Abstract
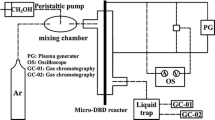
A novel corona inducing dielectric barrier discharge (CIDBD) and catalyst hybrid reactor was developed for reforming methane. This corona inducing technique allows dielectric barrier discharge (DBD) to occur uniformly in a large gap at relatively low applied voltage. Hydrogen production by reforming methane with steam and air was investigated with the hybrid reactor under atmospheric pressure and temperatures below 600°C. The effects of input power, O2/C molar ratio and preheat temperature on methane conversion and hydrogen selectivity were investigated experimentally. It was found that higher methane conversions were obtained at higher discharge power, and methane conversion increased significantly with input power less than 50 W; the optimized molar ratio of O2/C was 0.6 to obtain the highest hydrogen selectivity (112%); under the synergy of dielectric barrier discharge and catalyst, methane conversion was close to the thermodynamic equilibrium conversion rate.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cai J L, Wang G C, Li Y C, et al. Enrichment and hydrogen production by marine anaerobic hydrogen-producing microflora. Chinese Sci Bull, 2009, 54: 2656–2661
Jasinski M, Dors M, Mizeraczyk J. Production of hydrogen via methane reforming using atmospheric pressure microwave plasma. J Power Sources, 2008, 181: 41–45
Bromberg L, Cohn D R, Rabinovich A, et al. Plasma catalytic reforming of methane. Int J Hydrogen Energy, 1999, 24: 1131–1137
Indarto A. Hydrogen production from methane in a dielectric barrier discharge using oxide zinc and chromium as catalyst. J Chin Inst Chem Eng, 2008, 39: 23–28
Pietruszka B, Anklam K, Heintze M. Plasma-assisted partial oxidation of methane to synthesis gas in a dielectric barrier discharge. Appl Catal A: Gen, 2004, 261: 19–24
Hammer T, Kappes T, Baldauf M. Plasma catalytic hybrid processes: Gas discharge initiation and plasma activation of catalytic processes. Catal Today, 2004, 89: 5–14
Indarto A, Chio J W, Lee H, et al. The kinetic studies of direct methane oxidation to methanol in the plasma process. Chinese Sci Bull, 2008, 53: 2783–2792
Bromberg L, Cohn D R, Rabinovich A, et al. Emissions reductions using hydrogen from plasmatron fuel converters. Int J Hydrogen Energy, 2001, 26: 1115–1121
Petitpas G, Rollier J D, Darmon A, et al. A comparative study of non-thermal plasma assisted reforming technologies. Int J Hydrogen Energy, 2007, 32: 2848–2867
Cormier J M, Rusu I. Syngas production via methane steam reforming with oxygen: plasma reactors versus chemical reactors. J Phys D: Appl Phys, 2001, 34: 2798–2803
Zheng B, Yan J H, Li X D, et al. Plasma assisted dry methane reforming using gliding arc gas discharge: Effect of feed gases proportion. Int J Hydrogen Energy, 2008, 33: 5545–5553
Horng R F, Huang H H, Lai M P, et al. Characteristics of hydrogen production by a plasma-catalyst hybrid converter with energy saving schemes under atmospheric pressure. Int J Hydrogen Energy, 2008, 33: 3719–3727
Le H, Lobban L L, Mallinson R G. Some temperature effects on stability and carbon formation in low temperature ac plasma conversion of methane. Catal Today, 2004, 89: 15–20
Supat K, Chavadej S, Lobban L L, et al. Combined steam reforming and partial oxidation of methane to synthesis gas under electrical discharge. Ind Eng Chem Res, 2003, 42: 1654–1661
Bang C U, Hong Y C, Cho S C, et al. Methane augmented microwave plasma burner. IEEE Trans Plasma Sci, 2006, 34: 1751–1756
Nozaki T, Muto N, Kado S, et al. Dissociation of vibrationally excited methane on Ni catalyst Part 1 application to methane steam reforming. Catal Today, 2004, 89: 57–65
Wang B W, Zhang X, Liu Y W, et al. Conversion of CH4, steam and O2 to syngas and hydrocarbons via dielectric barrier discharge. J Nat Gas Chem, 2009, 18: 94–97
Nozaki T, Hiroyuki T, Okazaki K. Hydrogen Enrichment of Low-calorific fuels using barrier discharge enhanced Ni/Al2O3 bed reactor: Thermal and nonthermal effect of non-equilibrium plasma. Energ Fuel, 2006, 20: 339–345
Nozaki T, Fukui W, Okazaki K. Reaction enhancement mechanism of the nonthermal discharge and catalyst hybrid reaction for methane reforming. Energ Fuel, 2008, 22: 3600–3604
Yang Y. Methane conversion and reforming by nonthermal plasma on pins. Ind Eng Chem Res, 2002, 41: 5918–5926
Matin N S, Savadkoohi H A, Feizabadi S Y. Methane conversion to C2 hydrocarbons using dielectric-barrier discharge reactor: Effects of system variables. Plasma Chem Plasma Process, 2008, 28: 189–202
Thammanoon S, Piyaphon T, Sumaeth C. Partial oxidation of methane with air for synthesis gas production in a multistage gliding arc discharge system. Int J Hydrogen Energy, 2007, 32: 1067–1079
Raizer Y P. Gas Discharge Physics. Berlin: Springer-Verlag, 1991
Tang J, Duan Y X, Zhao W. Characterization and mechanism studies of dielectric barrier discharges generated at atmospheric pressure. Appl Phys Lett, 2010, 96: 191503
Lü J, Li Z H. Conversion of natural gas to C2 hydrocarbons via cold plasma technology. J Nat Gas Chem, 2010, 19: 375–379
Juurlink L B F, McCabe P R, Smith R R, et al. Eigenstate-resolved studies of gas-surface reactivity: CH4 (ν3) dissociation on Ni(100). Phys Rev Lett, 1999, 83: 868–871
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhou, Z., Zhang, J., Ye, T. et al. Hydrogen production by reforming methane in a corona inducing dielectric barrier discharge and catalyst hybrid reactor. Chin. Sci. Bull. 56, 2162–2166 (2011). https://doi.org/10.1007/s11434-011-4485-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4485-0