Abstract
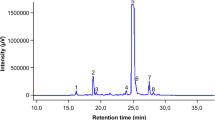
Mercury (II) is an important factor in hepatotoxicity that can enter the body through marine diets and amalgams. In the present study, the protective effect of the Eriobotrya japonica flower extract (EJFE) on HgCl2-induced hepatotoxicity was investigated. Five mg/kg of mercuric chloride in drinking water was given to rats either with saline or EJFE (100 and 200 mg/kg as intraperitoneal (IP)) for 30 d. The mercury levels in different groups of liver tissues of the rats were measured with flameless atomic absorption spectroscopy (F-AAS). Also, mercury accumulation in the liver of the rats was modeled by using a parallel chemical kinetic model. The results showed that HgCl2-induced oxidative damage led to a significant decrease in glutathione (GSH) and the total antioxidant capacity (TAC) levels, and to a significant increase in lipid peroxidation level. Accumulated mercury was 14.47% more in the livers of the stress groups than in those of the control groups (P<0.001), whereas the amount of Hg was adjusted to 13.49% and 13.93% in groups treated with 100 and 200 mg/kg of EJFE respectively, as compared with stress groups (P<0.001). HPLC analysis of EJFE revealed that hesperetin and gallic acid are the major antioxidants in EJFE. Results demonstrate that flowers of the Eriobotrya japonica cause a significant protection against HgCl2 induced hepatotoxicity in all diagnostic parameters by strengthening the antioxidant defense mechanisms and they may have a therapeutic function in free radical mediated diseases.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Langford N J, Ferner R E. Toxicity of mercury. J Hum Hypertens, 1999, 13: 651–656
Clarkson T W. The toxicology of mercury. Crit Rev Clin Lab Sci, 1997, 34: 369–403
Nath K A, Croatt A J, Likely S, et al. Renal oxidant injury and oxidant response induced by mercury. Kidney Int, 1996, 50: 1032–1043
Farina M, Soares F A, Zeni G, et al. Additive prooxidative effect of methylmercury and ebselen in liver from suckling rat pups. Toxicol Lett, 2004, 146: 227–235
Gstraunthaler G, Pfaller W, Kotanko P. Glutathione depletion and in vitro lipid peroxidation in mercury or maleate induced acute renal failure. Biochem Pharmacol, 1983, 32: 2969–2972
Zalups R K. Molecular interaction with mercury in the kidney. Pharmacol Rev, 2000, 52: 113–143
Stohs S J, Bagchi D. Oxidative mechanisms in the toxicity of metalions. Free Radic Biol Med, 1995, 18: 321–336
Huang Y L, Cheng S L, Lin T H. Lipid peroxidation in rats administrated with mercuric chloride. Biol Trace Elem Res, 1996, 52: 193–206
Girardi G, Elias M M. Mercuric chloride effects on rat renal redox enzymes activities: SOD protection. Free Radic Biol Med, 1995, 18: 61–66
Vaughan J G, Geissler C A. The New Oxford Book of Food Plants. New York: Oxford University Press, 1997. 110–111
de Tommasi N, Aquino R, de Simone F, et al. Plant metabolites, new sesquiterpene and ionone glycosides from Eryobotrya japonica. J Nat Prod, 1992, 55: 1025–1032
Ito H, Kobayashi E, Takamatsu Y, et al. Polyphenols from Eriobotrya japonica and their cytotoxicity against human oral tumor cell lines. Chem Pharm Bull, 2000, 48: 687–693
Ferreresa F, Gomesb D, Valentãob P, et al. Improved loquat (Eriobotrya japonica Lindl.) cultivars: Variation of phenolics and antioxidative potential. Food Chem, 2009, 114: 1019–1027
Peluso M R. Flavonoids attenuate cardiovascular disease, inhibit phosphodiesterase and modulate lipid homeostasis in adipose tissue and liver. Exp Biol Med, 2006, 231: 1287–1299
Hertog M C, Hollman P C H, Venema D P. Optimization of a quantitative HPLC determination of potentially anticarcinogenic flavonoids in vegetables and fruits. J Agric Food Chem, 1992, 40: 1591–1598
Sener G, Sehirli A O, Ayanoglu D, et al. Melatonin protects against mercury(II)-induced oxidative tissue damage in rats. Pharmacol Toxicol, 2003, 93: 290–296
Sener G, Sehirli O, Tozan A, et al. Ginkgo biloba extract protects against mercury(II)-induced oxidative tissue damage in rats. Food Chem Toxicol, 2007, 45: 543–550
Benzie I F, Strain J J. The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Anal Biochem, 1996, 239: 70–76
Wills E D. Mechanism of lipid peroxide formation in animal tissues. Biochem J, 1966, 99: 667–676
Draper H H, Hadley M. MDA determination as an index of lipid peroxidation. Methods Enzymol, 1990, 186: 421–430
Ellman G L. Tissue sulfhydryl groups. Arch Biochem Biophys, 1959, 82: 70–77
Teeny F M. Rapid method for the determination of mercury in fish tissue by atomic absorption spectroscopy. J Agric Food Chem, 1975, 23: 668–671
Zumkley H, Bertram H P, Lison A, et al. Al, Zn and Cu concentration in plasma and tissue in chronic renal insufficiency. Clin Nephrol, 1979, 12: 18–21
Li X, Zhang J Q, Liu W, et al. Serum levels of perfluorinated compounds in the general population in Shenzhen, China. Chin Sci Bull, 2011, 56: 3092–3099
Thompson J, Lorber M, Toms L M L, et al. Use of simple pharmacokinetic modeling to characterize exposure of Australians to perfluorooctanoic acid and perfluorooctane sulfonic acid. Environ Int, 2010, 36: 390–397
Hong Y, Lin S, Jiang Y, et al. Variation in contents of total phenolics, flavonoids and antioxidant activities in the leaves of 11 Eriobotrya species. Plant Food Hum Nutr, 2008, 63: 200–204
Goyer R A. Toxic Effects of Metals, In: Klaassen C D, Amdur M O, Doull J, eds. Casarett and Doull’s Toxicology. The Basic Science of Poisons. New York: McGraw-Hill Companies, 1996. 691–736
World Health Organization. Inorganic Mercury. Environmental Health Criteria 118. Geneva, 1991
Mahboob M, Shireen K F, Atkinson A, et al. Lipid peroxidation and oxidant enzyme activity in different organs of mice exposed to low level of mercury. J Environ Sci Health Part B, 2001, 36: 687–697
Alam M M, Javed K, Jafri M A. Effect of Rheum emodi (Revand Hindi) on renal functions in rats. J Ethnopharmacol, 2005, 96: 121–125
Alam M S, Kaur G, Jabbar Z, et al. Eruca sativa seeds possess antioxidant activity and exert a protective effect on mercuric chloride induced renal toxicity. Food Chem Toxicol, 2007, 45: 910–920
Van Acker S A, Van Den Berg D J, Tromp M N, et al. Structural aspects of antioxidant activity of flavanoids. Free Radic Biol Med, 1996, 20: 331–342
Ebrahimzadeh M A, Bahramian F. Antioxidant activity of Crataegus pentagina subsp. elbursis fruits extracts used in traditional medicine in Iran. Pak J Biol Sci, 2009, 12: 413–419
Yokota J, Takuma D, Hamada A, et al. Scavenging of ractive oxigen species (ROS) by Eriobotrya japonica seed extract. Biol Pharm Bull, 2006, 29: 467–471
Alia M, Horcajo C, Bravo L, et al. Effect of grape antioxidant dietary fiber on the total antioxidant capacity and the activity of liver antioxidant enzymes in rats. Nutr Res, 2003, 23: 1251–1267
Koba K, Matsuoka A, Osada K, et al. Effect of loquat (Eriobotrya japonica) extracts on LDL oxidation. Food Chem, 2007, 104: 308–316
Quig D. Cysteine metabolism and metal toxicity. Alter Med Rev, 1998, 3: 262–270
Sharma M K, Sharma A, Kumar A, et al. Spirulina fusifurmis provides protection against mercuric chloride induced oxidative stress in Swiss albino mice. Food Chem Toxicol, 2007, 45: 2412–2419
Huang Y, Li J, Cao Q, et al. Anti-oxidative effect of triterpene acids of Eriobotrya japonica (Thunb.) Lindl. Leaf in chronic bronchitis rats. Life Sci, 2006, 78: 2749–2757
Su L, Wang M, Yin S T, et al. The interaction of selenium and mercury in the accumulations and oxidative stress of rat tissues. Ecotox Environ Safe, 2008, 70: 483–489
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Esmaeili, A.H., Khavari-Nejad, R.A., Hajizadeh Moghaddam, A. et al. Effects of Eriobotrya japonica (Lindl.) flower extracts on mercuric chloride-induced hepatotoxicity in rats. Chin. Sci. Bull. 57, 3891–3897 (2012). https://doi.org/10.1007/s11434-012-5429-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-012-5429-z