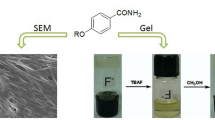
Abstract
The amidourea-based homoduplex was developed as a super organogelator, which could form stable gels in wide-tested solvents. And the reversible gel to solution transition was responsive to heat/cool and acid/base stimuli. The organogels were extensively investigated by 1H NMR, UV-visible absorption spectroscopy, fluorescence spectroscopy, scanning electron microscopy, transmission electron microscopy and powder X-ray diffraction. Based on these data, the gelation mechanism was rationally proposed. The hydrogen-bonded homoduplexes served as the basic assembling units, and further aggregated into three dimensional networks via π-π stacking and van der Waals interactions, which consequently led to the entangled fibers for the gel formation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Terech P, Weiss R G. Low molecular mass gelators of organic liquids and the properties of their gels. Chem Rev, 1997, 97: 3133–3159
Terech P, Weiss R G. Molecular Gels: Materials with Self-Assembled Fibrillar Networks. Dordrecht: Springer, 2006
Fages F. Low molecular mass gelators. Top Curr Chem, 2005, 256: 1–273
Steed J W. Supramolecular gel chemistry: Developments over the last decade. Chem Commun, 2011, 47: 1379–1383
de Jong J J D, Lucas L N, Kellogg R M, et al. Reversible optical transcription of supramolecular chirality into molecular chirality. Science, 2004, 304: 278–281
Ajayaghosh A, Praveen V K, Vijayakumar C, et al. Molecular wire encapsulated into π organogels: Efficient supramolecular light-harvesting antennae with color-tunable emission. Angew Chem Int Ed, 2007, 46: 6260–6265
Hirst A R, Escuder B, Miravet J F, et al. High-tech applications of self-assembling supramolecular nanostructured gel-phase materials: From regenerative medicine to electronic devices. Angew Chem Int Ed, 2008, 47: 8002–8018
Tao T, Assenmacher W, Peterlik H, et al. An air-stable organometallic low-molecular-mass gelator: Synthesis, aggregation, and catalytic application of a palladium pincer complex. Angew Chem Int Ed, 2007, 46: 6368–6371
George M, Weiss R G. Molecular organogels. Soft matter comprised of low-molecular-mass organic gelators and organic liquids. Acc Chem Res, 2006, 39: 489–497
Wang G T, Lin J B, Jiang X K, et al. Cholesterol-appended aromatic imine organogelators: A case study of gelation-driven component selection. Langmuir, 2009, 25: 8414–8418
Hu J, Zhang M, Ju Y. A simple oleanlic acid derivative as potent organogelator. Soft Matter, 2009, 5: 4971–4974
Lu J R, Hu J, Song Y, et al. A new dual-responsive organogel based on uracil-appended glycyrrhetinic acid. Org Lett, 2011, 13: 3372–3375
Suzuki M, Hanabusa K. L-Lysine-based low-molecular-weight gelators. Chem Soc Rev, 2009, 38: 967–975
Wang T Y, Li Y G, Liu M H. Gelation and self-assembly of glutamate bolaamphiphiles with hybrid linkers: Effect of the aromatic ring and alkyl spacers. Soft Matter, 2009, 5: 1066–1073
Tian Y, Zhang L, Duan P F, et al. Fabrication of organogels composed from carbon nanotubes through a supramolecular approach. New J Chem, 2010, 34: 2847–2852
Cai W, Wang G T, Xu Y X, et al. Vesicles and organogels from foldamers: A solvent-modulated self-assembling process. J Am Chem Soc, 2008, 130: 6936–6937
Cai W, Wang G T, Du P, et al. Foldamer organogels: A circular dichroism study of glucose-mediated dynamic helicity induction and amplification. J Am Chem Soc, 2008, 130: 13450–13459
Yang Y, Chen T, Xiang J F, et al. Mutual responsive hydrazide-based low-molecular-mass organic gelators: Probing gelation on the molecular level. Chem Eur J, 2008, 14: 5742–5746
Liu J W, Yang Y, Chen C F, et al. Novel anion-tuning supramolecular gels with dual-channel response: Reversible sol-gel transition and color changes. Langmuir, 2010, 26: 9040–9044
Liu J W, Ma J T, Chen C F. Structure-property relationship of a class of efficient organogelators and their multistimuli responsiveness. Tetrahedron, 2011, 67: 85–91
Wang C, Zhang D Q, Zhu D B. A low-molecular-mass gelator with an electroactive tetrathiafulvalene group: Tuning the gel formation by charge-transfer interaction and oxidation. J Am Chem Soc, 2005, 127: 16372–16373
Wang C, Zhang D Q, Xiang J F, et al. New organogels based on an anthracene derivative with one urea group and its photodimer: Fluorescence enhancement after gelation. Langmuir, 2007, 23: 9195–9200
Steed J W. Anion-tuned supramolecular gels: A natural evolution from urea supramolecular chemistry. Chem Soc Rev, 2010, 39: 3686–3699
Ajayaghosh A, Praveen V K. π-organogels of self-assembled p-phenylenevinylenes: Soft materials with distinct size, shape, and functions. Acc Chem Res, 2007, 40: 644–656
Ajayaghosh A, Praveen V K, Vijayakumar C. Organogels as scaffolds for excitation energy transfer and light harvesting. Chem Soc Rev, 2008, 37: 109–122
Wurthner F, Bauer C, Stepanenko V, et al. A black perylene bisimide super gelator with an unexpected J-type absorption band. Adv Mater, 2008, 20: 1695–1698
Prasanthkumar S, Saeki A, Seki S, et al. Solution phase epitaxial self-assembly and high charge-carrier mobility nanofibers of semiconducting molecular gelators. J Am Chem Soc, 2010, 132: 8866–8867
Piepenbrock M O M, Lloyd G O, Clarke N, et al. Metal- and anion-binding supramolecular gels. Chem Rev, 2010, 110: 1960–2004
Sobczuk A A, Tamaru S, Shinkai S. New strategy for controlling the oligothiophene aggregation mode utilizing the gel-to-sol phase transition induced by crown-alkali metal interactions. Chem Commun, 2011, 47: 3093–3095
Saez J A, Escuder B, Miravet J F. Selective catechol-triggered supramolecular gel disassembly. Chem Commun, 2010, 46: 7996–7998
Chen Q, Zhang D Q, Zhang G X, et al. New cholesterol-based gelators with maleimide unit and the relevant michael adducts: Chemoresponsive organogels. Langmuir, 2009, 25: 11436–11441
Lee D C, McGrath K K, Jang K. Nanofibers of asymmetrically substituted bisphenazine through organogelation and their acid sensing properties. Chem Commun, 2008, 44: 3636–3638
Li Y G, Liu K Q, Liu J, et al. Amino acid derivatives of cholesterol as “latent” organogelators with hydrogen chloride as a protonation reagent. Langmuir, 2006, 22: 7016–7020
Yang X Y, Zhang G X, Zhang D Q. Stimuli responsive gels based on low molecular weight gelators. J Mater Chem, 2012, 22: 38–50
Wang C, Chen Q, Sun F, et al. Multistimuli responsive organogels based on a new gelator featuring tetrathiafulvalene and azobenzene groups: reversible tuning of the gel-sol transition by redox reactions and light irradiation. J Am Chem Soc, 2010, 132: 3092–3096
Chen Q, Feng Y, Zhang D Q, et al. Light-triggered self-assembly of a spiropyran-functionalized dendron into nano-/micrometer-sized particles and photoresponsive organogel with switchable fluorescence. Adv Funct Mater, 2010, 20: 36–42
Chen Q, Zhang D Q, Zhang G X, et al. Multicolor tunable emission from organogels containing tetraphenylethene, perylenediimide, and spiropyran derivatives. Adv Funct Mater, 2010, 20: 3244–3251
Yoshio M, Shoji Y, Tochigi Y, et al. Electric field-assisted alignment of self-assembled fibers composed of hydrogen-bonded molecules having laterally fluorinated mesogens. J Am Chem Soc, 2009, 131: 6763–6767
Cravotto G, Cintas P. Molecular self-assembly and patterning induced by sound waves. The case of gelation. Chem Soc Rev, 2009, 38: 2684–2697
Li Y G, Wang T Y, Liu M H. Ultrasound induced formation of organogel from a glutamic dendron. Tetrahedron, 2007, 63: 7468–7473
Cao R K, Zhou J J, Wang W, et al. Oligoamide duplexes as organogelators. Org Lett, 2010, 12: 2958–2961
Chu W J, Yang Y, Chen C F. Multiple hydrogen-bond-mediated molecular duplexes based on the self-complementary amidourea motif. Org Lett, 2010, 12: 3156–3159
Hong Y N, Lam J W Y, Tang B Z. Aggregation-induced emission. Chem Soc Rev, 2011, 40: 5361–5388
Qu S N, Zhao L J, Yu Z X, et al. Nanoparticles, helical fibers, and nanoribbons of an achiral twin-tapered bi-1,3,4-oxadiazole derivative with strong fluorescence. Langmuir, 2009, 25: 1713–1717
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Chu, W., Chen, C. Amidourea-based homoduplex as a super organogelator. Chin. Sci. Bull. 57, 4278–4283 (2012). https://doi.org/10.1007/s11434-012-5440-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5440-4