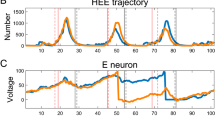
Abstract
The spiking activity of mitral cells (MC) in the olfactory bulb is a key attribute in olfactory sensory information processing to downstream cortical areas. A more detailed understanding of the modulation of MC spike statistics could shed light on mechanistic studies of olfactory bulb circuits and olfactory coding. We study the spike response of a recently developed single-compartment biophysical MC model containing seven known ionic currents and calcium dynamics subject to constant current input with background white noise. We observe rich spiking dynamics even with constant current input, including multimodal peaks in the interspike interval distribution (ISI). Although weak-to-moderate background noise for a fixed current input does not change the firing rate much, the spike dynamics can change dramatically, exhibiting non-monotonic spike variability not commonly observed in standard neuron models. We explain these dynamics with a phenomenological model of the ISI probability density function. Our study clarifies some of the complexities of MC spiking dynamics.





Similar content being viewed by others
Data Availability
This study did not use any experimental data.
References
Ashwin P, Coombes S, Nicks R (2016) Mathematical frameworks for oscillatory network dynamics in neuroscience. J Math Neurosci 6(1):2
Balu R, Larimer P, Strowbridge BW (2004) Phasic stimuli evoke precisely timed spikes in intermittently discharging mitral cells. J Neurophysiol 92(2):743–753
Barreiro A, Gautam S, Shew W, Ly C (2017) A theoretical framework for analyzing coupled neuronal networks: application to the olfactory system. PLoS Comput Biol 13:e1005780. https://doi.org/10.1371/journal.pcbi.1005780
Bhalla US, Bower JM (1993) Exploring parameter space in detailed single neuron models: simulations of the mitral and granule cells of the olfactory bulb. J Neurophysiol 69(6):1948–1965
Chen WR, Shepherd GM (1997) Membrane and synaptic properties of mitral cells in slices of rat olfactory bulb. Brain Res 745(1–2):189–196
Craft M, Barreiro A, Gautam S, Shew W, Ly C (2021) Differences in olfactory bulb mitral cell spiking with ortho- and retronasal stimulation revealed by data-driven models. PLoS Comput Biol 17(9):e1009169. https://doi.org/10.1371/journal.pcbi.1009169
Cury KM, Uchida N (2010) Robust odor coding via inhalation-coupled transient activity in the mammalian olfactory bulb. Neuron 68(3):570–585
Desmaisons D, Vincent JD, Lledo PM (1999) Control of action potential timing by intrinsic subthreshold oscillations in olfactory bulb output neurons. J Neurosci 19(24):10727–10737
DeVille R, Muratov C, Vanden-Eijnden E (2006) Non-meanfield deterministic limits in chemical reaction kinetics. J Chem Phys 124:231102
Doedel EJ (1981) Auto: a program for the automatic bifurcation analysis of autonomous systems. Congr Numer 30(265–284):25–93
Ermentrout B (1996) Type I membranes, phase-resetting curves, and synchrony. Neural Comput 8:979–1001
Ermentrout B (2002) Simulating, analyzing, and animating dynamical systems: a guide to XPPAUT for researchers and students. SIAM
Ermentrout B, Terman D (2010) Mathematical foundations of neuroscience, vol 64. Springer, USA
Friedrich RW, Laurent G (2004) Dynamics of olfactory bulb input and output activity during odor stimulation in zebrafish. J Neurophysiol 91(6):2658–2669
Gammaitoni L, Hänggi P, Jung P, Marchesoni F (1998) Stochastic resonance. Reviews of modern physics 70(1):223
Gardiner C (1985) Handbook of stochastic methods. Springer-Verlag, London
Gerstner W, Kistler W (2002) Spiking Neuron Models. Cambridge University Press, Cambridge
Kramers H (1940) Physica (Utrecht) 1940:284
Li G, Cleland TA (2013) A two-layer biophysical model of cholinergic neuromodulation in olfactory bulb. J Neurosci 33(7):3037–3058
Li G, Cleland TA (2017) A coupled-oscillator model of olfactory bulb gamma oscillations. PLoS Comput Biol 13(11):e1005760
Lindner B (2002) Coherence and stochastic resonance in nonlinear dynamical systems. Ph. D. thesis, Humboldt University of Berlin
Lindner B, Longtin A, Bulsara A (2003) Analytic expressions for rate and cv of a type i neuron driven by white gaussian noise. Neural Comput 15(8):1761–1788
Lundstrom B, Famulare M, Sorensen L, Spain W, Fairhall A (2009) Sensitivity of firing rate to input fluctuations depends on time scale separation between fast and slow variables in single neurons. J Comput Neurosci 27:277–290
Ly C, Barreiro AK, Gautam SH, Shew WL (2021) Odor-evoked increases in olfactory bulb mitral cell spiking variability. iScience 24(9): 102946
Ly C, Doiron B (2017) Noise-enhanced coding in phasic neuron spike trains. PLoS ONE 4:e0176963. https://doi.org/10.1371/journal.pone.0176963
Ly C, Ermentrout B (2011) Analytic approximations of statistical quantities and response of noisy oscillators. Physica D 240:719–731
Ly C, Tranchina D (2009) Spike train statistics and dynamics with synaptic input from any renewal process: a population density approach. Neural Comput 21:360–396. https://doi.org/10.1162/neco.2008.03-08-743
Marella S, Ermentrout B (2008) Class-ii neurons display a higher degree of stochastic synchronization than class-i neurons. Phys Rev E 77:041918
Marella S, Ermentrout B (2010) Amplification of asynchronous inhibition-mediated synchronization by feedback in recurrent networks. PLoS Comput Biol 6(2):e1000679
Migliore M, Hines ML, Shepherd GM (2005) The role of distal dendritic gap junctions in synchronization of mitral cell axonal output. J Comput Neurosci 18(2):151–161
Nesse WH, Del Negro CA, Bressloff PC (2008) Oscillation regularity in noise-driven excitable systems with multi-time-scale adaptation. Phys Rev Lett 101(8):088101
Ostojic S (2011) Interspike interval distributions of spiking neurons driven by fluctuating inputs. J Neurophysiol 106(1):361–373
Patel M, Rangan A (2021) Olfactory encoding within the insect antennal lobe: the emergence and role of higher order temporal correlations in the dynamics of antennal lobe spiking activity. J Theor Biol 522:110700
Plesser HE, Gerstner W (2000) Noise in integrate-and-fire neurons: from stochastic input to escape rates. Neural Comput 12:367–384
Pyzza PB, Newhall KA, Kovačič G, Zhou D, Cai D (2021) Network mechanism for insect olfaction. Cogn Neurodyn 15(1):103–129
Rall W, Shepherd GM, Reese TS, Brightman MW (1966) Dendrodendritic synaptic pathway for inhibition in the olfactory bulb. Exp Neurol 14(1):44–56
Reinker S, Li YX, Kuske R (2006) Noise-induced coherence and network oscillations in a reduced bursting model. Bull Math Biol 68(6):1401–1427
Reinker S, Puil E, Miura RM (2003) Resonances and noise in a stochastic hindmarsh-rose model of thalamic neurons. Bull Math Biol 65(4):641–663
Rinzel J, Ermentrout B (1989) Analysis of neural excitability and oscillations, In Methods in Neuronal Modeling: From Synapses to Networks, ed. Segev, I., 135–169. MIT Press
Risken H (1989) The Fokker-Planck equation: methods of solutions and applications. Springer-Verlag, New York, NY
Rospars JP, Lansky P, Chaput M, Duchamp-Viret P (2008) Competitive and noncompetitive odorant interactions in the early neural coding of odorant mixtures. J Neurosci 28(10):2659–2666
Sacerdote L, Villa A, Zucca C (2006) On the classification of experimental data modeled via a stochastic leaky integrate and fire model through boundary values. Bull Math Biol 68(6):1257–1274
Schwabedal JT, Pikovsky A (2013) Phase description of stochastic oscillations. Phys Rev Lett 110(20):204102
Schwalger T, Schimansky-Geier L (2008) Interspike interval statistics of a leaky integrate-and-fire neuron driven by gaussian noise with large correlation times. Phys Rev E 77(3):031914
Shmuel R, Secundo L, Haddad R (2019) Strong, weak and neuron type dependent lateral inhibition in the olfactory bulb. Sci Rep 9(1):1–11
Thomas PJ, Lindner B (2014) Asymptotic phase for stochastic oscillators. Phys Rev Lett 113(25):254101
Viertel R, Borisyuk A (2019) A computational model of the mammalian external tufted cell. J Theor Biol 462:109–121
Wang XJ (1993) Ionic basis for intrinsic 40 hz neuronal oscillations. NeuroReport 5(3):221–224
Wanner AA, Friedrich RW (2020) Whitening of odor representations by the wiring diagram of the olfactory bulb. Nat Neurosci 23(3):433–442
Wedgwood KC, Lin K, Thul R, Coombes S (2013) Phase-amplitude descriptions of neural oscillator models. J Mathemat Neurosci 3:2
Wilson D, Ermentrout B (2019) Augmented phase reduction of (not so) weakly perturbed coupled oscillators. SIAM Rev 61(2):277–315
Zhou P, Burton S, Urban N, Ermentrout B (2013) Impact of neuronal heterogeneity on correlated colored noise-induced synchronization. Frontiers in Computational Neuroscience 7:113. https://doi.org/10.3389/fncom.2013.00113
Acknowledgements
We thank Woodrow Shew, Shree Hari Gautam, and Andrea Barreiro for many conversations about olfactory bulb cells and circuits.
Funding
This work is funded by an NSF Grant (#IIS—1912338 for both CL and MC); the funding agency had no role in the design of the study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and analysis were performed by Cheng Ly and Michelle Craft. The first draft of the manuscript was written by Cheng Ly and Michelle Craft and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Code Availability
The code to implement the models is freely available at https://github.com/michellecraft64/MCuncoup.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A Viability of Alternative Approaches
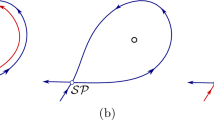
A common approach to reduce the number of state variables in describing (regular) spiking is to apply a phase reduction, which seems appealing because the results hold for relatively weak noise forcing where firing rates do not vary much. This motivated us to analyze the bifurcation between quiescence and spiking (using XPP-AUTO (Ermentrout 2002; Doedel 1981)), in the noiseless case (Fig. 6a). We see that the stable rest state loses stability via a saddle-node on invariant circle (SNIC) bifurcation as the applied current increases. However, we do not find actual periodic solutions for relevant firing rates with which to calculate commonly used entities for analysis like the phase-resetting curve (PRC). Curiously, the bifurcation diagram shows stable period solutions (green dots) that are interlaced with unstable periodic solutions. Figure 6b shows that even with \(I_{\text {app}}=144\,\mu \text {A/cm}^2\) where the diagram (Fig. 6a) suggests there is a well-behaved periodic solution, the voltage (and other variables) are not strictly periodic. Note that with \(I=144\), the firing rates are large (91.13 Hz) and not physiologically relevant. Also, for \(I=144\) although the PRC is numerically calculable (Fig. 6c), the rather large negative region does not resemble canonical PRCs associated with a SNIC (Rinzel and Ermentrout 1989; Ermentrout 1996).
Dynamics of noiseless MC model: phase reduction assumptions violated. a Bifurcation diagram of voltage varying I shows SNIC at onset of spiking. b The voltage traces in Fig.2a are not strictly periodic, even for an ideal well-behaved I value. For unrealistically high firing rates (\(I=144\,\mu \text {A/cm}^2\), firing rate: 91.13 Hz), we see the system is not periodic despite the bifurcation diagram suggesting it should be. The infinitesimal phase-resetting curve can be numerically calculated with XPP (Ermentrout 2002); notice that the negative region is rather large, not resembling canonical PRCs synonymous with a SNIC (Rinzel and Ermentrout 1989; Ermentrout 1996). This all suggests phase reduction descriptions would likely be inadequate to capture the observed phenomena in the regimes we are interested in (i.e., smaller I with physiological firing rates) (Color figure online)
Note that in a standard phase-reduced scalar model (Ermentrout and Terman 2010):
the ISI density can be approximated (assuming weak noise) via (Ly and Ermentrout 2011):
Since the formula closely resembles a normal distribution, the \(\sigma _{ISI}\) generally increases as \(\sigma \nearrow \). Even though there is multiplicative noise: \(\sigma \Delta (\Theta )\), these models would not help describe observations in Fig. 3.
Another common approach to model analysis with weak noise is use a potential well: \(\frac{dV}{dt}=-U'(V)+\sigma \xi (t)\), where U is the potential function, either principally derived from the system (simple) or ad hoc (high-dimensional). For example, for the leaky-integrate-and-fire model, \(U(V) \propto \frac{1}{2}(V-V_L)^2\) and V has a stable fixed point at the global minimum \(V=V_L\). This approach was pioneered in physics by Kramers (1940) and has been applied to several neural models where ‘exiting’ from the potential well from crossing a threshold is spiking. The rate of spiking is often \(\propto e^{-U/\sigma ^2}\), which is not directly related to spiking variability. The signal-to-noise ratio (SNR) in these systems, and in other applications of stochastic resonance, often have a maximal SNR value for an intermediate level of noise (Gammaitoni et al. 1998). However, this dynamic is associated with a minimum variability value in the denominator (ignoring the dynamics of the signal in the numerator) of SNR, rather than a maximal spiking variability for intermediate input noise level, as we have observed in the MC model.
Whether the potential well or ‘Arrhenius escape’ approach by Nesse et al. (2008) for a low-dimensional Fitzhugh–Nagumo model (with 1 activity variable x and a set of identical adaptation variables H endowed with multiple timescales that depend on x) could be successfully applied to our MC model is an open question. Nesse et al. (2008) exploited a separation of timescales, the slow variable was fixed and the mean first passage time (or escape) T could be calculated (in the fast variable) and set to the inverse of the mean firing rate: \(\lambda (H(t))=1/T(H(t))\). The ISI density is approximated with:
we see how the slow variation in H affects \(\rho (t)\). This framework successfully described the non-monotonic spiking dynamics (in the CV at least) in their model. As previously mentioned in the Discussion, an analogous approach would require identifying all of the effective timescales in our 13 variable model and having a significant separation of timescales when the neuron is excitable. Even if the slow variables are frozen, one would still have to calculate the mean first passage time with the remaining fast variables, which is generally not feasible unless the resulting dimension is small. Solving for the mean first passage time requires solving an ODE system derived from the backward Fokker–Planck equation, a PDE with the number of dimensions equal to the number of state variables (Gardiner 1985; Risken 1989). The viability and the accuracy of this approach for capturing our results is an open question but beyond the scope of this current study.
Appendix B Other Spike Statistics
The autocorrelation function (Eq (B3)) and power spectrum (Eq (B4)) of the MC model. Three values of input current: \(I=120\,\mu \text {A}/\text {cm}^2\) in a–c, \(I=130\,\mu \text {A}/\text {cm}^2\) in d–f, \(I=140\,\mu \text {A}/\text {cm}^2\) in g–i, with each panel showing the effects of increasing input noise \({\tilde{\sigma }}\). The effects of input noise are nonlinear and highly dependent on I (Color figure online)
We have focused on the ISI distribution, but there are other commonly used entities to characterize neural spike trains. For instance, the autocorrelation function (ACF) and power spectrum (P), defined below, are commonly used and can be unrelated to the ISI, in particular, when the system does not reset after a spike. Letting R(t) denote the spike train consisting of 0’s and 1’s, the (normalized) autocorrelation function is:
and the power spectrum:
Figure 7 shows these entities for the biophysical MC model with various applied current and input noise values.
With \(I=120\,\mu \text {A}/\text {cm}^2\), \(ACF(\tau )\) is relatively flat for these input noise values, while \(P(\omega )\) changes from having peaks at regularly spaced intervals with no noise (black) to being relatively flat with \({\tilde{\sigma }}>0\). With \(I=130\,\mu \text {A}/\text {cm}^2\), \(ACF(\tau )\) has local maxima at irregularly spaced \(\tau \) with no noise (black) that flatten out as input noise increases; the \(P(\omega )\) is similar to \(I=120,\mu \text {A}/\text {cm}^2\) but the curves have smaller values compared to \(I=120\). With \(I=140\,\mu \text {A}/\text {cm}^2\), \(ACF(\tau )\) indicates relatively regular spiking, although increased input noise shifts and broadens the peaks (same for \(P(\omega )\)). The flatter ACF with \(I=120\) compared to larger I indicates that the spiking has less temporal regularity, which is not surprising. For a given value of I (i.e., a row in Fig. 7), increasing input noise flattens the \(ACF(\tau )\) and shifts and/or diminishes peaks (local max), and for \(P(\omega )\) input noise can broaden/shift/diminish peaks. Overall, the effects of input noise are nonlinear and highly dependent on I.
The ACF and P were plotted using built-in functions in MATLAB, and there appears to be slight numerical round-off errors in the ACF, e.g., between the peaks in Fig. 7g, \(ACF\approx 0\), and likely in the P. Nevertheless, these plots given insight to some dynamics of various spike trains.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Craft, M., Ly, C. The Effects of Background Noise on a Biophysical Model of Olfactory Bulb Mitral Cells. Bull Math Biol 84, 107 (2022). https://doi.org/10.1007/s11538-022-01066-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01066-8