Abstract
Background
Cetuximab, a chimeric monoclonal antibody against EGFR sensitizes tumors to radiotherapy (RT), but is associated with skin and mucosal toxicity.
Objective
We report outcomes and tolerance of definitive RT in association with cetuximab in patients with locally advanced squamous cell carcinoma (LASCC) of the head and neck.
Patients and Methods
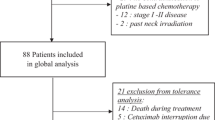
Between 2006 and 2011, 92 consecutive patients with LASCC of the head and neck were treated with RT and concomitant weekly cetuximab. Median age was 61.7 years. Most patients presented with oropharyngeal tumors (52.2%) and stage IV disease (77.2%).
Results
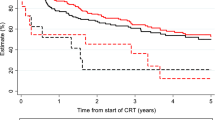
Sixty-nine patients received at least 7 cycles of cetuximab. Cetuximab was stopped at the first infusion following allergic reactions in four patients. During RT, 37% of patients developed grade ≥ 3 dermatitis; grade ≥ 2 cetuximab-induced rash occurred in 43 patients (46.7%). Severe mucositis (grade ≥ 3) affected 57.6% of patients. Ten percent of patients did not receive the full course of RT, and temporary discontinuation due to acute toxicity was frequent and affected 37 patients (53%). The median RT overall treatment time (OTT) in patients with interrupted RT was 56 days (47–75) compared to 51 days (47–65) in patients who did not require toxicity-related radiation interruptions (p < 0.05). After a median follow-up of 17.5 months (1.3–107.6) for all patients, median overall survival was 17.9 months (95% CI: 12.7–23.2), and loco-regional control (LRC) was 9.2 months (95% CI: 3.9–14.4). On multivariate analysis, hemoglobin concentration and occurrence of rash grade ≥ 2 were independent prognostic factors for LRC (p = 0.023 and p = 0.006, respectively). Lack of rash and extended OTT negatively impacted overall survival (p = 0.048 and 0.052, respectively).
Conclusions
Skin and mucosal toxicity remains an issue in patients with LASCC of the head and neck treated with concomitant cetuximab and RT. Severe toxicity leads to treatment interruptions and prolonged overall treatment time, with consequent decreased overall survival in these patients.




Similar content being viewed by others
References
Pignon JP, le Maitre A, Maillard E, Bourhis J, Group M-NC. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. Radiother Oncol. 2009;92(1):4–14.
Van den Bogaert W, Van der Leest A, Rijnders A, Delaere P, Thames H, van der Schueren E. Does tumor control decrease by prolonging overall treatment time or interrupting treatment in laryngeal cancer? Radiother Oncol. 1995;36(3):177–82.
Specenier P, Vermorken JB. Cetuximab: its unique place in head and neck cancer treatment. Biologics. 2013;7:77–90.
Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, Cohen RB, Jones CU, Sur R, Raben D, Jassem J, et al. Radiotherapy plus Cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006;354(6):567–78.
Giro C, Berger B, Bolke E, Ciernik IF, Duprez F, Locati L, Maillard S, Ozsahin M, Pfeffer R, Robertson AG, et al. High rate of severe radiation dermatitis during radiation therapy with concurrent Cetuximab in head and neck cancer: results of a survey in EORTC institutes. Radiother Oncol. 2009;90(2):166–71.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nuclear Med. 2009;50(Suppl 1):122S–50S.
Walsh L, Gillham C, Dunne M, Fraser I, Hollywood D, Armstrong J, Thirion P. Toxicity of Cetuximab versus cisplatin concurrent with radiotherapy in locally advanced head and neck squamous cell cancer (LAHNSCC). Radiother Oncol. 2011;98(1):38–41.
Koutcher L, Sherman E, Fury M, Wolden S, Zhang Z, Mo Q, Stewart L, Schupak K, Gelblum D, Wong R, et al. Concurrent cisplatin and radiation versus Cetuximab and radiation for locally advanced head-and-neck cancer. Int J Radiat Oncol Biol Phys. 2011;81(4):915–22.
Vera-Llonch M, Oster G, Hagiwara M, Sonis S. Oral mucositis in patients undergoing radiation treatment for head and neck carcinoma. Cancer. 2006;106(2):329–36.
Gonzalez Ferreira JA, Jaen Olasolo J, Azinovic I, Jeremic B. Effect of radiotherapy delay in overall treatment time on local control and survival in head and neck cancer: review of the literature. Rep Pract Oncol Radiother. 2015;20(5):328–39.
Bernier J, Russi EG, Homey B, Merlano MC, Mesia R, Peyrade F, Budach W. Management of radiation dermatitis in patients receiving Cetuximab and radiotherapy for locally advanced squamous cell carcinoma of the head and neck: proposals for a revised grading system and consensus management guidelines. Ann Oncol. 2011;22(10):2191–200.
Zecha JA, Raber-Durlacher JE, Nair RG, Epstein JB, Elad S, Hamblin MR, Barasch A, Migliorati CA, Milstein DM, Genot MT, et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: part 2: proposed applications and treatment protocols. Support Care Cancer. 2016;24(6):2793–805.
Kogawa T, Doi A, Shimokawa M, Fouad TM, Osuga T, Tamura F, Mizushima T, Kimura T, Abe S, Ihara H, et al. Early skin toxicity predicts better outcomes, and early tumor shrinkage predicts better response after Cetuximab treatment in advanced colorectal cancer. Target Oncol. 2015;10(1):125–33.
Petrelli F, Borgonovo K, Barni S. The predictive role of skin rash with Cetuximab and panitumumab in colorectal cancer patients: a systematic review and meta-analysis of published trials. Target Oncol. 2013;8(3):173–81.
Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, Sur RK, Raben D, Baselga J, Spencer SA, Zhu J, et al. Radiotherapy plus Cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between Cetuximab-induced rash and survival. Lancet Oncol. 2010;11(1):21–8.
Narayanaswamy RK, Potharaju M, Vaidhyswaran AN, Perumal K. Pre-radiotherapy Haemoglobin level is a prognosticator in locally advanced head and neck cancers treated with concurrent Chemoradiation. J Clin Diagn Res. 2015;9(6):XC14–8.
Magrini SM, Buglione M, Corvo R, Pirtoli L, Paiar F, Ponticelli P, Petrucci A, Bacigalupo A, Crociani M, Lastrucci L, et al. Cetuximab and radiotherapy versus cisplatin and radiotherapy for locally advanced head and neck cancer: a randomized phase II trial. J Clin Oncol. 2016;34(5):427–35.
Rosenthal DI, Harari PM, Giralt J, Bell D, Raben D, Liu J, Schulten J, Ang KK, Bonner JA. Association of Human Papillomavirus and p16 status with outcomes in the IMCL-9815 phase III registration trial for patients with Locoregionally advanced oropharyngeal squamous cell carcinoma of the head and neck treated with radiotherapy with or without Cetuximab. J Clin Oncol. 2016;34(12):1300–8.
Iyer NG, Tan DS, Tan VK, Wang W, Hwang J, Tan NC, Sivanandan R, Tan HK, Lim WT, Ang MK, et al. Randomized trial comparing surgery and adjuvant radiotherapy versus concurrent chemoradiotherapy in patients with advanced, nonmetastatic squamous cell carcinoma of the head and neck: 10-year update and subset analysis. Cancer. 2015;121(10):1599–607.
Petrelli F, Coinu A, Riboldi V, Borgonovo K, Ghilardi M, Cabiddu M, Lonati V, Sarti E, Barni S. Concomitant platinum-based chemotherapy or Cetuximab with radiotherapy for locally advanced head and neck cancer: a systematic review and meta-analysis of published studies. Oral Oncol. 2014;50(11):1041–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflict of Interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Roman, J., Dissaux, G., Gouillou, M. et al. Prolonged Overall Treatment Time and Lack of Skin Rash Negatively Impact Overall Survival in Locally Advanced Head and Neck Cancer Patients Treated with Radiotherapy and Concomitant Cetuximab. Targ Oncol 12, 505–512 (2017). https://doi.org/10.1007/s11523-017-0499-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-017-0499-0