Abstract
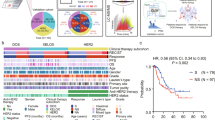
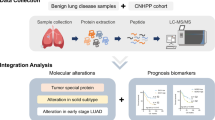
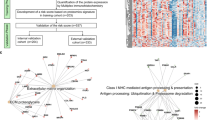
While precision medicine driven by genome sequencing has revolutionized cancer care, such as lung cancer, its impact on gastric cancer (GC) has been minimal. GC patients are routinely treated with chemotherapy, but only a fraction of them receive the clinical benefit. There is an urgent need to develop biomarkers or algorithms to select chemo-sensitive patients or apply targeted therapy. Here, we carried out retrospective analyses of 1,020 formalin-fixed, paraffin-embedded GC surgical resection samples from 5 hospitals and developed a mass spectrometry-based workflow for proteomic subtyping of GC. We identified two proteomic subtypes: the chemo-sensitive group (CSG) and the chemo-insensitive group (CIG) in the discovery set. The 5-year overall survival of CSG was significantly improved in patients who had received adjuvant chemotherapy after surgery compared with those who received surgery only (64.2% vs. 49.6%; Cox P-value=0.002), whereas no such improvement was observed in CIG (50.0% vs. 58.6%; Cox P-value=0.495). We validated these results in an independent validation set. Further, differential proteome analysis uncovered 9 FDA-approved drugs that may be applicable for targeted therapy of GC. A prospective study is warranted to test these findings for future GC patient care.
Similar content being viewed by others
References
Azmi, A.S., Uddin, M.H., and Mohammad, R.M. (2021). The nuclear export protein XPO1—from biology to targeted therapy. Nat Rev Clin Oncol 18, 152–169.
Bang, Y.J., Van Cutsem, E., Feyereislova, A., Chung, H.C., Shen, L., Sawaki, A., Lordick, F., Ohtsu, A., Omuro, Y., Satoh, T., et al. (2010). Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376, 687–697.
Bang, Y.J., Kim, Y.W., Yang, H.K., Chung, H.C., Park, Y.K., Lee, K.H., Lee, K.W., Kim, Y.H., Noh, S.I., Cho, J.Y., et al. (2012). Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): a phase 3 open-label, randomised controlled trial. Lancet 379, 315–321.
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R.L., Torre, L.A., and Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin 68, 394–424.
Burness, C.B., and Duggan, S.T. (2016). Trifluridine/tipiracil: A review in metastatic colorectal cancer. Drugs 76, 1393–1402.
Cancer Genome Atlas Research, N. (2014). Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513, 202–209.
Cardoso, F., van’t Veer, L.J., Bogaerts, J., Slaets, L., Viale, G., Delaloge, S., Pierga, J.Y., Brain, E., Causeret, S., DeLorenzi, M., et al. (2016). 70-Gene signature as an aid to treatment decisions in early-stage breast cancer. N Engl J Med 375, 717–729.
Chen, W., Zheng, R., Baade, P.D., Zhang, S., Zeng, H., Bray, F., Jemal, A., Yu, X.Q., and He, J. (2016). Cancer statistics in China, 2015. CA A Cancer J Clin 66, 115–132.
Cheong, J.H., Yang, H.K., Kim, H., Kim, W.H., Kim, Y.W., Kook, M.C., Park, Y.K., Kim, H.H., Lee, H.S., Lee, K.H., et al. (2018). Predictive test for chemotherapy response in resectable gastric cancer: a multi-cohort, retrospective analysis. Lancet Oncol 19, 629–638.
Collisson, E.A., Campbell, J.D., Brooks, A.N., Berger, A.H., Lee, W., Chmielecki, J., Beer, D.G., Cope, L., and Creighton, C.J. (2014). Comprehensive molecular profiling of lung adenocarcinoma. Nature 511, 543–550.
Coscia, F., Lengyel, E., Duraiswamy, J., Ashcroft, B., Bassani-Sternberg, M., Wierer, M., Johnson, A., Wroblewski, K., Montag, A., Yamada, S. D., et al. (2018). Multi-level proteomics identifies CT45 as a chemosensitivity mediator and immunotherapy target in ovarian cancer. Cell 175, 159–170.e16.
Ding, C., Jiang, J., Wei, J., Liu, W., Zhang, W., Liu, M., Fu, T., Lu, T., Song, L., Ying, W., et al. (2013). A fast workflow for identification and quantification of proteomes. Mol Cell Proteomics 12, 2370–2380.
Esserman, L.J., Yau, C., Thompson, C.K., van’t Veer, L.J., Borowsky, A.D., Hoadley, K.A., Tobin, N.P., Nordenskjöld, B., Fornander, T., Stål, O., et al. (2017). Use of molecular tools to identify patients with indolent breast cancers with ultralow risk over 2 decades. JAMA Oncol 3, 1503–1510.
Feng, J., Ding, C., Qiu, N., Ni, X., Zhan, D., Liu, W., Xia, X., Li, P., Lu, B., Zhao, Q., et al. (2017). Firmiana: towards a one-stop proteomic cloud platform for data processing and analysis. Nat Biotechnol 35, 409–412.
Feng, R.M., Zong, Y.N., Cao, S.M., and Xu, R.H. (2019). Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun 39, 22.
Hirsch, F.R., Scagliotti, G.V., Mulshine, J.L., Kwon, R., Curran Jr, W.J., Wu, Y.L., and Paz-Ares, L. (2017). Lung cancer: current therapies and new targeted treatments. Lancet 389, 299–311.
Hodson, R. (2016). Precision medicine. Nature 537, S49.
Huang, D., Lu, N., Fan, Q., Sheng, W., Bu, H., Jin, X., Li, G., Liu, Y., Li, X., Sun, W., et al. (2013). HER2 status in gastric and gastroesophageal junction cancer assessed by local and central laboratories: Chinese Results of the HER-EAGLE study PLoS ONE 8, e80290.
Imming, P., Sinning, C., and Meyer, A. (2006). Drugs, their targets and the nature and number of drug targets. Nat Rev Drug Discov 5, 821–834.
Ishizuka, J.J., Manguso, R.T., Cheruiyot, C.K., Bi, K., Panda, A., Iracheta-Vellve, A., Miller, B.C., Du, P.P., Yates, K.B., Dubrot, J., et al. (2019). Loss of ADAR1 in tumours overcomes resistance to immune checkpoint blockade. Nature 565, 43–48.
Kim, W.H., Gomez-Izquierdo, L., Vilardell, F., Chu, K.M., Soucy, G., Dos Santos, L.V., Monges, G., Viale, G., Brito, M.J., Osborne, S., et al. (2018). HER2 status in gastric and gastroesophageal junction cancer: results of the large, multinational HER-EAGLE study. Appl Immunohistochem Mol Morphol 26, 239–245.
Lei, Z., Tan, I.B., Das, K., Deng, N., Zouridis, H., Pattison, S., Chua, C., Feng, Z., Guan, Y.K., Ooi, C.H., et al. (2013). Identification of molecular subtypes of gastric cancer with different responses to PI3-kinase inhibitors and 5-fluorouracil. Gastroenterology 145, 554–565.
Liu, H., Golji, J., Brodeur, L.K., Chung, F.S., Chen, J.T., deBeaumont, R.S., Bullock, C.P., Jones, M.D., Kerr, G., Li, L., et al. (2019). Tumor-derived IFN triggers chronic pathway agonism and sensitivity to ADAR loss. Nat Med 25, 95–102.
Manzoni, C., Kia, D.A., Vandrovcova, J., Hardy, J., Wood, N.W., Lewis, P. A., and Ferrari, R. (2018). Genome, transcriptome and proteome: the rise of omics data and their integration in biomedical sciences. Brief Bioinf 19, 286–302.
Matsusaka, S., Nashimoto, A., Nishikawa, K., Miki, A., Miwa, H., Yamaguchi, K., Yoshikawa, T., Ochiai, A., Morita, S., Sano, T., et al. (2016). Clinicopathological factors associated with HER2 status in gastric cancer: results from a prospective multicenter observational cohort study in a Japanese population (JFMC44-1101). Gastric Cancer 19, 839–851.
Muzny, D.M., Bainbridge, M.N., Chang, K., Dinh, H.H., Drummond, J.A., Fowler, G., Kovar, C.L., Lewis, L.R., and Morgan, M.B. (2012). Comprehensive molecular characterization of human colon and rectal cancer. Nature 487, 330–337.
Noh, S.H., Park, S.R., Yang, H.K., Chung, H.C., Chung, I.J., Kim, S.W., Kim, H.H., Choi, J.H., Kim, H.K., Yu, W., et al. (2014). Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol 15, 1389–1396.
Oh, S.C., Sohn, B.H., Cheong, J.H., Kim, S.B., Lee, J.E., Park, K.C., Lee, S.H., Park, J.L., Park, Y.Y., Lee, H.S., et al. (2018). Clinical and genomic landscape of gastric cancer with a mesenchymal phenotype. Nat Commun 9, 1777.
Paik, S., Shak, S., Tang, G., Kim, C., Baker, J., Cronin, M., Baehner, F.L., Walker, M.G., Watson, D., Park, T., et al. (2004). A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med 351, 2817–2826.
Paik, S., Tang, G., Shak, S., Kim, C., Baker, J., Kim, W., Cronin, M., Baehner, F.L., Watson, D., Bryant, J., et al. (2006). Gene expression and benefit of chemotherapy in women with node-negative, estrogen receptor-positive breast cancer. J Clin Oncol 24, 3726–3734.
Park, S.H., Sohn, T.S., Lee, J., Lim, D.H., Hong, M.E., Kim, K.M., Sohn, I., Jung, S.H., Choi, M.G., Lee, J.H., et al. (2015). Phase III trial to compare adjuvant chemotherapy with capecitabine and cisplatin versus concurrent chemoradiotherapy in gastric cancer: final report of the adjuvant chemoradiotherapy in stomach tumors trial, including survival and subset analyses. J Clin Oncol 33, 3130–3136.
Sadanandam, A., Lyssiotis, C.A., Homicsko, K., Collisson, E.A., Gibb, W. J., Wullschleger, S., Ostos, L.C.G., Lannon, W.A., Grotzinger, C., Del Rio, M., et al. (2013). A colorectal cancer classification system that associates cellular phenotype and responses to therapy. Nat Med 19, 619–625.
Sasako, M., Sakuramoto, S., Katai, H., Kinoshita, T., Furukawa, H., Yamaguchi, T., Nashimoto, A., Fujii, M., Nakajima, T., and Ohashi, Y. (2011). Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol 29, 4387–4393.
Schirle, M., Bantscheff, M., and Kuster, B. (2012). Mass spectrometry-based proteomics in preclinical drug discovery. Chem Biol 19, 72–84.
Schwanhäusser, B., Busse, D., Li, N., Dittmar, G., Schuchhardt, J., Wolf, J., Chen, W., and Selbach, M. (2011). Global quantification of mammalian gene expression control. Nature 473, 337–342.
Schwartz, G.K., Winter, K., Minsky, B.D., Crane, C., Thomson, P.J., Anne, P., Gross, H., Willett, C., and Kelsen, D. (2009). Randomized phase II trial evaluating two paclitaxel and cisplatin-containing chemoradiation regimens as adjuvant therapy in resected gastric cancer (RTOG-0114). J Clin Oncol 27, 1956–1962.
Shah, M.A. (2015). Update on metastatic gastric and esophageal cancers. J Clin Oncol 33, 1760–1769.
Shen, L., Shan, Y.S., Hu, H.M., Price, T.J., Sirohi, B., Yeh, K.H., Yang, Y. H., Sano, T., Yang, H.K., Zhang, X., et al. (2013). Management of gastric cancer in Asia: resource-stratified guidelines. Lancet Oncol 14, e535–e547.
Sheng, W.Q., Huang, D., Ying, J.M., Lu, N., Wu, H.M., Liu, Y.H., Liu, J.P., Bu, H., Zhou, X.Y., and Du, X. (2013). HER2 status in gastric cancers: a retrospective analysis from four Chinese representative clinical centers and assessment of its prognostic significance. Ann Oncol 24, 2360–2364.
Sohn, B.H., Hwang, J.E., Jang, H.J., Lee, H.S., Oh, S.C., Shim, J.J., Lee, K. W., Kim, E.H., Yim, S.Y., Lee, S.H., et al. (2017). Clinical significance of four molecular subtypes of gastric cancer identified by The Cancer Genome Atlas Project. Clin Cancer Res 23, 4441–4449.
Song, N., Pogue-Geile, K.L., Gavin, P.G., Yothers, G., Kim, S.R., Johnson, N.L., Lipchik, C., Allegra, C.J., Petrelli, N.J., O’Connell, M.J., et al. (2016). Clinical outcome from oxaliplatin treatment in stage II/III colon cancer according to intrinsic subtypes. JAMA Oncol 2, 1162–1169.
Sparano, J.A., Gray, R.J., Makower, D.F., Pritchard, K.I., Albain, K.S., Hayes, D.F., Geyer Jr., C.E., Dees, E.C., Perez, E.A., Olson Jr., J.A., et al. (2015). Prospective validation of a 21-gene expression assay in breast cancer. N Engl J Med 373, 2005–2014.
Sparano, J.A., Gray, R.J., Ravdin, P.M., Makower, D.F., Pritchard, K.I., Albain, K.S., Hayes, D.F., Geyer Jr., C.E., Dees, E.C., Goetz, M.P., et al. (2019). Clinical and genomic risk to guide the use of adjuvant therapy for breast cancer. N Engl J Med 380, 2395–2405.
Van Cutsem, E., Sagaert, X., Topal, B., Haustermans, K., and Prenen, H. (2016). Gastric cancer. Lancet 388, 2654–2664.
Vargas, A.J., and Harris, C.C. (2016). Biomarker development in the precision medicine era: lung cancer as a case study. Nat Rev Cancer 16, 525–537.
Zhu, Y., Weiss, T., Zhang, Q., Sun, R., Wang, B., Yi, X., Wu, Z., Gao, H., Cai, X., Ruan, G., et al. (2019). High-throughput proteomic analysis of FFPE tissue samples facilitates tumor stratification. Mol Oncol 13, 2305–2328.
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFC1308900, 2017YFC0908404, 2018YFA0507503, 2017YFA0505103), Beijing Municipal Government Key Research and Development Program (Z181100001918020, Z161100002616036), the National Natural Science Foundation of China (31870828, 81972790, 81672319), the Guangdong Provincial Key R&D Programmes (2019B020229002), the Science and Technology Program of Guangzhou (201902020009), the National Key Basic Research Program of China (2014CBA02002), and the National Key Technology Support Program (2015BAI13B07). This work is dedicated to the memory of Dr. Bei Zhen. We thank the following people for their contributions: all the patients who participated in this study; the clinicians and staff of the institutions who assisted with the studies; and Drs. Jing Gao, Sai Ge, Yanyan Li, Xiaoyi Chong (Department of Gastrointestinal Oncology, Peking University Cancer Hospital and Institute) for assistance with data management and logistic support; and Dongmei Lin, Zhongwu Li, Bin Dong (Department of Pathology, Peking University Cancer Hospital and Institute) for assistance with sample evaluation.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Compliance and ethics
The author(s) declare that they have no conflict of interest.
Electronic supplementary material
11427_2021_1966_MOESM5_ESM.docx
Proteomics provides individualized options of precision medicine for patients with gastric cancer, approximately 374 KB.
Rights and permissions
About this article
Cite this article
Huang, W., Zhan, D., Li, Y. et al. Proteomics provides individualized options of precision medicine for patients with gastric cancer. Sci. China Life Sci. 64, 1199–1211 (2021). https://doi.org/10.1007/s11427-021-1966-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-021-1966-4