Abstract
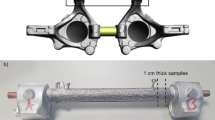
‘Compound casting’ simplifies joining processes by directly casting a metallic melt onto a solid metal substrate. A continuously metallurgic transition is very important for industrial applications, such as joint structures of spaceframe constructions in transport industry. In this project, ‘compound casting’ of light metals is investigated, aiming at weight-saving. The substrate used is a wrought aluminium alloy of type AA5xxx, containing magnesium as main alloying element. The melts are aluminium alloys, containing various alloying elements (Cu, Si, Zn), and magnesium. By replacing the natural oxygen layer with a zinc layer, the inherent wetting difficulties were avoided, and compounds with flawless interfaces were successfully produced (no contraction defects, cracks or oxides). Electron microscopy and EDX investigations as well as optical micrographs of the interfacial areas revealed their continuously metallic constitution. Diffusion of alloying elements leads to heat-treatable microstructures in the vicinity of the joining interfaces in Al-Al couples. This permits significant variability of mechanical properties. Without significantly cutting down on wettability, the formation of low-melting intermetallic phases (Al3Mg2 and Al12Mg17 IMPs) at the interface of Al-Mg couples was avoided by applying a protective coating to the substrate.
Similar content being viewed by others
References
Volder J P. An overview of a number of casting processes. Aluminium 1993, 4: 11–16
Pearson T, Wake S J. Improvements in the pretreatment of aluminium as a substrate for electrodeposition. Trans Inst Met Finish, 1997, 75: 93–97
Saito M, Maegawa T, Homma T. Electrochemical analysis of zincate treatments for Al and Al alloy films. Electrochim Acta, 2005, 51: 1017–1020
Qi G, Chen X, Shao Z. Influence of bath chemistry on zincate morphology on aluminium bond pad. Thin Solid Films, 2002, 406: 204–209
Tang Y C, Davenport A J. The effect of heat treatment and surface roughness on the zincate treatment of aluminium alloy 6082. Trans Inst Met Finish, 2001, 79: 85–89
Robertson S G, Ritchie I M, Druskovich D M. A kinetic and electrochemical study of the zincate immersion process for aluminium. J Appl Electrochem, 1995, 25: 659–666
Monteiro F J, Barbosa M A, Ross D H, et al. Pretreatments to improve the electrodeposits on aluminium. Surf Interface Anal, 1991, 17: 519–528
Zipperian D C, Raghavan S, Pritzker M D. Physical and chemical characteristics of the zincate immersion process for aluminium and aluminium alloys. J Electrochem Soc, 1987, 8B: C437–C438
Chen P-Y, Hussey C L. The electrodeposition of Mn and Zn-Mn alloys from the room-temperature tri-1-butylmethylammonium bis ((trifluoromethane)sulfonyl)imide ionic liquid. Electrochim Acta, 2007, 5: 1857–1864
Davis J R, ed. ASM Speciality Handbook—Aluminium and Aluminium Alloys. Materials Park, OH: ASM International, Materials Park, OH 440730002, USA, 1993
Andersson J O, Helander T, Hoglund L, et al. Thermo-Calc & DICTRA, computational tools for materials science. Calphad, 2002, 26: 273–312
Ansara I, Dinsdale A T, Rand MH, eds. Thermochemical Database for Light Metal Alloys. COST 507 Final Report Round 2, Vol. 2, Luxembourg, 1998
Prikhodovsky A. PhD Thesis, Aachen: RWTH Aachen University, 2000
Polmear I J. Light Alloys—Metallurgy of the Light Metals. Honeycombe R, Hancock P, eds. London: Arnold, 1995
Murayama M, Hono K. Pre-precipitate clusters and precipitation processes in Al-Mg-Si alloys. Acta Mater, 1999, 47: 1537–1548
Ronnhult T, Rilby U, Olefjord I. The surface state and weldability of aluminium alloys. Mater Sci Eng, 1980, 42: 329–336
Borrisutthekul R, Miyashita Y, Mutoh Y. Dissimilar material laser welding between magnesium alloy AZ31B and aluminium alloy A5052-O. Sci Techn Adv Mater, 2005, 6: 199–204
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papis, K.J.M., Loeffler, J.F. & Uggowitzer, P.J. Light metal compound casting. Sci. China Ser. E-Technol. Sci. 52, 46–51 (2009). https://doi.org/10.1007/s11431-008-0345-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11431-008-0345-9