Abstract
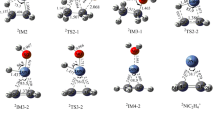
Density functional theory (DFT) study of reaction between vanadium trioxide cluster cation (VO +3 ) and ethylene (C2H4) to yield VO +2 + CH3CHO (acetaldehyde) and VO2CH +2 + HCHO (formaldehyde) is carried out. Structures of all reactants, products, intermediates, and transition state in the reaction have been optimized and characterized. The results show unexpected barriers in the reaction due to the existence of a η2-O2 moiety in the ground state structure of VO +3 . The initial reaction steps combining ethylene adsorption, C=C activation and O-O cleavage are proposed as rate limiting processes. Comparison of reactions of VO +3 + C2H4 with VO3 + C2H4 and VO +2 + C2H4 in the previous studies is made in detail. The results of this work may shed light on the understanding of C=C bond cleavage in related heterogeneous catalysis.
Similar content being viewed by others
References
Pimentel G C, Coonrod J A. Opportunities in Chemistry. Washington, DC: National Academy Press, 1985
Ogliaro F, Harris N, Cohen S, et al. A model “rebound” mechanism of hydroxylation by cytochrome P450: Stepwise and effectively concerted pathways and their reactivity patterns. J Am Chem Soc, 2000, 122: 8977–8989
Muetterties E L. Molecular Metal Clusters. Science, 1977, 196: 839
Bohme D K, Schwarz H. Gas-phase catalysis by atomic and cluster metal ions: The ultimate single-site catalysts. Angew Chem Int Ed, 2005, 44: 2336–2354
Weckhuysen B M, Keller D E. Chemistry, spectroscopy and the role of supported vanadium oxides in heterogeneous catalysis. Catal Today, 2003, 78: 25–46
Martinez-Huerta M V, Gao X, Tian H, et al. Oxidative dehydrogenation of ethane to ethylene over alumina-supported vanadium oxide catalysts: Relationship between molecular structures and chemical reactivity. Catal Today, 2006, 118: 279–287
Dunn J P, Stenger H G, Wachs I E. Oxidation of sulfur dioxide over supported vanadia catalysts: molecular structure-reactivity relationships and reaction kinetics. Catal Today, 1999, 51: 301–318
Harvey J N, Diefenbach J N, Schroder D, et al. Oxidation properties of the early transition-metal dioxide cations MO +2 (M = Ti, V, Zr, Nb) in the gas-phase. Int J Mass Spectrom., 1999, 182/183: 85–97
Bell R C, Castleman Jr A W. Reactions of Vanadium Oxide Cluster Ions with 1,3-Butadiene and Isomers of Butene. J Phys Chem A, 2002, 106: 9893–9899
Fielicke A, Rademann K. Stability and reactivity patterns of medium-sized vanadium oxide cluster cations VxO +y (4 ⩽ x ⩽ 14). Phys Chem Chem Phys, 2002, 4: 2621–2628
Zemski K A, Justes D R, Castleman A W. Studies of metal oxide clusters: elucidating reactive sites responsible for the activity of transition metal oxide catalysts. J Phys Chem B, 2002, 106: 6136–6148; and references there in.
Justes D R, Mitric R, Moore N A, et al. Theoretical and experimental consideration of the reactions between VxO +y and ethylene. J Am Chem Soc, 2003, 125: 6289–6299
Schröder D, Engeser M, Bronstrup M, et al. Ion chemistry of the hexanuclear methoxo-oxovanadium cluster V6O7(OCH3)12. Int J Mass Spectrom, 2003, 228: 743–757
Engeser M, Schlangen M, Schroder D, et al. Alkane oxidation by VO +2 in the gas phase: A unique dependence of reactivity on the chain length. Organometallics, 2003, 22: 3933–3943
Feyel S, Schroder D, Rozanska X, et al. Gas-phase oxidation of propane and 1-butene with [V3O7]+: Experiment and theory in concert. Angew Chem Int Ed, 2006, 45: 4677–4681
Feyel S, Dobler J, Schroder D, et al. Thermal activation of methane by tetranuclear [V4O10]+. Angew Chem Int Ed, 2006, 45: 4681–4685
Dong F, Heinbuch S, Xie Y, et al. Experimental and theoretical study of the reactions between neutral vanadium oxide clusters and ethane, ethylene, and acetylene. J Am Chem Soc, 2008, 130: 1932–1943
Calatayud M, Silvi B, Andrés J, et al. A theoretical study on the structure, energetics and bonding of VOx and VO +x (x = 1–4) systems. Chem Phys Lett, 2001, 333: 493–503
Calatayud M, Andrés J, Beltrán A. A Systematic density functional theory study of VxO +y and VxOy (x = 2–4, y = 2–10) Systems. J Phys Chem A, 2001, 105: 9760–9775
Gracia L, Sambrano J R, Safont V S, et al. Theoretical study on the molecular mechanism for the reaction of VOy2+ with C2H4. J Phys Chem A, 2003, 107: 3107–3120
Gracia L, Andrés J, Safont V S, et al. DFT Study of the reaction between VO +2 and C2H6. Organometallics, 2004, 23: 730–739
Gracia L, Sambrano J R, Andrés J, et al. Mechanistic insghts into the reaction between VO +2 and propene based on a DFT study. Organometallics, 2006, 25: 1643–1653
Vyboishchikov S F, Sauer J. (V2O5)n gas-phase clusters (n = 1–12) compared to V2O5 Crystal: DFT calculations. J Phys Chem A, 2001, 105: 8588–8598
Gracia L, Polo V, Sambrano J R, et al. Theoretical study on the reaction mechanism of VO +2 with propyne in gas phase. J Phys Chem A, 2008, 112: 1808–1816
Wang Z C, Xue W, Ma Y P, et al. Partial oxidation of propylene catalyzed by VO3 Clusters: A density functional theory study. J Phys Chem A, 2008, 112: 5984–5993
Ma Y P, Xue W, Wang Z C, et al. Acetylene cyclotrimerization catalyzed by TiO2 and VO2 in the gas phase: A DFT study. J Phys Chem A, 2008, 112: 3731–3741
Broclawik E, Haber J, Piskorz W. Molecular mechanism of C.H bond cleavage at transition metal oxide clusters. Chem Phys Lett, 2001, 333: 332–336
Gijzeman O L J, Lingen J N J, Lenthe J H V, et al. A new model for the molecular structure of supported vanadium oxide catalysts. Chem Phys Lett, 2004, 397: 277–281
Keller D E, de Groot F M F, Koningsberger D C, et al. VO4 Upside Down: A new molecular structure for supported VO4 Catalysts. J Phys Chem B, 2005, 109: 10223–10233
Frisch M J, Trucks G W, Schlegel H B, et al. Gaussian03 (Revision B.04); Pittsburgh, PA: Gaussian, Inc. 2003
Schlegel H B. Optimization of equilibrium geometries and transition structures. J Comput Chem, 1982, 3: 214–218
Peng C, Ayala P Y, Schlegel H B, et al. Using redundant internal coordinates to optimize equilibrium geometries and transition states. J Comput Chem, 1996, 17: 49–56
Berente I, Naray-Szabo G. Multicoordinate driven method for approximating enzymatic reaction paths: Automatic definition of the reaction coordinate using a subset of chemical coordinates. J Phys Chem A, 2006, 110: 772–778
Gonzalez C, Schlegel H B. An improved algorithm for reaction path following. J Chem Phys, 1989, 90: 2154–2161
Gonzalez C, Schlegel H B. Reaction path following in massweighted internal coordinates. J Phys Chem, 1990, 94: 5523–5527
Becke A D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A, 1988, 38: 3098–3100
Becke A D. Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys, 1993, 98: 5648–5652
Lee C, Yang W, Parr R G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B, 1988, 37: 785–789
Schafer A, Huber C, Ahlrichs R. Fully optimized contracted Gaussian basis sets of triple zeta valence quality for atoms Li to Kr. J Chem Phys, 1994, 100: 5829–5835
Boys S F, Bernardi F. The calculation of small molecular interactions by the differences of separate total energies: Some procedures with reduced errors. Mol Phys, 1970, 19: 553–566
Simon S, Duran M, Dannenberg J J. How does basis set superposition error change the potential surfaces for hydrogen-bonded dimers? J Chem Phys, 1996, 105: 11024–11031
Noodleman L. Valence bond description of antiferromagnetic coupling in transition metal dimmers. J Chem Phys, 1981, 74: 5737–5743
Caballo R, Castell O, Illas F, et al. Remarks on the proper use of the broken symmetry approach to magnetic coupling. J Phys Chem A, 1997, 101: 7860–7866
Ruszel M, Grzybowska B, Gasior M, et al. Effect of Au in V2O5/SiO2 and MoO3/SiO2 catalysts on physicochemical and catalytic properties in oxidation of C3 hydrocarbons and of CO. Catal Today, 2005, 99: 151–159
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Hundred Talents Fund, Chinese Academy of Sciences and National Natural Science Foundation of China (Grant No. 20703048)
About this article
Cite this article
Wang, Z., Ding, X., Ma, Y. et al. Theoretical study of partial oxidation of ethylene by vanadium trioxide cluster cation. Chin. Sci. Bull. 54, 2814–2821 (2009). https://doi.org/10.1007/s11434-009-0276-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-009-0276-2