Abstract
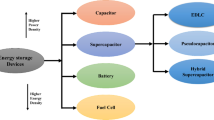
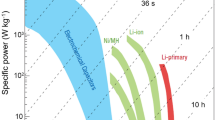
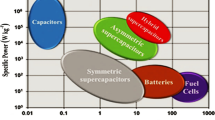
Both energy density and power density are crucial for a supercapacitor device, where the trade-off must be made between the two factors towards a practical application. Herein we focus on pseudocapacitance produced from the electrode and the electrolyte of supercapacitors to simultaneously achieve high energy density and power density. On the one hand, layered transition metal hydroxides (Ni(OH)2 and Co(OH)2) are introduced as electrodes, followed with exploration of the effect of the active materials and the substrate on the electrochemical behavior. On the other hand, various redox electrolytes are utilized to improve the specific capacitance of an electrolyte. The roadmap is to select an appropriate electrode and a dedicated electrolyte in order to achieve high electrochemical performance of the supercapacitors.
Similar content being viewed by others
References
Simon P, Gogotsi Y. Materials for electrochemical capacitors. Nat Mater, 2008, 7: 845–854
Wang G, Zhang L, Zhang J. A review of electrode materials for electrochemical supercapacitors. Chem Soc Rev, 2012, 41: 797–828
Service R F. Materials science-new ‘supercapacitor’ promises to pack more electrical punch. Science, 2006, 313: 902–902
Gogotsi Y, Simon P. True performance metrics in electrochemical energy storage. Science, 2011, 334: 917–918
Conway B E. Electrochemical Supercapacitors: Scientific Fundamentals and Technological Applications. Berlin: Springer, 1999
Thackeray M M, Wolverton C, Isaacs E D. Electrical energy storage for transportation—approaching the limits of, and going beyond, lithium-ion batteries. Energy Environ Sci, 2012, 5: 7854–7863
Becker H I. Low voltage electrolytic capacitor. US 2800616, 1957
Zhao M Q, Zhang Q, Huang J Q, et al. Towards high purity graphene/single-walled carbon nanotube hybrids with improved electrochemical capacitive performance. Carbon, 2013, 54: 403–411
Jiang J, Li Y, Liu J, et al. Recent advances in metal oxide-based electrode architecture design for electrochemical energy storage. Adv Mater, 2012, 24: 5166–5180
Zhong C, Deng Y, Hu W, et al. A review of electrolyte materials and compositions for electrochemical supercapacitors. Chem Soc Rev, 2015, doi: 10.1039/C5CS00303B
Zhang J, Zhao X S. On the configuration of supercapacitors for maximizing electrochemical performance. ChemSusChem, 2012, 5: 818–841
Tian W, Wang X, Zhi C, et al. Ni(OH)2 nanosheet @ Fe2O3 nanowire hybrid composite arrays for high-performance supercapacitor electrodes. Nano Energy, 2013, 2: 754–763
Salunkhe R R, Lin J, Malgras V, et al. Large-scale synthesis of coaxial carbon nanotube/Ni(OH)2 composites for asymmetric supercapacitor application. Nano Energy, 2015, 11: 211–218
Gao S, Sun Y, Lei F, et al. Ultrahigh energy density realized by a single-layer beta-Co(OH)2 all-solid-state asymmetric supercapacitor. Angew Chem Intern Ed, 2014, 53: 12789–12793
Cheng Y, Zhang H, Varanasi C V, et al. Improving the performance of cobalt–nickel hydroxide-based self-supporting electrodes for supercapacitors using accumulative approaches. Energy Environ Sci, 2013, 6: 3314–3321
Kulkarni S B, Jagadale A D, Kumbhar V S, et al. Potentiodynamic deposition of composition influenced Co1-x Nix LDHs thin film electrode for redox supercapacitors. Inter J Hydrogen Energy, 2013, 38: 4046–4053
Yu H, Wu J, Fan L, et al. An efficient redox-mediated organic electrolyte for high-energy supercapacitor. J Power Sources, 2014, 248: 1123–1126
Yu H, Wu J, Lin J, et al. A reversible redox strategy for swcnt-based supercapacitors using a high-performance electrolyte. ChemPhys Chem, 2013, 14: 394–399
Wu J, Yu H, Fan L, et al. A simple and high-effective electrolyte mediated with p-phenylenediamine for supercapacitor. J Mater Chem, 2012, 22: 19025–19030
Yu H, Fan L, Wu J, et al. Redox-active alkaline electrolyte for carbon-based supercapacitor with pseudocapacitive performance and excellent cyclability. RSC Adv, 2012, 2: 6736–6740
Yu H, Wu J, Fan L, et al. Application of a novel redox-active electrolyte in MnO2-based supercapacitors. Sci China Chem, 2012, 55: 1319–1324
Yu H, Wu J, Fan L, et al. A novel redox-mediated gel polymer electrolyte for high-performance supercapacitor. J Power Sources, 2012, 198: 402–407
Sun G, Li K, Sun C. Electrochemical performance of electrochemical capacitors using Cu(ii)-containing ionic liquid as the electrolyte. Micro Meso Mater, 2010, 128: 56–61
Yu H, Wu J, Fan L, et al. Improvement of the performance for quasisolid-state supercapacitor by using PVA–KOH–KI polymer gel electrolyte. Electrochim Acta, 2011, 56: 6881–6886
Senthilkumar S T, Selvan R K, Lee Y S, et al. Electric double layer capacitor and its improved specific capacitance using redox additive electrolyte. J Mater Chem A, 2013, 1: 1086–1095
Senthilkumar S T, Selvan R K, Ulaganathan M, et al. Fabrication of Bi2O3AC asymmetric supercapacitor with redox additive aqueous electrolyte and its improved electrochemical performances. Electrochim Acta, 2014, 115: 518–524
Chen H, Hu L, Chen M, et al. Nickel-cobalt layered double hydroxide nanosheets for high-performance supercapacitor electrode materials. Adv Func Mater, 2014, 24: 934–942
Jiang W, Yu D, Zhang Q, et al. Ternary hybrids of amorphous nickel hydroxide-carbon nanotube-conducting polymer for supercapacitors with high energy density, excellent rate capability, and long cycle life. Adv Func Mater, 2015, 25: 1063–1073
Zhao J, Chen J, Xu S, et al. Hierarchical NiMn layered double hydroxide/carbon nanotubes architecture with superb energy density for flexible supercapacitors. Adv Func Mater, 2014, 24: 2938–2946
Yang J, Yu C, Fan X, et al. 3D architecture materials made of NiCoAl-LDH nanoplates coupled with NiCo-carbonate hydroxide nanowires grown on flexible graphite paper for asymmetric supercapacitors. Adv Energy Mater, 2014, 4: 1400761
Gu C D, Ge X, Wang X L, et al. Cation–anion double hydrolysis derived layered single metal hydroxide superstructures for boosted supercapacitive energy storage. J Mater Chem A, 2015, 3: 14228–14238
Wang L, Wang D, Dong X Y, et al. Layered assembly of graphene oxide and Co–Al layered double hydroxide nanosheets as electrode materials for supercapacitors. Chem Comm, 2011, 47: 3556–3558
Vialat P, Mousty C, Taviot-Gueho C, et al. High-performing monometallic cobalt layered double hydroxide supercapacitor with defined local structure. Adv Func Mater, 2014, 24: 4831–4842
Della Noce R, Eugénio S, Silva T M, et al. Alpha-Co(OH)2/carbon nanofoam composite as electrochemical capacitor electrode operating at 2 V in aqueous medium. J Power Sources, 2015, 288: 234–242
Yan J, Wang Q, Wei T, et al. Recent advances in design and fabrication of electrochemical supercapacitors with high energy densities. Adv Energy Mater, 2014, 4: 1300816
Zhi M, Xiang C, Li J, et al. Nanostructured carbon-metal oxide composite electrodes for supercapacitors: A review. Nanoscale, 2013, 5: 72–88
Cheng J P, Zhang J, Liu F. Recent development of metal hydroxides as electrode material of electrochemical capacitors. RSC Adv, 2014, 4: 38893–38917
Augustyn V, Simon P, Dunn B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ Sci, 2014, 7: 1597–1614
Cao L, Xu F, Liang Y Y, et al. Preparation of the novel nanocomposite Co(OH)2/ultra-stable Y Zeolite and its application as a supercapacitor with high energy density advanced materials. Adv Mater, 2004, 16: 1853–1857
Cao L, Kong L B, Liang Y Y, et al. Preparation of novel nano-composite Ni(OH)2/usy material and its application for electrochemical capacitance storage. Chem Comm, 2004, 14: 1646–1647
Liang Y Y, Bao S J, Li H L. Nanocrystalline nickel cobalt hydroxides/ultrastable Y zeolite composite for electrochemical capacitors. J Solid State Electrochem, 2006, 11: 571–576
Yan J, Fan Z, Sun W, et al. Advanced asymmetric supercapacitors based on Ni(OH)2/graphene and porous graphene electrodes with high energy density. Adv Func Mater, 2012, 22: 2632–2641
Xiong X, Ding D, Chen D, et al. Three-dimensional ultrathin Ni-(OH)2 nanosheets grown on nickel foam for high-performance supercapacitors. Nano Energy, 2015, 11: 154–161
Lu Z, Chang Z, Zhu W, et al. Beta-phased Ni(OH)2 nanowall film with reversible capacitance higher than theoretical faradic capacitance. Chem Comm, 2011, 47: 9651–9653
Wang H L, Casalongue H S, Liang Y Y, et al. Ni(OH)2 nanoplates grown on graphene as advanced electrochemical pseudocapacitor materials. J Am Chem Soc, 2010, 132: 7472–7477
Wang X, Wang Y, Zhao C, et al. Electrodeposited Ni(OH)2 nanoflakes on graphite nanosheets prepared by plasma-enhanced chemical vapor deposition for supercapacitor electrode. New J Chem, 2012, 36: 1902–1906
Wang X, Liu J, Wang Y, et al. Ni(OH)2 nanoflakes electrodeposited on Ni foam-supported vertically oriented graphene nanosheets for application in asymmetric supercapacitors. Mater Res Bull, 2014, 52: 89–95
Wang Z, Wang X, Zhao Y X, et al. MnOx/Ni(OH)2 nanocomposite materials for high-performance electrochemical capacitor application. J Nano Res, 2012, 20: 53–60
Dong X, Wang L, Wang D, et al. Layer-by-layer engineered Co-Al hydroxide nanosheets/graphene multilayer films as flexible electrode for supercapacitor. Langmuir, 2012, 28: 293–298
Wang L, Dong Z H, Wang Z G, et al. Layered α-Co(OH)2 nanocones as electrode materials for pseudocapacitors: Understanding the effect of interlayer space on electrochemical activity. Adv Func Mater, 2013, 23: 2758–2764
Wang L, Lin C, Zhang F X, et al. Phase transformation guided singlelayer beta-Co(OH)2 nanosheets for pseudocapacitive electrodes. ACS Nano, 2014, 8: 3724–3734
Hercule K M, Wei Q, Khan A M, et al. Synergistic effect of hierarchical nanostructured MoO2/Co(OH)2 with largely enhanced pseudocapacitor cyclability. Nano Letters, 2013, 13: 5685–5691
Zhao C, Wang X, Wang S, et al. Synthesis of Co(OH)2/graphene/Ni foam nano-electrodes with excellent pseudocapacitive behavior and high cycling stability for supercapacitors. Int J Hydrogen Energy, 2012, 37: 11846–11852
Choi B G, Yang M, Jung S C, et al. Enhanced pseudocapacitance of ionic liquid/cobalt hydroxide nanohybrids. ACS Nano, 2013, 7: 2453–2460
Pan G X, Xia X, Cao F, et al. Porous Co(OH)2/Ni composite nanoflake array for high performance supercapacitors. Electrochim Acta, 2012, 63: 335–340
Tai Dam D, Lee J M. Ultrahigh pseudocapacitance of mesoporous nidoped Co(OH)2/ITO nanowires. Nano Energy, 2013, 2: 1186–1196
Liu X M, Zhang Y H, Zhang X G, et al. Studies on Me/Al-layered double hydroxides (Me=Ni and Co) as electrode materials for electrochemical capacitors. Electrochim Acta, 2004, 49: 3137–3141
Gupta V, Gupta S, Miura N. Potentiostatically deposited nanostructured CoxNi1-x layered double hydroxides as electrode materials for redox-supercapacitors. J Power Sources, 2008, 175: 680–685
Hu Z A, Xie Y L, Wang Y X, et al. Synthesis and electrochemical characterization of mesoporous CoxNi1-x layered double hydroxides as electrode materials for supercapacitors. Electrochim Acta, 2009, 54: 2737–2741
Sebastian M, Nethravathi C, Rajamathi M. Interstratified hybrids of α-hydroxides of nickel and cobalt as supercapacitor electrode materials. Mater Res Bull, 2013, 48: 2715–2719
Liu X, Huang J, Wei X, et al. Preparation and electrochemical performances of nanostructured CoxNi1-x(OH)2 composites for supercapacitors. J Power Sources, 2013, 240: 338–343
Roldán S, Granda M, Menéndez R, et al. Mechanisms of energy storage in carbon-based supercapacitors modified with a quinoid redoxactive electrolyte. J Phys Chem C, 2011, 115: 17606–17611
Roldan S, Blanco C, Granda M, et al. Towards a further generation of high-energy carbon-based capacitors by using redox-active electrolytes. Angew Chem Intern Ed, 2011, 50: 1699–1701
Roldán S, González Z, Blanco C, et al. Redox-active electrolyte for carbon nanotube-based electric double layer capacitors. Electrochim Acta, 2011, 56: 3401–3405
Roldán S, Granda M, Menéndez R, et al. Supercapacitor modified with methylene blue as redox active electrolyte. Electrochim Acta, 2012, 83: 241–246
Lota G, Milczarek G. The effect of lignosulfonates as electrolyte additives on the electrochemical performance of supercapacitors. Electrochem Comm, 2011, 13: 470–473
Zhou J, Yin Y, Mansour A N, et al. Experimental studies of mediatorenhanced polymer electrolyte supercapacitors. Electrochem Solid-State Lett, 2011, 14: A25–A28
Li Q, Li K, Sun C, et al. An investigation of Cu2+ and Fe2+ ions as active materials for electrochemical redox supercapacitors. J Electroanal Chem, 2007, 611: 43–50
Su L H, Zhang X G, Mi C H, et al. Improvement of the capacitive performances for Co–Al layered double hydroxide by adding hexacyanoferrate into the electrolyte. Phys Chem Chem Phys, 2009, 11: 2195–2202
Tian Y, Yan J, Xue R, et al. Capacitive properties of activated carbon in K4Fe(CN)6. J Electrochem Soc, 2011, 158: A818–A821
Zhao C, Zheng W, Wang X, et al. Ultrahigh capacitive performance from both Co(OH)2/graphene electrode and K3Fe(CN)6 electrolyte. Sci Rep, 2013, 3: 2986
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, H., Zhang, W., Chen, H. et al. Towards unlocking high-performance of supercapacitors: From layered transition-metal hydroxide electrode to redox electrolyte. Sci. China Technol. Sci. 58, 1779–1798 (2015). https://doi.org/10.1007/s11431-015-5930-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-015-5930-0