Abstract
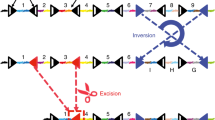
MADM (Mosaic Analysis with Double Markers) technology offers a genetic approach in mice to visualize and concomitantly manipulate genetically defined cells at clonal level and single cell resolution. MADM employs Cre recombinase/loxP-dependent interchromosomal mitotic recombination to reconstitute two split marker genes—green GFP and red tdTomato—and can label sparse clones of homozygous mutant cells in one color and wild-type cells in the other color in an otherwise unlabeled background. At present, major MADM applications include lineage tracing, single cell labeling, conditional knockouts in small populations of cells and induction of uniparental chromosome disomy to assess effects of genomic imprinting. MADM can be applied universally in the mouse with the sole limitation being the specificity of the promoter controlling Cre recombinase expression. Here I review recent developments and extensions of the MADM technique and give an overview of the major discoveries and progresses enabled by the implementation of the novel genetic MADM tools.
Similar content being viewed by others
References
Armakolas A, Klar A J (2006). Cell type regulates selective segregation of mouse chromosome 7 DNA strands in mitosis. Science, 311(5764): 1146–1149
Ayala R, Shu T, Tsai L H (2007). Trekking across the brain: the journey of neuronal migration. Cell, 128(1): 29–43
Badea T C, Wang Y, Nathans J (2003). A noninvasive genetic/pharmacologic strategy for visualizing cell morphology and clonal relationships in the mouse. J Neurosci, 23(6): 2314–2322
Barlow D P (2011). Genomic imprinting: a mammalian epigenetic discovery model. Annu Rev Genet, 45(1): 379–403
Bartolomei M S, Ferguson-Smith A C (2011). Mammalian genomic imprinting. Cold Spring Harb Perspect Biol, 3(7): 3
Bi W, Yan J, Stankiewicz P, Park S S, Walz K, Boerkoel C F, Potocki L, Shaffer L G, Devriendt K, Nowaczyk M J, Inoue K, Lupski J R (2002). Genes in a refined Smith-Magenis syndrome critical deletion interval on chromosome 17p11.2 and the syntenic region of the mouse. Genome Res, 12(5): 713–728
Blair S S (2003). Genetic mosaic techniques for studying Drosophila development. Development, 130(21): 5065–5072
Bonaguidi MA, Wheeler MA, Shapiro J S, Stadel R P, Sun G J, Ming G L, Song H (2011). In vivo clonal analysis reveals self-renewing and multipotent adult neural stem cell characteristics. Cell, 145(7): 1142–1155
Branda C S, Dymecki S M (2004). Talking about a revolution: The impact of site-specific recombinases on genetic analyses in mice. Dev Cell, 6(1): 7–28
Brennand K, Huangfu D, Melton D (2007). All beta cells contribute equally to islet growth and maintenance. PLoS Biol, 5(7): e163
Buckingham M E, Meilhac S M (2011). Tracing cells for tracking cell lineage and clonal behavior. Dev Cell, 21(3): 394–409
Cajal S R y (1911). Histology of the Nervous System of Man and Vertebrates. Oxford University Press, Inc, Oxford 1995
Translation Cepko C, Ryder E F, Austin C P, Walsh C, Fekete D M (1995). Lineage analysis using retrovirus vectors. Methods Enzymol, 254: 387–419
Chow B Y, Han X, Boyden E S (2012). Genetically encoded molecular tools for light-driven silencing of targeted neurons. Prog Brain Res, 196: 49–61
Cowan W M (1998). The emergence of modern neuroanatomy and developmental neurobiology. Neuron, 20(3): 413–426
De Paola V, Arber S, Caroni P (2003). AMPA receptors regulate dynamic equilibrium of presynaptic terminals in mature hippocampal networks. Nat Neurosci, 6(5): 491–500
Desgraz R, Herrera P L (2009). Pancreatic neurogenin 3-expressing cells are unipotent islet precursors. Development, 136(21): 3567–3574
Dessaud E, Yang L L, Hill K, Cox B, Ulloa F, Ribeiro A, Mynett A, Novitch B G, Briscoe J (2007). Interpretation of the sonic hedgehog morphogen gradient by a temporal adaptation mechanism. Nature, 450(7170): 717–720
Dymecki S M, Kim J C (2007). Molecular neuroanatomy’s “Three Gs”: a primer. Neuron, 54(1): 17–34
Espinosa J S, Luo L (2008). Timing neurogenesis and differentiation: insights from quantitative clonal analyses of cerebellar granule cells. J Neurosci, 28: 2301–2312
Espinosa J S, Wheeler D G, Tsien R W, Luo L (2009). Uncoupling dendrite growth and patterning: single-cell knockout analysis of NMDA receptor 2B. Neuron, 62(2): 205–217
Feil R, Brocard J, Mascrez B, LeMeur M, Metzger D, Chambon P (1996). Ligand-activated site-specific recombination in mice. Proc Natl Acad Sci USA, 93(20): 10887–10890
Feinberg A P (2007). Phenotypic plasticity and the epigenetics of human disease. Nature, 447(7143): 433–440
Feng G, Mellor R H, Bernstein M, Keller-Peck C, Nguyen Q T, Wallace M, Nerbonne J M, Lichtman J W, Sanes J R (2000). Imaging neuronal subsets in transgenic mice expressing multiple spectral variants of GFP. Neuron, 28(1): 41–51
Foo L C, Allen N J, Bushong E A, Ventura P B, Chung W S, Zhou L, Cahoy J D, Daneman R, Zong H, Ellisman M H, Barres B A (2011). Development of a method for the purification and culture of rodent astrocytes. Neuron, 71(5): 799–811
Franco S J, Müller U (2013). Shaping our minds: stem and progenitor cell diversity in the mammalian neocortex. Neuron, 77(1): 19–34
Gao P, Sultan K T, Zhang X J, Shi S H (2013). Lineage-dependent circuit assembly in the neocortex. Development, 140(13): 2645–2655
Gorski J A, Talley T, Qiu M, Puelles L, Rubenstein J L, Jones K R (2002). Cortical excitatory neurons and glia, but not GABAergic neurons, are produced in the Emx1-expressing lineage. J Neurosci, 22: 6309–6314
Hallonet M E, Le Douarin N M (1993). Tracing neuroepithelial cells of the mesencephalic and metencephalic alar plates during cerebellar ontogeny in quail-chick chimaeras. Eur J Neurosci, 5(9): 1145–1155
Hayashi S, McMahon A P (2002). Efficient recombination in diverse tissues by a tamoxifen-inducible form of Cre: a tool for temporally regulated gene activation/inactivation in the mouse. Dev Biol, 244(2): 305–318
Hegemann P, Möglich A (2011). Channelrhodopsin engineering and exploration of new optogenetic tools. Nat Methods, 8(1): 39–42
Hippenmeyer S, Johnson R L, Luo L (2013). Mosaic analysis with double markers reveals cell-type-specific paternal growth dominance. Cell Rep, 3: 960–967
Hippenmeyer S, Vrieseling E, Sigrist M, Portmann T, Laengle C, Ladle D R, Arber S (2005). A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol, 3(5): e159
Hippenmeyer S, Youn Y H, Moon H M, Miyamichi K, Zong H, Wynshaw-Boris A, Luo L (2010). Genetic mosaic dissection of Lis1 and Ndel1 in neuronal migration. Neuron, 68(4): 695–709
Imayoshi I, Ohtsuka T, Metzger D, Chambon P, Kageyama R (2006). Temporal regulation of Cre recombinase activity in neural stem cells. Genesis, 44(5): 233–238
Indra A K, Warot X, Brocard J, Bornert J M, Xiao J H, Chambon P, Metzger D (1999). Temporally-controlled site-specific mutagenesis in the basal layer of the epidermis: comparison of the recombinase activity of the tamoxifen-inducible Cre-ER(T) and Cre-ER(T2) recombinases. Nucleic Acids Res, 27(22): 4324–4327
Jefferis G S, Livet J (2012). Sparse and combinatorial neuron labelling. Curr Opin Neurobiol, 22(1): 101–110
Lao Z, Raju G P, Bai C B, Joyner A L (2012). MASTR: a technique for mosaic mutant analysis with spatial and temporal control of recombination using conditional floxed alleles in mice. Cell Rep, 2: 386–396
Lee T, Luo L (1999). Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron, 22(3): 451–461
Legué E, Joyner A L (2010). Genetic fate mapping using site-specific recombinases. Methods Enzymol, 477: 153–181
Lehtinen M K, Walsh C A (2011). Neurogenesis at the braincerebrospinal fluid interface. Annu Rev Cell Dev Biol, 27(1): 653–679
Lewandoski M (2001). Conditional control of gene expression in the mouse. Nat Rev Genet, 2(10): 743–755
Liang H, Xiao G, Yin H, Hippenmeyer S, Horowitz J M, Ghashghaei H T (2013). Neural development is dependent on the function of specificity protein 2 in cell cycle progression. Development, 140(3): 552–561
Liu C, Sage J C, Miller M R, Verhaak R G, Hippenmeyer S, Vogel H, Foreman O, Bronson R T, Nishiyama A, Luo L, Zong H (2011). Mosaic analysis with double markers reveals tumor cell of origin in glioma. Cell, 146(2): 209–221
Liu P, Jenkins N A, Copeland N G (2002). Efficient Cre-loxP-induced mitotic recombination in mouse embryonic stem cells. Nat Genet, 30(1): 66–72
Lui J H, Hansen D V, Kriegstein A R (2011). Development and evolution of the human neocortex. Cell, 146(1): 18–36
Luo L (2007). Fly MARCM and mouse MADM: genetic methods of labeling and manipulating single neurons. Brain Res Brain Res Rev, 55(2): 220–227
Mabb A M, Judson M C, Zylka M J, Philpot B D (2011). Angelman syndrome: insights into genomic imprinting and neurodevelopmental phenotypes. Trends Neurosci, 34(6): 293–303
Madisen L, Zwingman T A, Sunkin S M, Oh S W, Zariwala H A, Gu H, Ng L L, Palmiter R D, Hawrylycz M J, Jones A R, Lein E S, Zeng H (2010). A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci, 13(1): 133–140
Marín O, Valiente M, Ge X, Tsai L H (2010). Guiding neuronal cell migrations. Cold Spring Harb Perspect Biol, 2(2): a001834
McConnell S K (1988). Fates of visual cortical neurons in the ferret after isochronic and heterochronic transplantation. J Neurosci, 8: 945–974
Merkle F T, Mirzadeh Z, Alvarez-Buylla A (2007). Mosaic organization of neural stem cells in the adult brain. Science, 317(5836): 381–384
Metzger D, Chambon P (2001). Site- and time-specific gene targeting in the mouse. Methods, 24(1): 71–80
Ming G L, Song H (2011). Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron, 70(4): 687–702
Miyoshi G, Hjerling-Leffler J, Karayannis T, Sousa V H, Butt S J, Battiste J, Johnson J E, Machold R P, Fishell G (2010). Genetic fate mapping reveals that the caudal ganglionic eminence produces a large and diverse population of superficial cortical interneurons. J Neurosci, 30: 1582–1594
Morgan T H (1914). Mosaics and gynandromorphs in Drosophila. Proc Soc Exp Biol Med, 11(6): 171–172
Muzumdar M D, Luo L, Zong H (2007). Modeling sporadic loss of heterozygosity in mice by using mosaic analysis with double markers (MADM). Proc Natl Acad Sci USA, 104(11): 4495–4500
Nelson S B, Sugino K, Hempel C M (2006). The problem of neuronal cell types: a physiological genomics approach. Trends Neurosci, 29(6): 339–345
Nicholls R D, Knepper J L (2001). Genome organization, function, and imprinting in Prader-Willi and Angelman syndromes. Annu Rev Genomics Hum Genet, 2(1): 153–175
Ninkovic J, Gotz M (2013). Fate specification in the adult brain-lessons for eliciting neurogenesis from glial cells. BioEssays, 35: 242–252
Novak A, Guo C, Yang W, Nagy A, Lobe C G (2000). Z/EG, a double reporter mouse line that expresses enhanced green fluorescent protein upon Cre-mediated excision. Genesis, 28(3–4): 147–155
Petersen P H, Zou K, Hwang J K, Jan Y N, Zhong W (2002). Progenitor cell maintenance requires numb and numblike during mouse neurogenesis. Nature, 419(6910): 929–934
Reiner O, Carrozzo R, Shen Y, Wehnert M, Faustinella F, Dobyns W B, Caskey C T, Ledbetter D H (1993). Isolation of a Miller-Dieker lissencephaly gene containing G protein beta-subunit-like repeats. Nature, 364(6439): 717–721
Ross M E, Walsh C A (2001). Human brain malformations and their lessons for neuronal migration. Annu Rev Neurosci, 24(1): 1041–1070
Sanes J R (1989). Analysing cell lineage with a recombinant retrovirus. Trends Neurosci, 12(1): 21–28
Schnütgen F, Doerflinger N, Calléja C, Wendling O, Chambon P, Ghyselinck N B (2003). A directional strategy for monitoring Cremediated recombination at the cellular level in the mouse. Nat Biotechnol, 21(5): 562–565
Shaner N C, Campbell R E, Steinbach P A, Giepmans B N, Palmer A E, Tsien R Y (2004). Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nat Biotechnol, 22(12): 1567–1572
Smith G B, Fitzpatrick D (2012). Specifying cortical circuits: a role for cell lineage. Neuron, 75(1): 4–5
Soriano P (1999). Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat Genet, 21(1): 70–71
Stern C (1936). Somatic Crossing over and Segregation in Drosophila Melanogaster. Genetics, 21(6): 625–730
Tasic B, Miyamichi K, Hippenmeyer S, Dani V S, Zeng H, Joo W, Zong H, Chen-Tsai Y, Luo L (2012). Extensions of MADM (mosaic analysis with double markers) in mice. PLoS ONE, 7(3): e33332
Tronche F, Kellendonk C, Kretz O, Gass P, Anlag K, Orban P C, Bock R, Klein R, Schütz G (1999). Disruption of the glucocorticoid receptor gene in the nervous system results in reduced anxiety. Nat Genet, 23(1): 99–103
Tsai J W, Chen Y, Kriegstein A R, Vallee R B (2005). LIS1 RNA interference blocks neural stem cell division, morphogenesis, and motility at multiple stages. J Cell Biol, 170(6): 935–945
Walsh C, Cepko C L (1992). Widespread dispersion of neuronal clones across functional regions of the cerebral cortex. Science, 255(5043): 434–440
Wingate R J, Hatten M E (1999). The role of the rhombic lip in avian cerebellum development. Development, 126(20): 4395–4404
Woodruff A, Xu Q, Anderson S A, Yuste R (2009). Depolarizing effect of neocortical chandelier neurons. Front Neural Circuits 3: 15
Wynshaw-Boris A, Pramparo T, Youn Y H, Hirotsune S (2010). Lissencephaly: mechanistic insights from animal models and potential therapeutic strategies. Semin Cell Dev Biol, 21(8): 823–830
Xu Q, Tam M, Anderson S A (2008). Fate mapping Nkx2.1-lineage cells in the mouse telencephalon. J Comp Neurol, 506(1): 16–29
Xu T, Rubin GM (1993). Analysis of genetic mosaics in developing and adult Drosophila tissues. Development, 117(4): 1223–1237
Yang S B, Mclemore K D, Tasic B, Luo L, Jan Y N, Jan L Y (2012). Kv1.1-dependent control of hippocampal neuron number as revealed by mosaic analysis with double markers. J Physiol, 590(Pt 11): 2645–2658
Yingling J, Toyo-Oka K, Wynshaw-Boris A (2003). Miller-Dieker syndrome: analysis of a human contiguous gene syndrome in the mouse. Am J Hum Genet, 73(3): 475–488
Yingling J, Youn Y H, Darling D, Toyo-Oka K, Pramparo T, Hirotsune S, Wynshaw-Boris A (2008). Neuroepithelial stem cell proliferation requires LIS1 for precise spindle orientation and symmetric division. Cell, 132(3): 474–486
Youn Y H, Pramparo T, Hirotsune S, Wynshaw-Boris A (2009). Distinct dose-dependent cortical neuronal migration and neurite extension defects in Lis1 and Ndel1 mutant mice. J Neurosci, 29: 15520–15530
Young P, Qiu L, Wang D, Zhao S, Gross J, Feng G (2008). Singleneuron labeling with inducible Cre-mediated knockout in transgenic mice. Nat Neurosci, 11(6): 721–728
Zhang F, Aravanis A M, Adamantidis A, de Lecea L, Deisseroth K (2007). Circuit-breakers: optical technologies for probing neural signals and systems. Nat Rev Neurosci, 8(8): 577–581
Zhu X, Bergles D E, Nishiyama A (2008). NG2 cells generate both oligodendrocytes and gray matter astrocytes. Development, 135(1): 145–157
Zhuo L, Theis M, Alvarez-Maya I, Brenner M, Willecke K, Messing A (2001). hGFAP-cre transgenic mice for manipulation of glial and neuronal function in vivo. Genesis, 31(2): 85–94
Zong H, Espinosa J S, Su H H, Muzumdar M D, Luo L (2005). Mosaic analysis with double markers in mice. Cell, 121(3): 479–492
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hippenmeyer, S. Dissection of gene function at clonal level using mosaic analysis with double markers. Front. Biol. 8, 557–568 (2013). https://doi.org/10.1007/s11515-013-1279-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-013-1279-6