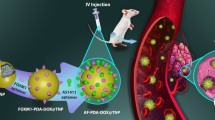
Abstract
In this study, an amphiphilic copolymer folate-succinyl-methionine-chitosan-octyl (FSMCO) was successfully synthesized step by step for self-assembling polymeric micelles. The copolymers formed micelle-like nanoparticles by their amphiphilic characteristics and structures were examined by UV-Vis absorption and Fourier transform spectroscopy. The sizes of blank and ICG derivative-loaded micelles measured by dynamic light scattering were about 170 and 140 nm, respectively, which were spherical in shape with an average zeta potential of −10 mV. Further studies on the stability showed that the micellar solutions maintain their sizes at room temperature for 1 month without distinct aggregation or dissociation. ICG derivative was much better photostable after being entrapped by the new carrier. The prepared FSMCO micelles displayed a good drug loading content (11.7%), entrapment efficiency (66.5%) and sustained release rate for the model drug fluorescein. The copolymers demonstrated weeny cytotoxicity toward Bel-7402, L02 and A549 cells when incubated for 2 d. Ligands modified micelles endowed preferable cell targeting capability and beautiful cell inhibition of HCPT-FSMCO on Bel-7402 tumor cells. This kind of polymeric micelles may be a promising nanovehicle in delivering near-infrared dyes for tumors imaging and chemotherapeutic drugs for cancer therapeutics.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Werner M E, Karve S, Sukumar R, et al. Folate-targeted nanoparticle delivery of chemo- and radiotherapeutics for the treatment of ovarian cancer peritoneal metastasis. Biomaterials, 2011, 32: 8548–8554
Barreto J A, Malley W O, Kubeil M, et al. Nanomaterials: Applications in cancer imaging and therapy. Adv Healthcare Mater, 2011, 23: 18–40
Maeda H, Bharate G Y, Daruwalla J. Polymeric drugs for efficient tumor-targeted drug delivery based on EPR-effect. Eng Polymers Control Drug Delivery Target, 2009, 71: 409–419
Low P S, Kularatne S A. Folate-targeted therapeutic and imaging agents for cancer. Curr Opin Chem Biol, 2009, 13: 256–262
Low P S, Henne W A, Doorneweerd D D. Discovery and development of folic-acid-based receptor targeting for imaging and therapy of cancer and inflammatory diseases. Acc Chem Res, 2008, 41: 120–129
Xia W, Low P S. Folate-targeted therapies for cancer. J Med Chem, 2010, 53: 6811–6824
Finkelstein J D. Methionine metabolism in mammals. J Nutr Biochem, 1990, 1: 228–237
Lin J, Raoof D A, Thomas D G, et al. L-type amino acid transporter-1 overexpression and melphalan sensitivity in Barrett’s adenocarcinoma. Neoplasia, 2004, 6: 74–84
Halpern B C, Clark B R, Hardy D N, et al. The effect of replacement of methionine by homocystine on survival of malignant and normal adult mammalian cells in culture. Proc Natl Acad Sci USA, 1974, 71: 1133–1136
Cavuoto P, Fenech M F. A review of methionine dependency and the role of methionine restriction in cancer growth control and life-span extension. Cancer Treatment Rev, 2012, 38: 726–736
Pavillard V, Nicolaou A, Double J A, et al. Methionine dependence of tumours: A biochemical strategy for optimizing paclitaxel chemosensitivity in vitro. Biochem Pharmacol, 2006, 71: 772–778
Maltzahn G V, Park J H, Lin K Y, et al. Nanoparticles that communicate in vivo to amplify tumour targeting. Nat Mater, 2011, 10: 545–551
Zhu H Y, Liu F, Guo J, et al. Folate-modified chitosan micelles with enhanced tumor targeting evaluated by near infrared imaging system. Carbohydrate Polymers, 2011, 86: 1118–1129
Wang X H, Tian Q, Wang W, et al. In vitro evaluation of polymeric micelles based on hydrophobically-modified sulfated chitosan as a carrier of doxorubicin. J Mater Sci Mater Med, 2012, 23: 1663–1674
Al Kobiasi M, Chua B Y, Tonkin D, et al. Control of size dispersity of chitosan biopolymer microparticles and nanoparticles to influence vaccine trafficking and cell uptake. J Biomed Mater Res Part A, 2012, 100A: 1859–1867
Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J Immunol Methods, 1983, 65: 55–63
Lian H, Sun J, Yu Y P, et al. Supramolecular micellar nanoaggregates based on a novel chitosan/vitamin E succinate copolymer for paclitaxel selective delivery. Inter J Nanomed, 2011, 6: 3323–3334
Luo Y L, Yao X J, Yuan J F, et al. Preparation and drug controlled-release of polyion complex micelles as drug delivery systems. Colloids and Surfaces B: Biointerfaces, 2009, 68: 218–224
Quinnones J P, Gothelf K V, Kjems J, et al. Self-assembled nanoparticles of glycol chitosan-Ergocalciferol succinate conjugate, for controlled release. Carbohydrate Polymers, 2012, 88: 1373–1377
Gaumet M, Vargas A, Gurny R, et al. Nanoparticles for drug delivery: The need for precision in reporting particle size parameters. Eur J Pharm Biopharm, 2008, 69: 1–9
Matsumura Y, Maeda H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res, 1986, 46: 6387–6392
Fan L, Li F, Zhang H T, et al. Co-delivery of PDTC and doxorubicin by multifunctional micellar nanoparticles to achieve active targeted drug delivery and overcome multidrug resistance. Biomaterials, 2010, 31: 5634–5642
Sun P J, Zhou D H, Gan Z H. Novel reduction-sensitive micelles for triggered intracellular drug release. J Contr Release, 2011, 155: 96–103
Chavanpatil M D, Khdair A, Panyam J. Nanoparticles for cellular drug delivery: Mechanisms and factors influencing delivery. J Nanosci Nanotechnol, 2006, 6: 2651–2663
Ozdemir V, Jones B W, Glatt S J, et al. Shifting emphasis from pharmacogenomics to theragnostics. Nat Biotechnol, 2006, 24: 942–946
Liang N, Sun S P, Li X F, et al. α-Tocopherol succinate-modified chitosan as a micellar delivery system for paclitaxel: Preparation, characterization and in vitro/in vivo evaluations. Inter J Pharmaceutics, 2012, 423: 480–488
Hu B, Ting Y W, Zeng X X, et al. Cellular uptake and cytotoxicity of chitosan-caseinophosphopeptides nanocomplexes loaded with epigallocatechin gallate. Carbohydrate Polymers, 2012, 89: 362–370
Chen H Y, Li B W, Ren X Y, et al. Multifunctional near-infrared-emitting nano-conjugates based on gold clusters for tumor imaging and therapy. Biomaterials, 2012, 33: 8461–8476
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Chen, Y., Cao, J., Zhu, H. et al. Synthesis and evaluation of methionine and folate co-decorated chitosan self-assembly polymeric micelles as a potential hydrophobic drug-delivery system. Chin. Sci. Bull. 58, 2379–2386 (2013). https://doi.org/10.1007/s11434-013-5733-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5733-2