Abstract
Cortical cerebral ischemia elicits neuroinflammation as well as secondary neuronal degeneration in remote areas. Locally distinct and specific secondary neurodegeneration affecting thalamic nuclei connected to cortical areas highlights such processes. Osteopontin (OPN) is a cytokine-like glycoprotein that is excreted in high amounts after cerebral ischemia and exerts various immunomodulatory functions. We here examined putative protective effects of OPN in secondary thalamic degeneration. We subjected male Wistar rats to photothrombosis and subsequently injected OPN or placebo intracerebroventricularly. Immunohistochemical and fluorescence staining was used to detect the extent of neuronal degeneration and microglia activation. Ex vivo autoradiography with radiotracers available for human in vivo PET studies, i.e., cis-4-[18F]Fluor-d-Proline (D-cis-[18F]FPro), and [6-3H]thymidine ([3H]thymidine), confirmed degeneration and proliferation, respectively. We found secondary neurodegeneration in the thalamus characterized by microglial activation and neuronal loss. Neuronal loss was restricted to areas of microglial infiltration. Treatment with OPN significantly decreased neurodegeneration, inflammation and microglial proliferation. Microglia displayed morphological signs of activation without expressing markers of M1 or M2 polarization. D-cis-[18F]FPro-uptake mirrored attenuated degeneration in OPN-treated animals. Notably, [3H]thymidine and BrdU-staining revealed increased stem cell proliferation after treatment with OPN. The data suggest that OPN is able to ameliorate secondary neurodegeneration in thalamic nuclei. These effects can be visualized by radiotracers D-cis-[18F]FPro and [3H]thymidine, opening new vistas for translational studies.

Intracerebroventricular injection of osteopontin attenuates thalamic degeneration after cortical ischemia (pink area). Disruption of thalamocortical connections (blue) and degeneration of thalamic nuclei (encircled) leads to microglia activation. Osteopontin protects from both neurodegeneration and microglia activation as assessed by histological analysis and autoradiograpic studies.







Similar content being viewed by others
References
Becerra-Calixto A, Cardona-Gómez GP (2017) The role of astrocytes in neuroprotection after brain stroke: potential in cell therapy. Front Mol Neurosci 10. https://doi.org/10.3389/fnmol.2017.00088
Bhatt A, Fan L-W, Pang Y (2014) Strategies for myelin regeneration: lessons learned from development. Neural Regen Res 9:1347–1350. https://doi.org/10.4103/1673-5374.137586
Butler WT (1989) The nature and significance of osteopontin. Connect Tissue Res 23:123–136
Butler WT (1995) Structural and functional domains of osteopontin. Ann N Y Acad Sci 760:6–11
Chamberlain KA, Nanescu SE, Psachoulia K, Huang JK (2016) Oligodendrocyte regeneration: its significance in myelin replacement and neuroprotection in multiple sclerosis. Neuropharmacology 110:633–643. https://doi.org/10.1016/j.neuropharm.2015.10.010
Chávez-Galán L, Olleros ML, Vesin D, Garcia I (2015) Much More than M1 and M2 Macrophages, There are also CD169+ and TCR+ Macrophages. Front Immunol 6:263. https://doi.org/10.3389/fimmu.2015.00263
Chen W, Ma Q, Suzuki H et al (2011) Osteopontin reduced hypoxia-ischemia neonatal brain injury by suppression of apoptosis in a rat pup model. Stroke J Cereb Circ 42:764–769. https://doi.org/10.1161/STROKEAHA.110.599118
De Vloo P, Morlion B, van Loon J, Nuttin B (2017) Animal models for central poststroke pain: a critical comprehensive review. Pain 158:17–29. https://doi.org/10.1097/j.pain.0000000000000722
Denes A, Vidyasagar R, Feng J et al (2007) Proliferating resident microglia after focal cerebral ischaemia in mice. J Cereb Blood Flow Metab 27:1941–1953. https://doi.org/10.1038/sj.jcbfm.9600495
Denhardt DT, Noda M, O’Regan AW et al (2001) Osteopontin as a means to cope with environmental insults: regulation of inflammation, tissue remodeling, and cell survival. J Clin Invest 107:1055–1061
Doyle KP, Yang T, Lessov NS et al (2008) Nasal administration of osteopontin peptide mimetics confers neuroprotection in stroke. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 28:1235–1248. https://doi.org/10.1038/jcbfm.2008.17
Ellison JA, Velier JJ, Spera P et al (1998) Osteopontin and its integrin receptor alpha(v)beta3 are upregulated during formation of the glial scar after focal stroke. Stroke J Cereb Circ 29:1698–1706 discussion 1707
Freret T, Chazalviel L, Roussel S et al (2006) Long-term functional outcome following transient middle cerebral artery occlusion in the rat: correlation between brain damage and behavioral impairment. Behav Neurosci 120:1285–1298. https://doi.org/10.1037/0735-7044.120.6.1285
Fu G-X, Xu C-C, Zhong Y et al (2012) Aldosterone-induced osteopontin expression in vascular smooth muscle cells involves MR, ERK, and p38 MAPK. Endocrine 42:676–683. https://doi.org/10.1007/s12020-012-9675-2
Geisler S, Willuweit A, Schroeter M et al (2013) Detection of remote neuronal reactions in the thalamus and Hippocampus induced by rat glioma using the PET tracer cis-4-[18F]fluoro-풟-proline. J Cereb Blood Flow Metab 33:724–731. https://doi.org/10.1038/jcbfm.2013.8
Giachelli CM, Liaw L, Murry CE et al (1995) Osteopontin expression in cardiovascular diseases. Ann N Y Acad Sci 760:109–126
Gliem M, Krammes K, Liaw L et al (2015) Macrophage-derived osteopontin induces reactive astrocyte polarization and promotes re-establishment of the blood brain barrier after ischemic stroke. Glia. https://doi.org/10.1002/glia.22885
Hedtjärn M, Mallard C, Hagberg H (2004) Inflammatory gene profiling in the developing mouse brain after hypoxia-ischemia. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 24:1333–1351. https://doi.org/10.1097/01.WCB.0000141559.17620.36
Hu X, Li P, Guo Y et al (2012) Microglia/macrophage polarization dynamics reveal novel mechanism of injury expansion after focal cerebral ischemia. Stroke J Cereb Circ 43:3063–3070. https://doi.org/10.1161/STROKEAHA.112.659656
Hucklenbroich J, Klein R, Neumaier B et al (2014) Aromatic-turmerone induces neural stem cell proliferation in vitro and in vivo. Stem Cell Res Ther 5:100. https://doi.org/10.1186/scrt500
Hur EM, Youssef S, Haws ME et al (2007) Osteopontin-induced relapse and progression of autoimmune brain disease through enhanced survival of activated T cells. Nat Immunol 8:74–83. https://doi.org/10.1038/ni1415
Iizuka H, Sakatani K, Young W (1990) Neural damage in the rat thalamus after cortical infarcts. Stroke 21:790–794
Ikeshima-Kataoka H, Matsui Y, Uede T (2018) Osteopontin is indispensable for activation of astrocytes in injured mouse brain and primary culture. Neurol Res 40:1071–1079. https://doi.org/10.1080/01616412.2018.1517995
Itoh K, Maki T, Lok J, Arai K (2015) Mechanisms of cell-cell interaction in oligodendrogenesis and remyelination after stroke. Brain Res 1623:135–149. https://doi.org/10.1016/j.brainres.2015.04.039
Kanazawa M, Ninomiya I, Hatakeyama M, Takahashi T, Shimohata T (2017) Microglia and Monocytes/Macrophages Polarization Reveal Novel Therapeutic Mechanism against Stroke. Int J Mol Sci 18(10):2135
Kato A, Okura T, Hamada C et al (2014) Cell stress induces upregulation of osteopontin via the ERK pathway in type II alveolar epithelial cells. PLoS One 9:e100106. https://doi.org/10.1371/journal.pone.0100106
Klein R, Blaschke S, Neumaier B et al (2014) The synthetic NCAM mimetic peptide FGL mobilizes neural stem cells in vitro and in vivo. Stem Cell Rev 10:539–547. https://doi.org/10.1007/s12015-014-9512-5
Klein R, Mahlberg N, Ohren M et al (2016) The neural cell adhesion molecule-derived (NCAM)-peptide FG loop (FGL) mobilizes endogenous neural stem cells and promotes endogenous regenerative capacity after stroke. J Neuroimmune Pharmacol Off J Soc NeuroImmune Pharmacol 11:708–720. https://doi.org/10.1007/s11481-016-9694-5
Kumar S, Sharma P, Kumar D et al (2013) Functional characterization of stromal osteopontin in melanoma progression and metastasis. PLoS One 8:e69116. https://doi.org/10.1371/journal.pone.0069116
Ladwig A, Walter HL, Hucklenbroich J et al (2017) Osteopontin augments M2 microglia response and separates M1- and M2-polarized microglial activation in permanent focal cerebral ischemia. Mediators Inflamm 2017. https://doi.org/10.1155/2017/7189421
Langen K-J, Salber D, Hamacher K et al (2007) Detection of secondary thalamic degeneration after cortical infarction using cis-4-18F-fluoro-D-proline. J Nucl Med Off Publ Soc Nucl Med 48:1482–1491. https://doi.org/10.2967/jnumed.107.041699
Lee MY, Shin SL, Choi YS et al (1999) Transient upregulation of osteopontin mRNA in hippocampus and striatum following global forebrain ischemia in rats. Neurosci Lett 271:81–84
Matsumoto H, Kumon Y, Watanabe H et al (2008) Accumulation of macrophage-like cells expressing NG2 proteoglycan and Iba1 in ischemic core of rat brain after transient middle cerebral artery occlusion. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 28:149–163. https://doi.org/10.1038/sj.jcbfm.9600519
McKee MD, Nanci A (1996) Secretion of Osteopontin by macrophages and its accumulation at tissue surfaces during wound healing in mineralized tissues: a potential requirement for macrophage adhesion and phagocytosis. Anat Rec 245:394–409. https://doi.org/10.1002/(SICI)1097-0185(199606)245:2<394::AID-AR19>3.0.CO;2-K
Meller R, Stevens SL, Minami M et al (2005) Neuroprotection by osteopontin in stroke. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 25:217–225. https://doi.org/10.1038/sj.jcbfm.9600022
Morales-Ibanez O, Domínguez M, Ki SH et al (2013) Human and experimental evidence supporting a role for osteopontin in alcoholic hepatitis. Hepatol Baltim Md 58:1742–1756. https://doi.org/10.1002/hep.26521
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol 8:958–969. https://doi.org/10.1038/nri2448
Myers R, Manjil LG, Cullen BM et al (1991) Macrophage and astrocyte populations in relation to [3H]PK 11195 binding in rat cerebral cortex following a local ischaemic lesion. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 11:314–322. https://doi.org/10.1038/jcbfm.1991.64
Myles T, Leung LLK (2008) Thrombin hydrolysis of human osteopontin is dependent on thrombin anion-binding exosites. J Biol Chem 283:17789–17796. https://doi.org/10.1074/jbc.M708629200
Ogawa T, Yoshida Y, Okudera T et al (1997) Secondary thalamic degeneration after cerebral infarction in the middle cerebral artery distribution: evaluation with MR imaging. Radiology 204:255–262. https://doi.org/10.1148/radiology.204.1.9205256
Orihuela R, McPherson CA, Harry GJ (2016) Microglial M1/M2 polarization and metabolic states. Br J Pharmacol 173:649–665. https://doi.org/10.1111/bph.13139
Pappata S, Levasseur M, Gunn RN et al (2000) Thalamic microglial activation in ischemic stroke detected in vivo by PET and [11C]PK1195. Neurology 55:1052–1054
Perego C, Fumagalli S, Simoni M-GD (2011) Temporal pattern of expression and colocalization of microglia/macrophage phenotype markers following brain ischemic injury in mice. J Neuroinflammation 8:174. https://doi.org/10.1186/1742-2094-8-174
Rabenstein M, Hucklenbroich J, Willuweit A, et al (2015) Osteopontin mediates survival, proliferation and migration of neural stem cells through the chemokine receptor CXCR4. Stemcell Res Ther under revision
Rabenstein M, Vay SU, Flitsch LJ, Fink GR, Schroeter M, Rueger MA (2016) Osteopontin directly modulates cytokine expression of primary microglia and increases their survival. J Neuroimmunol 299:130–138. https://doi.org/10.1016/j.jneuroim.2016.09.009
Rogall R, Rabenstein M, Vay S et al (2018) Bioluminescence imaging visualizes osteopontin-induced neurogenesis and neuroblast migration in the mouse brain after stroke. Stem Cell Res Ther 9:182. https://doi.org/10.1186/s13287-018-0927-9
Rueger MA, Backes H, Walberer M et al (2010) Noninvasive imaging of endogenous neural stem cell mobilization in vivo using positron emission tomography. J Neurosci 30:6454–6460. https://doi.org/10.1523/JNEUROSCI.6092-09.2010
Rueger MA, Muesken S, Walberer M et al (2012) Effects of minocycline on endogenous neural stem cells after experimental stroke. Neuroscience 215:174–183. https://doi.org/10.1016/j.neuroscience.2012.04.036
Schroeter M, Jander S, Witte OW, Stoll G (1999) Heterogeneity of the microglial response in photochemically induced focal ischemia of the rat cerebral cortex. Neuroscience 89:1367–1377
Schroeter M, Zickler P, Denhardt DT et al (2006) Increased thalamic neurodegeneration following ischaemic cortical stroke in osteopontin-deficient mice. Brain J Neurol 129:1426–1437. https://doi.org/10.1093/brain/awl094
Smirkin A, Matsumoto H, Takahashi H et al (2010) Iba1(+)/NG2(+) macrophage-like cells expressing a variety of neuroprotective factors ameliorate ischemic damage of the brain. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 30:603–615. https://doi.org/10.1038/jcbfm.2009.233
Stoll G, Jander S, Schroeter M (1998) Inflammation and glial responses in ischemic brain lesions. Prog Neurobiol 56:149–171
Streit WJ, Kreutzberg GW (1987) Lectin binding by resting and reactive microglia. J Neurocytol 16:249–260
Vay SU, Flitsch LJ, Rabenstein M et al (2018) The plasticity of primary microglia and their multifaceted effects on endogenous neural stem cells in vitro and in vivo. J Neuroinflammation 15:226. https://doi.org/10.1186/s12974-018-1261-y
Villa P, van Beek J, Larsen AK et al (2007) Reduced functional deficits, neuroinflammation, and secondary tissue damage after treatment of stroke by nonerythropoietic erythropoietin derivatives. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab 27:552–563. https://doi.org/10.1038/sj.jcbfm.9600370
Walberer M, Rueger MA, Simard M-L et al (2010) Dynamics of neuroinflammation in the macrosphere model of arterio-arterial embolic focal ischemia: an approximation to human stroke patterns. Exp Transl Stroke Med 2:22. https://doi.org/10.1186/2040-7378-2-22
Wang KX, Denhardt DT (2008) Osteopontin: role in immune regulation and stress responses. Cytokine Growth Factor Rev 19:333–345. https://doi.org/10.1016/j.cytogfr.2008.08.001
Wang X, Louden C, Yue TL et al (1998) Delayed expression of osteopontin after focal stroke in the rat. J Neurosci 18:2075–2083
Xia C-Y, Zhang S, Gao Y et al (2015) Selective modulation of microglia polarization to M2 phenotype for stroke treatment. Int Immunopharmacol 25:377–382. https://doi.org/10.1016/j.intimp.2015.02.019
Xiong X-Y, Liu L, Yang Q-W (2016) Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke. Prog Neurobiol 142:23–44. https://doi.org/10.1016/j.pneurobio.2016.05.001
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in the studies involving animals were in accordance with the ethical standards of the institution at which the studies were conducted.
Appendix
Appendix
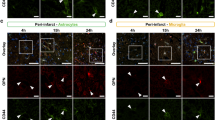
Markers of microglial activation are attenuated by OPN treatment. (a) Isolectin B4 (IB4) and Iba1 double-immunofluorescence staining show attenuated microglial activation in the ipsilateral thalamus of OPN-treated animals. (b) Magnified images of IB4/Iba1 staining showing several double positive cells (white arrow heads) in an exemplary placebo-treated animal compared to an OPN-treated animal with sparse overlap. (c) Double immunofluorescence staining of MHC class II/ Ox6 and Iba1 delineating a distinct population of microglia with upregulated MHC II receptors in the ipsilateral thalamus after stroke. Quantification showed a trend towards decreased upregulation in OPN-treated animals. White arrow heads indicate double labeled cells. Scale bar is 100 μm in a, b and close ups in c (lower row) and 500 μm in 4x pictures of entire thalamus in c (upper row). Bar graphs represent mean ± s.e.m. * p < 0.05
Rights and permissions
About this article
Cite this article
Ladwig, A., Rogall, R., Hucklenbroich, J. et al. Osteopontin Attenuates Secondary Neurodegeneration in the Thalamus after Experimental Stroke. J Neuroimmune Pharmacol 14, 295–311 (2019). https://doi.org/10.1007/s11481-018-9826-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11481-018-9826-1