Abstract
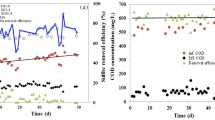
Sulfate reducing bacteria (SRB) is identified as the primary organisms responsible for the treatment of heavy metal wastewater. However, most heavy metals can inhibit the growth of SRB during heavy metal treatment processes. Sulfide is a metabolic product of SRB and it can precipitate or reduce heavy metals. This study focused on the effects of sulfide on SRB resistance to Cu(II), Hg(I) and Cr(VI) toxicity. First, we considered the existence style of various heavy metals with and without sulfide addition by sequential extraction experiments. Second, the particle size distribution was evaluated and the cell structure during the metabolism of a SRB culture, containing different heavy metals, was analyzed by particle size distribution and TEM analyses. Third, the evolution of sulfate under the influence of different concentrations of heavy metals with and without sulfide addition was investigated to evaluate SRB activity. The results indicated that sulfide played an important role in alleviating and even eliminating the toxicity of Cu(II), Hg(II) and Cr(VI). We also discuss the mechanism of sulfide on SRB resistance to Cu(II), Hg(I) and Cr(VI) toxicity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sampaio R M M, Timmers R A, Xu Y, et al. Selective precipitition of Cu from Zn in a pS controlled continuously stirred tank reactor. J Hazard Mater, 2009, 165: 256–265
Glombitza F. Treatment of acid lignite mine flooding water by means of microbial sulfate reduction. Waste Manage, 2001, 21: 197–203
Tabak H H, Scharp R, Burckle J, et al. Advances in biotreatment of acid mine drainage and biorecovery of metals: 1. Metal precipitation for recovery and recycle. Biodegradation, 2003, 14: 423–436
Foucher S, Battaglia-Brunet F, Ignatiadis I, et al. Treatment by sulfate-reducing bacteria of Chessy acid-mine drainage and metals recovery. Chem Eng Sci, 2001, 56: 1639–1645
Tatsunori N, Satoshi H, Akihiko M, et al. Distribution and diversity of thermophilic sulfate-reducing bacteria within a Cu-Pb-Zn mine (Toyoha, Japan). FEMS Microbiol Ecol, 2002, 41: 199–209
Sierra-Alvarez R, Karri S, Freeman S, et al. Biological treatment of heavy metals in acid mine drainage using sulfate reducing bioreactors. Water Sci Technol, 2006, 54: 179–185
Fang H H P, Xu L C, Chan K Y. Effects of toxic metals and chemicals on biofilm and biocorrosion. Water Res, 2002, 36: 4709–4716
Poulson S R, Colbery P J S, Drever J I. Toxicity of heavy metals(Ni, Zn) to Desulfovibrio desulfuricals. Geomicrobiol J, 1997, 14: 41–49
Olive J H, Li H G, Jin M C. Effects of metal additions on sulfate reduction activity in wastewaters. Toxicol Environ Chem, 1994, 46: 197–212
Maria T A, Carla C, Bo M. Precipitation of Zn(II), Cu(II) and Pb(II) at bench-scale using biogenic hydrogen sulfide from the utilization of volatile fatty acids. Chemosphere, 2007, 66: 1677–1683
Jiménez-Rodríguez A M, Durán-Barrantes M M, Borja R, et al. Heavy metals removal from acid mine drainage water using biogenic hydrogen sulphide and effluent from anaerobic treatment: Effect of pH. J Hazard Mater, 2009, 165: 759–765
Tebo B, Obraztsova A Y. Sulfate-reducing bacterium grows with Cr(VI), U(VI), Mn(IV) and Fe(III) as electron acceptors. FEMS Microbiol Lett, 1998, 162: 193–198
Cetin D, Dönmez S, Dönmez G. The treatment of textile wastewater including chromium (VI) and reactive dye by sulfate-reducing bacterial enrichment. J Environ Manage, 2008, 88: 76–82
Postgate J R. The Sulphate Reducing Bacteria. New York: Cambridge University Press, 1984
Tessier A, Campbell P G C, Bisson M. Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem, 1979, 51: 844–851
Chang I S, Groh J L, Ramsey M M, et al. Differential expression of desulfovibrio vulgaris genes in response to Cu(II) and Hg(II) toxicity. Appl Environ Microbiol, 2004, 70: 1847–1851
Tony J, David L P. Adsorption of Pb(II), Cu(II), Cd(II), Zn(II), Ni(II), Fe(II), and As(V) on bacterially produced metal sulfides. J Colloid Interface Sci, 2004, 275: 61–71
Ekstrom E B, Morel F M M. Cobalt limitation of growth and mercury methylation in sulfate reducing bacteria. Environ Sci Technol, 2008, 42: 93–99
Zhao X H, Rockne K J, Hurley R K, et al. Characterization of methyl mercury in dental wastewater and correlation with sulfate reducing bacterial DNA. Environ Sci Technol, 2008, 42: 2780–2786
Drott A, Lambertsson L, Bjorn E, et al. Importance of dissolved neutral mercury sulfides for methyl mercury production in contaminated sediments. Environ Sci Technol, 2007, 41: 2270–2276
Somasundaram V, Philip L, Murty Bhallamudi S. Experimental and mathematical modeling studies on Cr(VI) reduction by CRB, SRB and IRB. individually and incombination. J Hazard Mater, 2009, 172: 606–617
Guillen-Jimenez F D M, Morales-Barrera L, Morales-Jimenez J, et al. Modulation of tolerance to Cr(VI) and Cr(VI) reduction by sulfate ion in a Candida yeast strain isolated from tannery wastewater. J Ind Microbiol Biotechnol, 2008, 35: 1277–1287
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Sheng, Y., Cao, H., Li, Y. et al. Effects of sulfide on sulfate reducing bacteria in response to Cu(II), Hg(II) and Cr(VI) toxicity. Chin. Sci. Bull. 56, 862–868 (2011). https://doi.org/10.1007/s11434-011-4397-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4397-z