Abstract
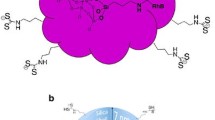
Gold@silica core–shell nanoparticles were prepared with various gold core diameters (ranging from 20 to 150 nm) and silica thicknesses (ranging from 10 to 30 nm). When the gold diameter is increased, the size dispersion became larger, leading to a broader plasmon band. Then, silicon carbide (SiC) nanoparticles were covalently immobilized onto silica to obtain hybrid (Au@SiO2) SiC nanoparticles. The absorption properties of these hybrid nanoparticles showed that an excess of SiC nanoparticles in the dispersion can be identified by a strong absorption in the UV region. Compared to SiC reference samples, a blue shift of the fluorescence emission, from 582 to 523 nm, was observed, which was previously attributed to the strong surface modification of SiC when immobilized onto silica. Finally, the influence of several elaboration parameters (gold diameter, silica thickness, SiC concentration) on fluorescence enhancement was investigated. It showed that the highest enhancements were obtained with 10 nm silica thickness, low concentration of SiC nanoparticles, and surprisingly, with a 20-nm gold core diameter. This last result could be attributed to the broad plasmon band of big gold colloids. In this case, SiC emission strongly overlapped gold absorption, leading to possible quenching of SiC fluorescence by energy transfer.






Similar content being viewed by others
References
Lakowicz JR, Geddes CD (2005) Radiative decay engineering. Vol. 8—topics in fluorescence spectroscopy. Springer, New York
Lakowicz JR (2006) Plasmonics in biology and plasmon-controlled fluorescence. Plasmonics 1:5–33
Lakowicz JR (2001) Radiative decay engineering: biophysical and biomedical applications. Anal Biochem 298:1–24
Bharadwaj P, Novotny L (2007) Spectral dependence of single molecule fluorescence enhancement. Opt Express 15:14266–14274
Tovmachenko OG et al (2006) Fluorescence enhancement by metal-core/silica-shell nanoparticles. Adv Mater 18:91–95
Liu N et al (2006) Hybrid gold/silica/nanocrystal-quantum-dot superstructures: synthesis and analysis of semiconductor–metal interactions. J Am Chem Soc 128:15362–15363
Zhang F et al (2010) Fabrication of Ag@SiO2@Y2O3:Er nanostructures for bioimaging: tuning of the upconversion fluorescence with silver nanoparticles. J Am Chem Soc 132:2850–2851
Jin Y, Gao X (2009) Plasmonic fluorescent quantum dots. Nat Nanotechnol 4:571–576
Zhang J et al (2007) Metal-enhanced single-molecule fluorescence on silver particle monomer and dimer: coupling effect between metal particles. Nano Lett 7:2101–2107
Li C et al (2012) Metal-enhanced fluorescence of carbon dots adsorbed Ag@SiO2 core-shell nanoparticles. RSC Adv 2:1765–1768
Nychyporuk T et al (2011) Strong photoluminescence enhancement of silicon quantum dots by their near-resonant coupling with multi-polar plasmonic hot spots. Nanoscale 3:2472–2475
Sui N et al. (2012) Plasmon-controlled fluorescence in (Au@SiO2)SiC nanohybrids: enhanced, narrower and blue-shifted emission of SiC nanoparticles. J Nanopart Res 14:1004
Zhang Y et al (2011) Metal-enhanced photoluminescence from carbon nanodots. Chem Commun 47:5313–5315
Zakharko Y et al Plasmon-enhanced photoluminescence of SiC quantum dots for cell imaging applications. Plasmonics. doi:10.1007/s11468-012-9364-2
Frens G (1973) Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nature Phys Sci 241:20–22
Liz-Marzan LM et al (1996) Synthesis of nanosized gold-silica core-shell particles. Langmuir 12:4329–4335
Stöber W et al (1968) Controlled growth of monodisperse silica spheres in the micron size range. J Colloid Interface Sci 26:62–69
Botsoa J et al (2008) Application of 3C-SiC quantum dots for living cell imaging. Appl Phys Lett 92:173902
Westcott SL et al (1998) Formation and adsorption of clusters of gold nanoparticles onto functionalized silica nanoparticle surfaces. Langmuir 14:5396–5401
Patungwasa W, Hodak JH (2008) pH tunable morophology of the gold nanoparticles produced by citrate reduction. Mater Chem Phys 108:45–54
Zakharko Y et al (2010) Influence of the interfacial chemical environment on the luminescence of 3C-SiC nanoparticles. J Appl Phys 107:013503
Fan J et al (2008) 3C-SiC nanocrystals as fluorescent biological labels. Small 4:1058–1062
Maye MM et al (2010) Photoluminescence enhancement in CdSe/ZnS–DNA linked–Au nanoparticle heterodimers probed by single molecule spectroscopy. Chem Commun 46:6111–6113
Burikov S et al (2010) Raman and IR spectroscopy research on hydrogen bonding in water-ethanol systems. Mol Phys 108:2427–2436
Förster T (1965) Light and organic crystals: delocalized excitation and excitation transfer. Academic, New York
Botsoa J (2008) Synthèse de nanostructures de carbure de sillicium et étude de leurs propriétés optiques. PhD Dissertation, Institut National des Sciences Appliquées de Lyon
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sui, N., Monnier, V., Zakharko, Y. et al. Fluorescent (Au@SiO2)SiC Nanohybrids: Influence of Gold Nanoparticle Diameter and SiC Nanoparticle Surface Density. Plasmonics 8, 85–92 (2013). https://doi.org/10.1007/s11468-012-9425-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11468-012-9425-6