–402
Harrison BD, Jones RAC (1971) Factor affecting the development of spraing in potato tubers infected with potato mop-top virus. Ann Appl Biol 68:281–289
Harrison BD, Reavy B (2002) Potato mop-top virus. In: Descriptions of Plant Viruses, No. 389. Commonwealth mycological institute/association of applied biologists, Kew, Surrey, England
Harrison JG, Searle RJ, Williams NA (1997) Powdery scab disease of potato-a review. Plant Pathol 46:1–25
Hims MJ, Preece TF (1975) Spongospora subterranea f. sp. subterranea. In: Descriptions of Pathogenic Fungi and Bacteria, No. 477. Commonwealth Mycological Institute/ Association of Applied Biologists, Kew, Surrey, England
Iftikhar S, Rattu AR, Asad S, Burney K (2007) Susceptibility of potato cultivars to Spongospora subterranea under field conditions. Pak J Bot 39:1329–1333
Jones RAC (1988) Epidemiology and control of potato mop-top virus. In: Cooper JI, Asher MJC (eds) Developments in applied biology II. Viruses with fungal vectors. Association of Applied Biologists, Wellesbourne, pp 255–270
Jones RAC, Harrison BD (1969) The behaviour of potato mop-top virus in soil, and evidence for its transmission by Spongospora subterranea (Wallr.) Lagerh. Ann Appl Biol 63:1–17
Kallender H, Buck KW, Brunt AA (1990) Association of three RNA molecules with potato mop-top virus. Neth J Plant Pathol 96:47–50
King AMQ, Lefkowitz E, Adams MJ, Carstens EB (eds) (2012) Virus taxonomy-ninth report of the international committee on taxonomy of viruses. Elsevier/Academic Press, London
Kurppa AHJ (1989a) The distribution and incidence of mop-top virus in Finland as determined in 1987 and on the variation of disease symptoms in infected potatoes. Ann Agric Fenn 28:285–295
Kurppa AHJ (1989b) Reaction of potato cultivars to primary and secondary infection by potato mop-top furovirus and strategies for virus detection. EPPO Bull 19:593–598
Lambert DH, Levy L, Mavrodieva VA, Johnson SB, Babcock MJ, Vayda ME (2003) First report of Potato mop-top virus on potato from the United States. Plant Dis 87:872
Latvala-Kilby S, Aura JM, Pupola N, Hannukkala A, Valkonen JPT (2009) Detection of Potato mop-top virus in potato tubers and sprouts: combinations of RNA2 and RNA3 variants and incidence of symptomless infections. Phytopathology 99:519–531
Melander M, Lee M, Sandgren M (2001) Reduction of potato mop-top virus accumulation and incidence in tubers of potato transformed with a modified triple gene block gene of PMTV. Mol Breed 8:197–206
Merz U (2008) Powdery scab of potato-occurrence, life cycle and epidemiology. Am J Potato Res 85:241–246
Merz U, Fallon R (2009) Review: powdery scab of potato-increased knowledge of pathogen biology and disease epidemiology for effective disease management. Potato Res 52:17–37
Nielsen SL, Mølgaard JP (1997) Incidence, appearance and development of potato mop-top furovirus-induced spraing in potato cultivars and the influence on yield, distribution in Denmark and detection of the virus in tubers by ELISA. Potato Res 40:101–110
Palukaitis P (2012) Resistance to viruses of potato and their vectors. Plant Pathol J 28:348–358
Ramesh SV, Raikhy G, Brown CR, Whitworth JL, Pappu HR (2014) Complete genomic characterization of a potato mop-top virus isolate from the United States. Arch Virol. doi:10.1007/s00705-014-2214-0
Reavy B, Arif M, Kashiwazaki S, Webster KD, Barker H (1995) Immunity to potato mop-top virus in Nicotiana benthamiana plants expressing the coat protein gene is effective against fungal inoculation of the virus. Mol Plant-Microbe Interact 8:286–291
Reavy B, Sandgren M, Barker H, Heino P, Oxelfelt P (1997) A coat protein transgene from a Scottish isolate of potato mop-top virus mediates strong resistance against Scandinavian isolates which have similar coat protein genes. Eur J Plant Pathol 103:829–834
Sandgren M (1995) Potato mop-top virus (PMTV) distribution in Sweden, development of symptoms during storage and cultivar trials in field and glasshouse. Potato Res 38:379–389
Sandgren M, Savenkov EI, Valkonen JPT (2001) The readthrough region of Potato mop-top virus (PMTV) coat protein encoding RNA, the second largest RNA of PMTV genome, undergoes structural changes in naturally infected and experimentally inoculated plants. Arch Virol 146:467–477
Sandgren M, Plaisted RL, Watanabe KN, Olsson S, Valkonen JPT (2002) Evaluation of some North and South American potato breeding lines for resistance to Potato mop-top virus in Sweden. Am Potato J 79:205–210
Santala J, Samuilova O, Hannukkala A, Latvala S, Kortemaa H, Beuch U, Kvarnheden A, Persson P, Topp K, Ørstad K, Spetz C, Nielsen SL, Kirk HG, Budziszewska M, Wieczorek P, Obrepalska-Steplowska A, Pospieszny H, Kryszczuk A, Sztangret-Wisniewska J, Yin Z, Chrzanowska M, Zimnoch-Guzowska E, Jackeviciene E, Taluntyte L, Pupola N, Mihailova J, Lielmane I, Jarvekulg L, Kotkas K, Rogozina E, Sozonov A, Tikhonovich I, Horn P, Broer I, Kuusiene S, Staniulis J, Uth JG, Adam G, Valkonen JPT (2010) Detection, distribution and control of Potato mop-top virus, a soil-borne virus, in northern Europe. Ann Appl Biol 157:163–178
Savenkov EI, Sandgren M, Valkonen JPT (1999) Complete sequence of RNA 1 and the presence of tRNA-like structure in all RNAs of Potato mop-top virus, genus Pomovirus. J Gen Virol 80:2779–2784
Solomon-Blackburn RM, Barker H (2001a) Breeding virus resistance potatoes (Solanum tuberosum): a review of traditional and molecular approaches. Heredity 86:17–35
Solomon-Blackburn RM, Barker H (2001b) A review of host major-gene resistance to potato viruses X, Y, A and V in potato: genes genetics and mapped locations. Heredity 86:8–16
Solomon-Blackburn RM, Wastie RL (1988) Management of potato mop-top virus and its vector by breeding and selection. In: Cooper JI, Asher MJC (eds) Developments in applied biology II. Viruses with fungal vectors. Association of Applied Biologists, Wellesbourne, pp 271–279
Tenorio J, Franco Y, Chuquillanqui C, Owens RA, Salazar LF (2006) Reaction of potato varieties to Potato mop-top virus infection in the Andes. Am J Potato Res 83:423–431
Valkonen JPT (2015) Elucidation of virus-host interactions to enhance breeding for control of virus diseases in potato. Breed Sci 65:69–76
Verwoerd TC, Dekker BMM, Hoekema A (1989) A small scale procedure for rapid isolation of plant RNAs. Nucleic Acids Res 17:2362
Whitworth JL, Crosslin JM (2013) Detection of Potato mop-top virus (Furovirus) on potato in southeast Idaho. Plant Dis 97:149
Xu H, DeHaan T–L, Boar SHD (2004) Detection and confirmation of Potato mop-top virus in potatoes produced in the United States and Canada. Plant Dis 88:363–367
Zamyatnin AA, Solovyev AG, Savenkov EI, Germundsson A, Sandgren M, Valkonen JPT, Morozov SY (2004) Transient coexpression of individual genes encoded by the triple gene block (TGB) of Potato mop-top virus reveals requirements for TGBp1 trafficking. Mol Plant-Microbe Interact 17:921–930
Acknowledgments
This work was funded by Higher Education Commission, Islamabad, Pakistan under National Research Program for Universities (NRPU) through Grant No. 20-1182/R and D/08.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arif, M., Ruby, G., Rehman, A. et al. Screening of Potato Cultivars to Potato Mop-Top Virus in Northwestern Pakistan. Potato Res. 59, 295–312 (2016). https://doi.org/10.1007/s11540-016-9324-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11540-016-9324-x