Abstract
The re-emergence of syphilis has become a global public health issue, and more persons are getting infected, especially in developing countries. This has also led to an increase in the incidence of human immunodeficiency virus (HIV) infections as some studies have shown in the recent decade. This paper investigates the synergistic interaction between HIV and syphilis using a mathematical model that assesses the impact of syphilis treatment on the dynamics of syphilis and HIV co-infection in a human population where HIV treatment is not readily available or accessible to HIV-infected individuals. In the absence of HIV, the syphilis-only model undergoes the phenomenon of backward bifurcation when the associated reproduction number (\({\mathcal {R}}_{T}\)) is less than unity, due to susceptibility to syphilis reinfection after recovery from a previous infection. The complete syphilis–HIV co-infection model also undergoes the phenomenon of backward bifurcation when the associated effective reproduction number (\({\mathcal {R}}_{C}\)) is less than unity for the same reason as the syphilis-only model. When susceptibility to syphilis reinfection after treatment is insignificant, the disease-free equilibrium of the syphilis-only model is shown to be globally asymptotically stable whenever the associated reproduction number (\({\mathcal {R}}_{T}\)) is less than unity. Sensitivity and uncertainty analysis show that the top three parameters that drive the syphilis infection (with respect to the associated response function, \({\mathcal {R}}_{T}\)) are the contact rate (\(\beta _S\)), modification parameter that accounts for the increased infectiousness of syphilis-infected individuals in the secondary stage of the infection (\(\theta _1\)) and treatment rate for syphilis-only infected individuals in the primary stage of the infection (\(r_1\)). The co-infection model was numerically simulated to investigate the impact of various treatment strategies for primary and secondary syphilis, in both singly and dually infected individuals, on the dynamics of the co-infection of syphilis and HIV. It is observed that if concerted effort is exerted in the treatment of primary and secondary syphilis (in both singly and dually infected individuals), especially with high treatment rates for primary syphilis, this will result in a reduction in the incidence of HIV (and its co-infection with syphilis) in the population.







Similar content being viewed by others
References
Aadland D, Finnoff D, Huang K (2013) Syphilis cycles. B E J Econ Finance 13(1):297–348
Abdallah SW, Estomih SM, Oluwole DM (2012) Mathematical modelling of HIV/AIDS dynamics with treatment and vertical transmission. Appl Math 2(3):77–89
Allen SF, Abbasi AJ (2003) Syphilis and human immunodeficiency virus co-infection. J Natl Med Assoc 95:363–382
Anderson RM, May RM (1987) Transmission dynamics of HIV infection. Nature 326:137–142
Blower SM, Dowlatabadi H (1994) Sensitivity and uncertainty analysis of complex models of disease transmission. Int Stat Rev 62(2):229–243
Castillo-Chavez C, Song B (2004) Dynamical models of tuberculosis and their applications. Math Biosci Eng 2:361–404
Centers for Disease Control and Prevention (CDC) (October, 2016) Sexually transmitted disease (STDs) syphilis-CDC fact sheet. www.cdc.gov. Accessed in October 2016
Chesson HW, Pinkerton SD, Irwin KL, Rein D, Kassler WJ (1999) HIV cases attributable to syphilis in the USA: estimates from a simplified transmission model. AIDS 13:1387–1396
Chesson HW, Pinkerton SD, Voigt R (2003) Hiv infection and associated costs attributable to syphilis co-infection among African Americans. Am J Public Health 93:943–948
Ciesielski C, Ginsberg MB, Robertson BJ, Luo CC, DeMaria A Jr, Ridzon R, Gallagher K (1997) Simultaneous transmission of human immuodeficiency virus and hepatitis C virus from a needle-stick. N Engl J Med 336(13):919–22
Clark GE, Danbolt N (1964) The Oslo study of the national cause of untreated syphilis: an epidemiological investigation based on the study of the Boeck-Bruusgaard material. Med Clin North Am 48:612–623
Communicable Disease Report (CDR) (2003) Recent developments in syphilis epidemiology. Communicable Disease Reports, 13
Czelusta A, Yen-Moore A, Vander Straten M et al (2000) An overview of sexually transmitted diseases Part 3. Sexually transmitted diseases in HIV-infected patients. J Am Acad Dermatol 43:450–451
Dickerson MC, Johnston J, Delea TE (1996) The causal role of genital ulcer disease as a risk factor for the transmission of HIV. Sex Transm Dis 23:433–440
Diekmann O, Heesterbeek JAP (2000) Mathematical epidemiology of infectious diseases: model building, analysis and interpretation. Wiley, New York
Domegan L, Cronin M, Thornton L, Creamer E, O’Lorcain P, Hopkins S (2002) Enhanced surveillance of syphilis. Epi-Insight 3:2–3
Elbasha EH (2013) Model for hepatitis C virus transmission. Math Biosci 10(4):1045–1065
Global Health Magazine (December 15, 2011) Fighting syphilis and HIV in women and children: lessons from Uganda and Zambia. www.pedaids.org/news/P220
Grassly C, Fraser C, Garnett GP (2005) Host immunity and synchronised epidemics of syphilis across the United States. Nature 433:417–421
Greenbalt RM, Lukehart SA, Plummer FA et al (1988) Genital ulceration as a risk factor for HIV infection. AIDS 2:47–50
Gumel AB (2012) Causes of backward bifurcation in some epidemiological models. J Math Anal Appl 395:355–365
Gwanzura L, Latif A et al (1999) Syphilis serology and HIV infection in Harare Zimbabwe. Sex Transm Infect 75:426–430
Hadeler K, van den Driessche P (1997) Backward bifurcation in epidemic control. Math Biosci 146:15–35
Hethcote HW (2000) The mathematics of infectious diseases. Soc Ind Appl Math Rev 42:599–653
Iboi E, Okuonghae D (2016) Population dynamics of a mathematical model for syphilis. Appl Math Model 40:3573–3590
La Salle J, Lefschetz S (1976) The stability of dynamical systems. SIAM, Philadelphia
Lakshmikantham S, Leela S, Martynyuk AA (1989) Stability analysis of non-linear systems. Marcel Dekker Inc., New York
Liming C, Xuezhi L, Mini G, Baozhu G (2008) Stability analysis of HIV/AIDS epidemic model with treatment. J Comput Appl Math 229:313–323
Lynn WA, Lightman S (2004) Syphilis and HIV: a dangerous combination. Lancet Infect Dis 4:456–466
Milner F, Zhoa R (2010) A new mathematical model of syphilis. Math Model Nat Phenom 5(6):96–108
Morbidity and Mortality Weekly Report (MMWR) (1998) HIV prevention through early detection and treatment of other sexually transmitted diseases-United States Recommendations of the Advisory Committee for HIV and STD prevention. MMWR Recomm Rep 47(RR-12):P1–24
Naresh R, Tripathi A, Omar S (2006) Modelling the spread of AIDS epidemic with vertical transmission. Appl Math Comput 178:262–272
Naresh R, Agraj T, Dileep S (2008) Modelling and analysis of the spread of AIDS epidemic with immigration of HIV infectives. Math Comput Modell 49:880–892
Okuonghae D, Omosigho SE (2011) Analysis of a mathematical model for tuberculosis: what could be done to increase case detection. J Theor Biol 269:31–45
Okuonghae D, Ikhimwin BO (2016) Dynamics of a mathematical model for tuberculosis with variability in susceptibility and disease progressions due to difference in awareness level. Front Microbiol 6:1530
Pourbohloul B, Rekart ML, Brunham RC (2003) Impact of mass treatment on syphilis transmission: a mathematical modelling approach. Sex Transm Dis 30:297–305
Rottingen JA, Cameron DW, Garnett GP (2001) A systematic review of the epidemiological interactions between classic sexually transmitted diseases and HIV. Sex Transm Dis 28:581–597
Saad-Roy CM, Shuai Z, van den Driessche P (2016) A mathematical model of syphilis transmission in an MSM population. Math Biosci 277:59–70
Sanchez MA, Blower SM (1997) Uncertainty and sensitivity analysis of the basic reproductive rate. Am J Epidemiol 145(12):1127–1137
Sharomi O, Podder CN, Gumel AB, Song B (2008) Mathematical modelling of the transmission dynamics of HIV/TB co-infection in the presence of treatment. Math Biosci Eng 5(1):145–174
Stoddart CA, Reyes RA (2006) Models of HIV-1 disease: a review of current status. Drug Discov Today Dis Models 3(1):113–119
USAIDS (2013) Access to antiretroviral therapy in Africa: Status Report on Progress Toward 2015 Target. www.unaids.org/en/resources/documents/2013/2015219
van den Driessche P, Watmough J (2002) Reproduction number and sub-threshold endemic equilibria for computational models of disease transmission. Math Biosci 180:29–48
World Health Organisation (WHO) (2012) Global incidence and prevalence of selected curable sexually transmitted infections. www.who.int/reproduvtivehealth/publications/rtis/stisestimates/en/
World Health Organisation (WHO) (January, 2016) Global Health Observatory Data. www.who.int/gho/hiv/en, 2014. Accessed in January 2016
World Health Organisation (WHO) (January, 2016) Eliminating congenital syphilis. www.who.int/reproductive-health/stis/syphilis.html. Accessed in January 2016
Acknowledgements
The authors will like to thank the anonymous reviewers for their invaluable and constructive comments.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Derivation of Model Equations
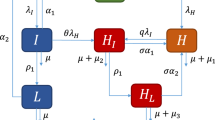
The susceptible population S(t) is generated by the recruitment of individuals (assumed susceptible) into the population at a rate \(\Lambda \). The population of individuals in the S(t) compartment is reduced due to HIV and syphilis infections at the rates \(\lambda _H\), \(\lambda _S\), \(\lambda _{SH}\) and \(\lambda _{HS}\). The population is further reduced by natural death (at a rate \(\mu \); natural death occurs in all epidemiological compartments at this rate). So that
The population of individuals exposed to syphilis is increased by those who acquire syphilis infection at the rates \(\lambda _S\) and \(\lambda _{SH}\) and those reinfected with syphilis after treatment, at the rates \(\alpha _1\lambda _S\) and \(\alpha _2\lambda _{SH}\), where \(\alpha _1\) and \(\alpha _2\) are modification parameters which account for the susceptibility of singly infected individuals with syphilis who recovered from syphilis. This population is reduced by progression to primary syphilis, at the rate \(\gamma _1\), those who get infected with HIV at the rates \(\lambda _H\) and \(\lambda _{HS}\) and then by natural death. Hence
The population of infected individuals with primary syphilis is increased by those who progressed from the exposed stage and reduced by those who get infected with HIV at the rates \(\phi _1\lambda _H\) and \(\phi _2\lambda _{HS}\) (where \(\phi _1\) and \(\phi _2\) are modification parameters which account for their increased susceptibility to HIV infection). This population is further reduced by those who recover at the rate \(r_1\), those who progress to the secondary stage of syphilis, at the rate \(\gamma _2\), and by natural death. So that we have
The population of infected individuals with secondary syphilis is increased by those who progressed from the primary stage and reduced by those who get infected with HIV at the rates \(\phi _3\lambda _H\) and \(\phi _4\lambda _{HS}\) (where \(\phi _3\) and \(\phi _4\) are modification parameters which account for their increased susceptibility to HIV infection). This population is further reduced by those who recover at the rate \(r_2\), those who progress to the early latent stage of syphilis, at the rate \(\gamma _3\), and by natural death. Hence
The population of infected individuals with early latent syphilis is increased by those who progressed from the secondary stage and reduced by those who get infected with HIV at the rates \(\phi _5\lambda _H\) and \(\phi _6\lambda _{HS}\) (where \(\phi _5\) and \(\phi _6\) are modification parameters which account for their increased susceptibility to HIV infection). This population is further reduced by those who recover at the rate \(r_3\), those who progress to the late latent stage of syphilis, at the rate \(\gamma _4\), and finally by natural death. Hence
The population of infected individuals with late latent syphilis is increased by those who progressed from the early latent stage and reduced by those who get infected with HIV at the rates \(\phi _7\lambda _H\) and \(\phi _8\lambda _{HS}\) (where \(\phi _7\) and \(\phi _8\) are modification parameters which account for their increased susceptibility to HIV infection). This population is further reduced by those who recover at the rate \(r_4\), those who progress to the tertiary stage of syphilis, at the rate \(\gamma _5\), and by natural death. Hence
The population of infected individuals with tertiary syphilis is increased by those who progress from the late latent stage and diminished by those who get infected with HIV at the rates \(\phi _9\lambda _H\) and \(\phi _{10}\lambda _{HS}\) (where \(\phi _9\) and \(\phi _{10}\) are modification parameters which account for their increased susceptibility to HIV infection). This population is further reduced by those who recover, at the rate \(r_5\), and natural mortality. We therefore have that
The population of those recovered from syphilis is increased by all singly infected individuals who recover from syphilis at the rates \(r_1\), \(r_2\), \(r_3\), \(r_4\) and \(r_5\) and reduced by the those who get reinfected with syphilis, those who get infected with HIV, at the rates \(\lambda _H\) and \(\lambda _{HS}\), and also by natural death. We then have that
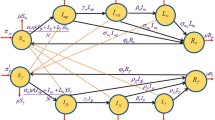
The population of individuals infected with HIV only is increased by HIV infection of susceptible individuals and individuals who recovered from a previous syphilis infection at the rates \(\lambda _H\) and \(\lambda _{HS}\). The population is further increased by HIV-infected individuals treated for syphilis at the primary, secondary, early latent, late latent and tertiary stages, at the rates \(r_{S1}\), \(r_{S2}\), \(r_{S3}\), \(r_{S4}\) and \(r_{S5}\), respectively. The population is however diminished by those who get infected with syphilis at the rates \(\psi _1\lambda _S\) and \(\psi _2\lambda _{SH}\) (where \(\psi _1\) and \(\psi _2\) are modification parameters which account for the increased susceptibility of HIV-infected individuals to syphilis infection), as well as through natural mortality and an additional disease-induced death, at the rate \(d_{H1}\). Hence
The population of HIV-infected individuals who are exposed to syphilis is increased by individuals with HIV who get infected with syphilis and those exposed to syphilis who get infected with HIV at the rates \(\lambda _H\) and \(\lambda _{HS}\) but diminished by the progression of these individuals to the primary stage of syphilis infection, at the rate \(\gamma _{S1}\), by natural mortality and disease-induced mortality, at the rates \(\mu \) and \(d_{H2}\), respectively. So we have
The population of HIV-infected individuals in the primary stage of syphilis is increased by those with primary syphilis who get infected with HIV at the rates \(\phi _1\lambda _H\) and \(\phi _2\lambda _{HS}\), and by individuals who progressed from the exposed stage of syphilis infection, at the rate \(\gamma _{S1}\). This population is decreased by those who progress to secondary stage of syphilis, at the rate \(\gamma _{S2}\), by those who are treated for syphilis, at the rate \(r_{S1}\) and by disease-induced mortality, at the rate \(d_{H3}\), as well as natural mortality. Hence
The population of HIV-infected individuals in the secondary stage of syphilis is increased by those with secondary syphilis who get infected with HIV, at the rates \(\phi _3\lambda _H\) and \(\phi _4\lambda _{HS}\), and by individuals who progressed from the primary stage of syphilis infection, at the rate \(\gamma _{S2}\). This population is decreased by those who progress to early latent stage of syphilis, at the rate \(\gamma _{S3}\), by those who are treated for syphilis, at the rate \(r_{S2}\), and by disease-induced mortality, at the rate \(d_{H4}\), as well as natural mortality. Hence
The population of HIV-infected individuals in the early latent stage of syphilis is increased by those with early latent syphilis who get infected with HIV, at the rates \(\phi _5\lambda _H\) and \(\phi _6\lambda _{HS}\), and by individuals who progressed from the secondary stage of syphilis infection at the rate \(\gamma _{S3}\). This population is decreased by those who progress to late latent stage of syphilis, at the rate \(\gamma _{S4}\), by those who are treated for syphilis, at the rate \(r_{S3}\), and by disease-induced mortality, at the rate \(d_{H5}\), as well as natural mortality. Hence
The population of HIV-infected individuals in the late latent stage of syphilis is increased by those with late latent syphilis who get infected with HIV, at the rates \(\phi _7\lambda _H\) and \(\phi _8\lambda _{HS}\), and by individuals who progressed from the early latent stage of syphilis infection, at the rate \(\gamma _{S4}\). This population is decreased by those who progress to the tertiary stage of syphilis, at the rate \(\gamma _{S5}\), by those who are treated for syphilis, at the rate \(r_{S4}\), and by disease-induced mortality, at the rate \(d_{H6}\), as well as natural mortality. Hence
The population of HIV-infected individuals in the tertiary stage of syphilis is increased by those with tertiary syphilis who get infected with HIV, at the rates \(\phi _9\lambda _H\) and \(\phi _{10}\lambda _{HS}\), and by individuals who progressed from the late latent stage of syphilis infection, at the rate \(\gamma _{S5}\). This population is decreased by those who are treated for syphilis, at the rate \(r_{S5}\), and by disease-induced mortality, at the rate \(d_{H7}\), as well as natural mortality. Hence
Appendix B: Positivity and Boundedness of Solutions
The basic qualitative properties of model (5) are given below. We claim the following.
Theorem 7.1
System (5) preserves positivity of solutions. That is, solutions with positive initial conditions remain positive for all time t.
Proof
Let
From the first equation of model (5), it follows that
when solved leads to
Using the same approach, we can equally show that all other state variable of model (5) will remain positive for all time \(t>0\). \(\square \)
We now prove the positive invariance of the region, \({\mathcal {D}}\).
Lemma 7.1
Consider the biologically feasible region
The closed set \({\mathcal {D}}\) is positively invariant and a global attractor of all positive solution of model (5).
Proof
Adding the equations of model (5) gives
Since the right-hand side of the above equality is bounded by \(\Lambda -\mu N\), a standard comparison theorem (Lakshmikantham et al. 1989) can be used to show that
In particular, \(N(t)\le \frac{\Lambda }{\mu }\) if \(N(0)\le \frac{\Lambda }{\mu }\) for all \(t>0\). Thus, \({\mathcal {D}}\) is a positively invariant set under the flow described by the model. The solutions with initial condition in \({\mathcal {D}}\) remain in \({\mathcal {D}}\) with respect to the model. Hence, the system is mathematically and epidemiologically well posed in \({\mathcal {D}}\) (Hethcote 2000). \(\square \)
Appendix C: Proof of Theorem 3.4
Proof
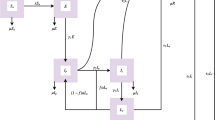
For system (13), let \(S=x_1\), \(E_S=x_2\), \(I_P=x_3\), \(I_S=x_4\), \(L_{S1}=x_5\), \(L_{S2}=x_6\), \(I_T=x_7\) and \(R_S=x_8\). So, the system of equations can be written as
Consider the case with \(\beta _S = \beta _S^{*}\) a bifurcation parameter. Solving for \(\beta _S = \beta _S^{*}\) from \({\mathcal {R}}_T = 1\) gives
The Jacobian of the transformed system evaluated at the DFE with \(\beta _S = \beta _S^{*}\) is given by
We obtain the right eigenvectors associated with the zero eigenvalue which is given as \({\mathbf {w}} = (\omega _1, \omega _2,...,\omega _{8})^T\), where
The components of the left eigenvector of \(J(\xi _{S0}) |_{\beta _S = \beta _S^*}\), \({\mathbf {v}} = (\nu _1, \nu _2,...,\nu _{8})\), satisfying \(\mathbf {v.w}=1\), are
It follows from Theorem 4.1 in Castillo-Chavez and Song (2004), if we compute the associated nonzero partial derivatives of F(x) (evaluated at the DFE \(\xi _{S0}\)), that the associated bifurcation coefficients a and b, defined by
are computed to be
and
with \(\nu _2\), \(\omega _2\), \(\omega _3\), \(\omega _4\), \(\omega _5\), \(\omega _6\), \(\omega _7\) and \(\omega _8\) positive.
Since the bifurcation coefficient \(b>0\), it follows from Theorem 4.1 in Castillo-Chavez and Song (2004) that system (13) will undergo a backward bifurcation if the backward bifurcation coefficient \(a>0\). This is possible if
Clearly, if \(\alpha _1=0\), then \(a<0\) and model (13) will not exhibit the backward bifurcation phenomenon at \({\mathcal {R}}_T=1\). \(\square \)
Appendix D: Proof of Theorem 3.5
Proof
Consider the following linear Lyapunov function
with Lyapunov derivative
It follows from (29), noting that \(S(t)\le N(t)\) and \(N(t)\le \frac{\Lambda }{\mu }\) in \({\mathcal {D}}_2\) for all \(t>0\), that
Hence, \(\dot{{\mathcal {V}}_2}\le 0\) if \({\mathcal {R}}_{T} \le 1\) with \(\dot{{\mathcal {V}}_2}= 0\) if and only if \(E_S=I_P=I_S=L_{S1}=L_{S2}=I_T=R_S=0\). Therefore, \({\mathcal {V}}_2\) is a Lyapunov function in \({\mathcal {D}}_2\), and it follows from LaSalles invariance principle Salle and Lefschetz (1976) that every solution to the equations in (13) with initial conditions in \({\mathcal {D}}_2\) converges to \(\xi _{S0}\) as \(t\rightarrow \infty \). That is,
Substituting \(E_S=I_P=I_S=L_{S1}=L_{S2}=I_T=R_S=0\) into the first equation in model (13) with \(\alpha _1=0\), gives \(S(t)\rightarrow \frac{\Lambda }{\mu }\) as \(t\rightarrow \infty \). Thus, \((S, E_S, I_P, I_S, L_{S1}, L_{S2}, I_T, R_S)\rightarrow (\frac{\Lambda }{\mu }, 0, 0, 0, 0, 0, 0, 0)\) as \(t\rightarrow \infty \) for \({\mathcal {R}}_{T}\le 1\), so the DFE, \(\xi _{S0}\), of model (13) with \(\alpha _1=0\), is globally asymptotically stable in \({\mathcal {D}}_2\) if \({\mathcal {R}}_{T}\le 1\). \(\square \)
Appendix E: Proof of Theorem 3.6
Proof
Consider model (13) (with (22) and \(\alpha _1=0\)) and \({\mathcal {R}}_{T}>1\), so that the associated unique endemic equilibrium exists. Also, consider the nonlinear Lyapunov function of the Goh–Volterra type
with Lyapunov derivative,
It can be shown from model (13) with \(\alpha _1=0\) that, at steady state,
Substituting the time derivatives of \(E_S\), \(I_P\) and \(I_S\), in (13) (with \(\alpha _1=0\)) as well as the relations in (27), into (26), we have that (after several algebraic calculations)
Finally, since the arithmetic mean is greater than the geometric mean, the following inequalities hold.
Thus we have that \(\dot{{\mathcal {F}}_2}\le 0\) for \({\mathcal {R}}_{T}>1\). Since the relevant variables in the equations for \(L_{S1}\), \(L_{S2}\), \(I_T\) and \(R_S\) are at endemic steady state, it follows that these can be substituted into the equations for \(L_{S1}\), \(L_{S2}\), \(I_T\) and \(R_S\) so that
\((L_{S1}(t), L_{S2}(t), I_T(t), R_S(t))\rightarrow (L_{S1}^{**}, L_{S2}^{**}, I_T^{**}, R_S^{**})\) as \(t\rightarrow \infty \)
Hence, \({\mathcal {F}}_2\) is a Lyapunov function in \({\mathcal {D}}_{3}|{\mathcal {D}}_{4}\) \(\square \)
Appendix F: Proof of Theorem 4.1
Proof
For system (5), let \(S=x_1\), \(E_S=x_2\), \(I_P=x_3\), \(I_S=x_4\), \(L_{S1}=x_5\), \(L_{S2}=x_6\), \(I_T=x_7\), \(R_S=x_8\), \(H=x_9\), \(E_H=x_{10}\), \(H_P=x_{11}\), \(H_S=x_{12}\), \(H_{S1}=x_{13}\), \(H_{S2}=x_{14}\) and \(H_T=x_{15}\). So, the system of equations can be written as
where
The Jacobian of the transformed system evaluated at the DFE with is given by
where
Without loss of generality, consider the case when \({\mathcal {R}}_{T}>{\mathcal {R}}_{H}\) and \({\mathcal {R}}_{C}=1\), so that \({\mathcal {R}}_{T}=1\). Furthermore, let \(\beta _S=\beta _S^*\) be a bifurcation parameter. Solving for \(\beta _S\) from \({\mathcal {R}}_{T}=1\) gives
We obtain the right eigenvectors associated with the zero eigenvalue which is given as \({\mathbf {w}} = (\omega _1, \omega _2,...,\omega _{15})^T\), where
The components of the left eigenvector of \(J(\xi _0) |_{\beta _S = \beta _S^*}\), \({\mathbf {v}} = (\nu _1, \nu _2,...,\nu _{15})\), satisfying \(\mathbf {v.w}=1\), are
It follows from Theorem 4.1 in Castillo-Chavez and Song (2004), if we compute the associated nonzero partial derivatives of F(x) (evaluated at the DFE \(\xi _{S0}\)), that the associated bifurcation coefficients a and b, defined by
are computed to be
and
with \(\nu _2\), \(\omega _2\), \(\omega _3\), \(\omega _4\), \(\omega _5\), \(\omega _6\), \(\omega _7\) and \(\omega _8\) positive.
Since the bifurcation coefficient \(b>0\), it follows from Theorem 4.1 in Castillo-Chavez and Song (2004) that system (5) will undergo a backward bifurcation if the backward bifurcation coefficient \(a>0\). This is possible if
Clearly, if \(\alpha _1=0\), then \(a<0\) and model (5) will not exhibit the backward bifurcation phenomenon at \({\mathcal {R}}_C=1\). \(\square \)
Appendix G: Discussion on Some Numerical Simulations
Figure 8 shows that, with increasing treatment rate for primary and secondary syphilis for both singly and dually infected individuals, there is a decrease in the cumulative incidence of HIV amongst individuals exposed to syphilis when the HIV infection is caused by dually infected individuals. This conclusion is also reached when we observe the impact of increasing treatment rates for primary and secondary syphilis (in both singly and dually infected individuals) on the cumulative incidence of HIV amongst individuals in the primary stage of syphilis infection (Fig. 9), secondary stage of syphilis infection (Fig. 10), early latent stage of syphilis infection (Fig. 11), late latent stage of syphilis infection (Fig. 12) and tertiary stage of syphilis infection (Fig. 13). However, when our interest is on investigating the impact of treatment of primary (and secondary) syphilis, for both singly and dually infected individuals, on the cumulative incidence of HIV amongst the aforementioned subpopulations, when the HIV infections are caused by HIV-only infected individuals, we observe that the relatively low treatment rates for primary and secondary syphilis (\(r_1=r_2=r_{s1}=r_{s2}=5\)) were losing its effectiveness on HIV control in the target subpopulations in the long run (see Figs. 8a, 9a, 10a, 11a, 12a, 13a). However, with higher treatment rates for primary and secondary syphilis (in both singly and dually infected individuals), i.e. \(r_1=r_2=r_{S1}=r_{S2}=10\), \(r_1=r_2=r_{S1}=r_{S2}=12\) and \(r_1=r_2=r_{S1}=r_{S2}=15\), we observe a reduction in the cumulative incidence of HIV amongst the subpopulations, just as we observed when the HIV infections were caused by dually infected individuals (as we can see in Figs. 8b, 9b, 10b, 11b, 12b, 13b).
Plots of cumulative incidence of HIV amongst individuals exposed to syphilis, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3. b \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Plots of cumulative incidence of HIV amongst infected individuals in the primary stage of syphilis, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3. b \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Plots of cumulative incidence of HIV amongst infected individuals in the secondary stage of syphilis infection, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3. b \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Plots of cumulative incidence of HIV amongst infected in the early latent stage of syphilis infection, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3. b Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Plots of cumulative incidence of HIV amongst infected in the late latent stage of syphilis infection, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). b Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Plots of cumulative incidence of HIV amongst infected in the tertiary stage of syphilis infection, due to infections caused by singly and dually infected individuals. a Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=0\) (\(\mathcal {R_T}=5.6756\)), \(r_1=r_2=r_{S1}=r_{S2}=5\) (\(\mathcal {R_T}=1.4446\)) and \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3. b Here, we apply three different strategies, namely: \(r_1=r_2=r_{S1}=r_{S2}=10\) (\(\mathcal {R_T}=0.7864\)), \(r_1=r_2=r_{S1}=r_{S2}=12\) (\(\mathcal {R_T}=0.6621\)) and \(r_1=r_2=r_{S1}=r_{S2}=15\) (\(\mathcal {R_T}=0.5341\)) and \(\mathcal {R_H} = 2.2043\). All other parameter values are as given in Table 3
Figure 14 reveals that increasing the treatment rates for singly infected individuals with primary and secondary syphilis infection, \(r_1\) (Fig. 14a) and \(r_2\) (Fig. 14a), respectively, results in a positive population-level impact on the burden of syphilis, as we observe a reduction in the prevalence of primary syphilis (\(I_P\)), secondary syphilis (\(I_S\)), early latent syphilis (\(L_{S1}\)), but a marginal effect on the prevalence of late latent syphilis (\(L_{S2}\)), in the long run.
Figure 15a shows that when the treatment rate of singly infected individuals with primary syphilis, (\(r_1\)), is varied, it has a positive population-level impact on the number of infected individuals with HIV and tertiary syphilis (\(H_T\)) but a marginal effect on the number of infected individuals with tertiary syphilis (\(I_T\)), in the long run, while Fig. 15b shows a decrease in the prevalence of HIV and syphilis co-infection as we increase the syphilis treatment rate of dually infected individuals with HIV and primary syphilis (\(r_{S1}\)), as expected.
Plots of the number of infected individuals with primary syphilis, (\(I_P\)), secondary syphilis, (\(I_S\)), early latent syphilis, (\(L_{S1}\)), and late latent syphilis, (\(L_{S2}\)). a Here, we vary the treatment rate of singly infected individuals with primary syphilis, (\(r_1\)), from 2 to 10, where \(\beta _S = 10\) and \(\beta _H = 0.6\), with \({\mathcal {R}}_H = 1.6532\). All other parameter values are as given in Table 3. b Here, we vary the treatment rate of singly infected individuals with secondary syphilis, (\(r_2\)), from 2 to 10, where \(\beta _S = 10\) and \(\beta _H = 0.6\), with \({\mathcal {R}}_H = 1.6532\). All other parameter values are as given in Table 3
Plots of the number of a infected persons with tertiary syphilis, (\(I_T\)), and HIV with tertiary syphilis, (\(H_T\)), where \(\beta _S = 10\), \(\beta _H = 0.6\) and \({\mathcal {R}}_H = 1.6532\) with variations in \(r_1\). b HIV-infected individuals with primary syphilis (\(H_P\)), secondary syphilis (\(H_S\)), early latent syphilis (\(H_{S1}\)) and late latent syphilis (\(H_{S2}\)), where \(\beta _S = 6\) and \(\beta _H = 1\), with \({\mathcal {R}}_H = 2.7554\). All other parameter values are as given in Table 3. a Treatment rate of singly infected individuals with primary syphilis (\(r_1\)) is varied from 2 to 10. b Treatment rate of dually infected individuals with primary syphilis (\(r_{S1}\)) is varied from 2 to 10
Rights and permissions
About this article
Cite this article
Nwankwo, A., Okuonghae, D. Mathematical Analysis of the Transmission Dynamics of HIV Syphilis Co-infection in the Presence of Treatment for Syphilis. Bull Math Biol 80, 437–492 (2018). https://doi.org/10.1007/s11538-017-0384-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0384-0