Abstract
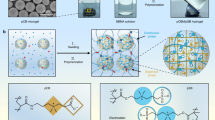
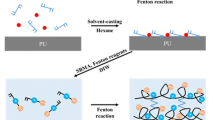
Zwitterions have aroused much interest to endow implantable medical devices with anti-fouling and anti-thrombosis performance, due to their ability to form a hydrated layer that can provide a good barrier against protein and cell adhesion. Herein, tyramine modified sulfobetaine-derived sodium hyaluronan (HST) hydrogel coating was fabricated, in which hyaluronan (HA) was used as polysaccharide skeleton to graft zwitterionic sulfobetaine, and tyramine was introduced as crosslinker to construct both the network of hydrogel and a strong covalent bond between coating and substrate. Hydrogel coating was prepared by spin coating or painting HST prepolymer solution under ultraviolet light irradiation. The obtained HST hydrogel coating shows good stability. Moreover, in addition to its outstanding anti-fouling performance and good biocompatibility, it can effectively prevent thrombosis in blood circulation ex vivo. This work offers a universal strategy to prepare a high-performance anti-fouling and anti-thrombosis coating, which is expected to promote the development of functional coatings for biomedical materials.
Similar content being viewed by others
References
Carpenter A W, Schoenfisch M H. Nitric oxide release: Part ii. Therapeutic applications. Chem Soc Rev, 2012, 41: 3742–3752
Jaffer I H, Fredenburgh J C, Hirsh J, et al. Medical device-induced thrombosis: What causes it and how can we prevent it? J Thromb Haemost, 2015, 13: S72–S81
Werner C, Maitz M F, Sperling C. Current strategies towards hemocompatible coatings. J Mater Chem, 2007, 17: 3376–3384
Al-Qahtani M, Safan A, Jassim G, et al. Efficacy of anti-microbial catheters in preventing catheter associated urinary tract infections in hospitalized patients: A review on recent updates. J Infect Public Health, 2019, 12: 760–766
Peng L, Chang L, Si M, et al. Hydrogel-coated dental device with adhesion-inhibiting and colony-suppressing properties. ACS Appl Mater Interfaces, 2020, 12: 9718–9725
Christensen K, Larsson R, Emanuelsson H, et al. Heparin coating of the stent graft—effects on platelets, coagulation and complement activation. Biomaterials, 2001, 22: 349–355
Lee S J, Jo H H, Lim K S, et al. Heparin coating on 3D printed poly(l-lactic acid) biodegradable cardiovascular stent via mild surface modification approach for coronary artery implantation. Chem Eng J, 2019, 378: 122116
Riedl C R, Witkowski M, Plas E, et al. Heparin coating reduces encrustation of ureteral stents: A preliminary report. Int J Antimicrob Agents, 2002, 19: 507–510
Goh S C, Luan Y, Wang X, et al. Polydopamine-polyethylene glycolalbumin antifouling coatings on multiple substrates. J Mater Chem B, 2018, 6: 940–949
Kim S, Gim T, Kang S M. Versatile, tannic acid-mediated surface pegylation for marine antifouling applications. ACS Appl Mater Interfaces, 2015, 7: 6412–6416
Yang C, Ding X, Ono R J, et al. Brush-like polycarbonates containing dopamine, cations, and PEG providing a broad-spectrum, antibacterial, and antifouling surface via one-step coating. Adv Mater, 2014, 26: 7346–7351
He M, Gao K, Zhou L, et al. Zwitterionic materials for antifouling membrane surface construction. Acta Biomater, 2016, 40: 142–152
Yao M, Sun H, Guo Z, et al. A starch-based zwitterionic hydrogel coating for blood-contacting devices with durability and bio-functionality. Chem Eng J, 2021, 421: 129702
Zhu Y, Wang J, Zhang F, et al. Zwitterionic nanohydrogel grafted pvdf membranes with comprehensive antifouling property and superior cycle stability for oil-in-water emulsion separation. Adv Funct Mater, 2018, 28: 1804121
Hoshi R A, Van Lith R, Jen M C, et al. The blood and vascular cell compatibility of heparin-modified eptfe vascular grafts. Biomaterials, 2013, 34: 30–41
Niimi Y, Ichinose F, Ishiguro Y, et al. The effects of heparin coating of oxygenator fibers on platelet adhesion and protein adsorption. Anesth Analg, 1999, 89: 573
Bannan S, Danby A, Cowan D, et al. Low heparinization with heparin-bonded bypass circuits: Is it a safe strategy? Ann Thoracic Surg, 1997, 63: 663–668
Conn G, Kidane A G, Punshon G, et al. Is there an alternative to systemic anticoagulation, as related to interventional biomedical devices? Expert Rev Med Devices, 2006, 3: 245–261
Rong Y, Zhang Z, He C L, et al. Matrix metalloproteinase-sensitive poly(ethylene glycol)/peptide hydrogels as an interactive platform conducive to cell proliferation during 3D cell culture. Sci China Tech Sci, 2021, 64: 1285–1294
Sigal G B, Mrksich M, Whitesides G M. Effect of surface wettability on the adsorption of proteins and detergents. J Am Chem Soc, 1998, 120: 3464–3473
Chen X, Lin Z, Feng Y, et al. Zwitterionic PMCP-modified polycaprolactone surface for tissue engineering: Antifouling, cell adhesion promotion, and osteogenic differentiation properties. Small, 2019, 15: 1903784
Goda T, Konno T, Takai M, et al. Biomimetic phosphorylcholine polymer grafting from polydimethylsiloxane surface using photo-induced polymerization. Biomaterials, 2006, 27: 5151–5160
Chen S, Zheng J, Li L, et al. Strong resistance of phosphorylcholine self-assembled monolayers to protein adsorption: Insights into non-fouling properties of zwitterionic materials. J Am Chem Soc, 2005, 127: 14473–14478
Iwasaki Y, Ishihara K. Cell membrane-inspired phospholipid polymers for developing medical devices with excellent biointerfaces. Sci Tech Adv Mater, 2012, 13: 064101
Lalani R, Liu L. Electrospun zwitterionic poly(sulfobetaine methacrylate) for nonadherent, superabsorbent, and antimicrobial wound dressing applications. Biomacromolecules, 2012, 13: 1853–1863
Jiang S, Cao Z. Ultralow-fouling, functionalizable, and hydrolyzable zwitterionic materials and their derivatives for biological applications. Adv Mater, 2010, 22: 920–932
Wen C, Guo H, Bai H, et al. Beetle-inspired hierarchical antibacterial interface for reliable fog harvesting. ACS Appl Mater Interfaces, 2019, 11: 34330–34337
Ukita R, Wu K, Lin X, et al. Zwitterionic poly-carboxybetaine coating reduces artificial lung thrombosis in sheep and rabbits. Acta Biomater, 2019, 92: 71–81
Yang L, Wu H, Liu Y, et al. A robust mussel-inspired zwitterionic coating on biodegradable poly(L-lactide) stent with enhanced anticoagulant, anti-inflammatory, and anti-hyperplasia properties. Chem Eng J, 2022, 427: 130910
Zhang L, Cao Z, Bai T, et al. Zwitterionic hydrogels implanted in mice resist the foreign-body reaction. Nat Biotechnol, 2013, 31: 553–556
Kang H, Jeong W, Hong D. Antifouling surface coating using droplet-based si-arget atrp of carboxybetaine under open-air conditions. Langmuir, 2019, 35: 7744–7750
Ma Q, Zhang H, Zhao J, et al. Fabrication of cell outer membrane mimetic polymer brush on polysulfone surface via raft technique. Appl Surf Sci, 2012, 258: 9711–9717
Huang P, Yang C, Liu J, et al. Improving the oral delivery efficiency of anticancer drugs by chitosan coated polycaprolactone-grafted hyaluronic acid nanoparticles. J Mater Chem B, 2014, 2: 4021–4033
Zhang E, Guo Q, Ji F, et al. Thermoresponsive polysaccharide-based composite hydrogel with antibacterial and healing-promoting activities for preventing recurrent adhesion after adhesiolysis. Acta Biomater, 2018, 74: 439–453
Dong D, Hao T, Wang C, et al. Zwitterionic starch-based hydrogel for the expansion and “sternness” maintenance of brown adipose derived stem cells. Biomaterials, 2018, 157: 149–160
Dong D, Li J, Cui M, et al. In situ “clickable” zwitterionic starch-based hydrogel for 3d cell encapsulation. ACS Appl Mater Interfaces, 2016, 8: 4442–4455
Applegate M B, Partlow B P, Coburn J, et al. Photocrosslinking of silk fibroin using riboflavin for ocular prostheses. Adv Mater, 2016, 28: 2417–2420
Peng W, Liu P, Zhang X, et al. Multi-functional zwitterionic coating for silicone-based biomedical devices. Chem Eng J, 2020, 398: 125663
Parada G, Yu Y, Riley W, et al. Ultrathin and robust hydrogel coatings on cardiovascular medical devices to mitigate thromboembolic and infectious complications. Adv Healthcare Mater, 2020, 9: 2001116
Yu Y, Yuk H, Parada G A, et al. Multifunctional “hydrogel skins” on diverse polymers with arbitrary shapes. Adv Mater, 2019, 31: 1807101
Han X X, Li L, Xie T, et al. “Ferrero-like” nanoparticles knotted injectable hydrogels to initially scavenge ROS and lastingly promote vascularization in infarcted hearts. Sci China Tech Sci, 2020, 63: 2435–2448
Bi B, Liu H, Kang W, et al. An injectable enzymatically crosslinked tyramine-modified carboxymethyl chitin hydrogel for biomedical applications. Colloids Surfs B-Biointerfaces, 2019, 175: 614–624
Raia N R, Partlow B P, McGill M, et al. Enzymatically crosslinked silk-hyaluronic acid hydrogels. Biomaterials, 2017, 131: 58–67
Wang J, Sun H, Li J, et al. Ionic starch-based hydrogels for the prevention of nonspecific protein adsorption. Carbohydrate Polyms, 2015, 117: 384–391
Kim K S, Park S J, Yang J A, et al. Injectable hyaluronic acid-tyramine hydrogels for the treatment of rheumatoid arthritis. Acta Biomater, 2011, 7: 666–674
Kurisawa M, Chung J E, Yang Y Y, et al. Injectable biodegradable hydrogels composed of hyaluronic acid-tyramine conjugates for drug delivery and tissue engineering. Chem Commun, 2005, 4312–4314
Fan Z W, Duan L J, Gao G H. Self-healing carrageenan-driven Polyacrylamide hydrogels for strain sensing. Sci China Tech Sci, 2020, 63: 2677–2686
Zhao R, Yang H, Nie B Q, et al. Highly transparent, antifreezing and stretchable conductive organohydrogels for strain and pressure sensors. Sci China Tech Sci, 2021, 64: 2532–2540
Zhang F, Hu C, Yang L, et al. A conformally adapted all-in-one hydrogel coating: Towards robust hemocompatibility and bactericidal activity. J Mater Chem B, 2021, 9: 2697–2708
Kim E, Kim M H, Song J H, et al. Dual crosslinked alginate hydrogels by riboflavin as photoinitiator. Int J Biol Macromolecules, 2020, 154: 989–998
Kobayashi M, Terayama Y, Yamaguchi H, et al. Wettability and antifouling behavior on the surfaces of superhydrophilic polymer brushes. Langmuir, 2012, 28: 7212–7222
Lv J, Zhang G, Zhang H, et al. Improvement of antifouling performances for modified PVDF ultrafiltration membrane with hydrophilic cellulose nanocrystal. Appl Surf Sci, 2018, 440: 1091–1100
Hu W, Zhang Z, Lu S, et al. Assembled anti-adhesion polypropylene mesh with self-fixable and degradable in situ mussel-inspired hydrogel coating for abdominal wall defect repair. Biomater Sci, 2018, 6: 3030–3041
Nanda H S, Shah A H, Wicaksono G, et al. Nonthrombogenic hydrogel coatings with carbene-cross-linking bioadhesives. Biomacromolecules, 2018, 19: 1425–1434
Gao F, Hu Y, Gong Z, et al. Fabrication of chitosan/heparinized graphene oxide multilayer coating to improve corrosion resistance and biocompatibility of magnesium alloys. Mater Sci Eng-C, 2019, 104: 109947
Gao F, Hu Y, Li G, et al. Layer-by-layer deposition of bioactive layers on magnesium alloy stent materials to improve corrosion resistance and biocompatibility. Bioactive Mater, 2020, 5: 611–623
Chang Y, Chang W J, Shih Y J, et al. Zwitterionic sulfobetaine-grafted poly(vinylidene fluoride) membrane with highly effective blood compatibility via atmospheric plasma-induced surface copolymerization. ACS Appl Mater Interfaces, 2011, 3: 1228–1237
Shih Y J, Chang Y. Tunable blood compatibility of polysulfobetaine from controllable molecular-weight dependence of zwitterionic non-fouling nature in aqueous solution. Langmuir, 2010, 26: 17286–17294
Kim M S, Ahn H H, Shin Y N, et al. An in vivo study of the host tissue response to subcutaneous implantation of PLGA- and/or porcine small intestinal submucosa-based scaffolds. Biomaterials, 2007, 28: 5137–5143
Morais J M, Papadimitrakopoulos F, Burgess D J. Biomaterials/tissue interactions: Possible solutions to overcome foreign body response. AAPS J, 2010, 12: 188–196
Hong J K, Gao L, Singh J, et al. Evaluating medical device and material thrombosis under flow: Current and emerging technologies. Biomater Sci, 2020, 8: 5824–5845
Chen S, Li L, Zhao C, et al. Surface hydration: Principles and applications toward low-fouling/nonfouling biomaterials. Polymer, 2010, 51: 5283–5293
Damodaran V B, Murthy N S. Bio-inspired strategies for designing antifouling biomaterials. Biomater Res, 2016, 20: 18
Andrews R K, Berndt M C. Platelet physiology and thrombosis. Thrombosis Res, 2004, 114: 447–453
Zimmerman G A, Weyrich A S. Signal-dependent protein synthesis by activated platelets. Arterioscler Thromb Vasc Biol, 2008, 28: s17–s24
Ruggeri Z M, Mendolicchio G L. Adhesion mechanisms in platelet function. Circ Res, 2007, 100: 1673–1685
Ruggeri Z M. Von Willebrand factor, platelets and endothelial cell interactions. J Thromb Haemost, 2003, 1: 1335–1342
Yu W, Koc J, Finlay J A, et al. Layer-by-layer constructed hyaluronic acid/chitosan multilayers as antifouling and fouling-release coatings. Biointerphases, 2019, 14: 051002
Gorbet M B, Sefton M V. Biomaterial-associated thrombosis: Roles of coagulation factors, complement, platelets and leukocytes. Biomaterials, 2004, 25: 5681–5703
Lavery K S, Rhodes C, Mcgraw A, et al. Anti-thrombotic technologies for medical devices. Adv Drug Deliver Rev, 2017, 112: 2–11
Gorbet M, Sperling C, Maitz M F, et al. The blood compatibility challenge. Part 3: Material associated activation of blood cascades and cells. Acta Biomater, 2019, 94: 25–32
L’Acqua C, Hod E. New perspectives on the thrombotic complications of haemolysis. Br J Haematol, 2015, 168: 175–185
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was supported by the National Key Research and Development Program of China (Grant No. 2018YFC1105500), the National Natural Science Foundation of China (Grant Nos. U20A20261, 31870948, 31971250, and 52073205), and the Natural Science Foundation of Tianjin (Grant Nos. 20JCYBJC00660 and 21JCYBJC00560).
Supporting Information
The supporting information is available online at https://tech.scichina.com and https://link.springer.com. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Guo, Z., Yao, M., Sun, H. et al. Tyramine-enhanced zwitterion hyaluronan hydrogel coating for anti-fouling and anti-thrombosis. Sci. China Technol. Sci. 65, 1828–1844 (2022). https://doi.org/10.1007/s11431-022-2048-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-022-2048-1