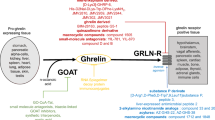
Abstract
Ghrelin O-acyltransferase (GOAT), a member of MBOATs family, is essential for octanoylation of ghrelin, which is required for active ghrelin to bind with and activate its receptor. GOAT is expressed mainly in the stomach, pancreas and hypothalamus. Levels of GOAT are altered by energy status. GOAT contains 11 transmembrane helices and one reentrant loop. Its invariant residue His-338 and conserved Asn-307 are located in the endoplasmic reticulum lumen and cytosol respectively. GOAT contributes to the regulation of food intake and energy expenditure, as well as glucose and lipids homeostasis. Deletion of GOAT blocks the acylation of ghrelin leading to subsequent impairment in energy homeostasis and survival when mice are challenged with high energy diet or severe caloric restriction. GO-CoA-Tat, a peptide GOAT inhibitor, attenuates acyl-ghrelin production and prevents weight gain induced by a medium-chain triglycerides-rich high fat diet. Further, GO-CoA-Tat increases glucose- induced insulin secretion. Overall, inhibition of GOAT is a novel strategy for treatment of obesity and related metabolic disorders.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alen, F., Crespo, I., Ramirez-Lopez, M.T., Jagerovic, N., Goya, P., de Fonseca, F.R., de Heras, R.G., and Orio, L. (2013). Ghrelin-induced orexigenic effect in rats depends on the metabolic status and is counteracted by peripheral CB1 receptor antagonism. PLoS One 8, e60918.
An, W., Li, Y., Xu, G., Zhao, J., Xiang, X., Ding, L., Li, J., Guan, Y., Wang, X., Tang, C., Li, X., Mulholland, M., and Zhang, W. (2010). Modulation of ghrelin O-acyltransferase expression in pancreatic islets. Cell Physiol. Biochem 26, 707–716.
Asakawa, A., Inui, A., Kaga, T., Yuzuriha, H., Nagata, T., Ueno, N., Makino, S., Fujimiya, M., Niijima, A., Fujino, M.A., and Kasuga, M. (2001). Ghrelin is an appetite-stimulatory signal from stomach with structural resemblance to motilin. Gastroenterology 120, 337–345.
Barnett, B.P., Hwang, Y., Taylor, M.S., Kirchner, H., Pfluger, P.T., Bernard, V., Lin, Y.Y., Bowers, E.M., Mukherjee, C., Song, W.J., Longo, P.A., Leahy, D.J., Hussain, M.A., Tschop, M.H., Boeke, J.D., and Cole, P.A. (2010). Glucose and weight control in mice with a designed ghrelin O-acyltransferase inhibitor. Science 330, 1689–1692.
Bowers, C.Y., Reynolds, G.A., and Momany, F.A. (1984). New advances on the regulation of growth hormone (GH) secretion. Internat. J. Neurol 18, 188–205.
Cai, H., Cong, W.N., Daimon, C.M., Wang, R., Tschop, M.H., Sevigny, J., Martin, B., and Maudsley, S. (2013). Altered lipid and salt taste responsivity in ghrelin and GOAT null mice. PLoS One 8, e76553.
Chen, M.H., Li, Y.J., Kawakami, T., Xu, S.M., and Chuang, P.T. (2004). Palmitoylation is required for the production of a soluble multimeric Hedgehog protein complex and long-range signaling in vertebrates. Genes Dev 18, 641–659.
Cummings, D.E., Purnell, J.Q., Frayo, R.S., Schmidova, K., Wisse, B.E., and Weigle, D.S. (2001). A preprandial rise in plasma ghrelin levels suggests a role in meal initiation in humans. Diabetes 50, 1714–1719.
Davies, J.S., Kotokorpi, P., Eccles, S.R., Barnes, S.K., Tokarczuk, P.F., Allen, S.K., Whitworth, H.S., Guschina, I.A., Evans, B.A., Mode, A., Zigman, J.M., and Wells, T. (2009). Ghrelin induces abdominal obesity via GHS-R-dependent lipid retention. Mol Endocrinol 23, 914–924.
Davis, J.F., Perello, M., Choi, D.L., Magrisso, I.J., Kirchner, H., Pfluger, P.T., Tschoep, M., Zigman, J.M., and Benoit, S.C. (2012). GOAT induced ghrelin acylation regulates hedonic feeding. Hormones Behav 62, 598–604.
De Winter, B.Y., De Man, J.G., Seerden, T.C., Depoortere, I., Herman, A.G., Peeters, T.L., and Pelckmans, P.A. (2004). Effect of ghrelin and growth hormone-releasing peptide 6 on septic ileus in mice. Neurogastroenterol Motil 16, 439–446.
Esler, W.P., Rudolph, J., Claus, T.H., Tang, W., Barucci, N., Brown, S.E., Bullock, W., Daly, M., Decarr, L., Li, Y., Milardo, L., Molstad, D., Zhu, J., Gardell, S.J., Livingston, J.N., and Sweet, L.J. (2007). Small-molecule ghrelin receptor antagonists improve glucose tolerance, suppress appetite, and promote weight loss. Endocrinology 148, 5175–5185.
Finger, B.C., Dinan, T.G., and Cryan, J.F. (2012). Diet-induced obesity blunts the behavioural effects of ghrelin: studies in a mouse-progressive ratio task. Psychopharmacology 220, 173–181.
Fujino, K., Inui, A., Asakawa, A., Kihara, N., Fujimura, M., and Fujimiya, M. (2003). Ghrelin induces fasted motor activity of the gastrointestinal tract in conscious fed rats. J Physiol 550, 227–240.
Fukuda, H., Mizuta, Y., Isomoto, H., Takeshima, F., Ohnita, K., Ohba, K., Omagari, K., Taniyama, K., and Kohno, S. (2004). Ghrelin enhances gastric motility through direct stimulation of intrinsic neural pathways and capsaicin-sensitive afferent neurones in rats. Scandinavian J Gastroenterol 39, 1209–1214.
Gahete, M.D., Cordoba-Chacon, J., Salvatori, R., Castano, J.P., Kineman, R.D., and Luque, R.M. (2010). Metabolic regulation of ghrelin O-acyl transferase (GOAT) expression in the mouse hypothalamus, pituitary, and stomach. Mol. Cell Endocrinol 317, 154–160.
Garner, A.L., and Janda, K.D. (2011). A small molecule antagonist of ghrelin O-acyltransferase (GOAT). Chem Commun 47, 7512–7514.
Goebel-Stengel, M., Hofmann, T., Elbelt, U., Teuffel, P., Ahnis, A., Kobelt, P., Lambrecht, N.W., Klapp, B.F., and Stengel, A. (2013). The ghrelin activating enzyme ghrelin-O-acyltransferase (GOAT) is present in human plasma and expressed dependent on body mass index. Peptides 43, 13–19.
Gonzalez, C.R., Vazquez, M.J., Lopez, M., and Dieguez, C. (2008). Influence of chronic undernutrition and leptin on GOAT mRNA levels in rat stomach mucosa. J Mol Endocrinol 41, 415–421.
Gutierrez, J.A., Solenberg, P.J., Perkins, D.R., Willency, J.A., Knierman, M.D., Jin, Z., Witcher, D.R., Luo, S., Onyia, J.E., and Hale, J.E. (2008). Ghrelin octanoylation mediated by an orphan lipid transferase. Proc Natl Acad Sci USA 105, 6320–6325.
Heppner, K.M., Muller, T.D., Kirchner, H., Perez-Tilve, D., Pfluger, P.T., Tschop, M.H., and Hofmann, S.M. (2013). The role of ghrelinoctanoyl- acyl-transferase in thermoregulation. J Endocrinol Invest 36, 180–184.
Hofmann, K. (2000). A superfamily of membrane-bound O-acyltransferases with implications for wnt signaling. Trends Biochem Sci 25, 111–112.
Howard, A.D., Feighner, S.D., Cully, D.F., Arena, J.P., Liberator, P.A., Rosenblum, C.I., Hamelin, M., Hreniuk, D.L., Palyha, O.C., Anderson, J., Paress, P.S., Diaz, C., Chou, M., Liu, K.K., McKee, K.K., Pong, S.S., Chaung, L.Y., Elbrecht, A., Dashkevicz, M., Heavens, R., Rigby, M., Sirinathsinghji, D.J., Dean, D.C., Melillo, D.G., Patchett, A.A., Nargund, R., Griffin, P.R., DeMartino, J.A., Gupta, S.K., Schaeffer, J.M., Smith, R.G., and van der Ploeg, L.H. (1996). A receptor in pituitary and hypothalamus that functions in growth hormone release. Science 273, 974–977.
Jaszberenyi, M., Bujdoso, E., Bagosi, Z., and Telegdy, G. (2006). Mediation of the behavioral, endocrine and thermoregulatory actions of ghrelin. Horm Behav 50, 266–273.
Kang, K., Schmahl, J., Lee, J.M., Garcia, K., Patil, K., Chen, A., Keene, M., Murphy, A., and Sleeman, M.W. (2012). Mouse ghrelin-Oacyltransferase (GOAT) plays a critical role in bile acid reabsorption. FASEB J 26, 259–271.
Khalil, E.M., De Angelis, J., Ishii, M., and Cole, P.A. (1999). Mechanism-based inhibition of the melatonin rhythm enzyme: pharmacologic exploitation of active site functional plasticity. Proc Natl Acad Sci USA 96, 12418–12423.
Kirchner, H., Gutierrez, J.A., Solenberg, P.J., Pfluger, P.T., Czyzyk, T.A., Willency, J.A., Schurmann, A., Joost, H.G., Jandacek, R.J., Hale, J.E., Heiman, M.L., and Tschop, M.H. (2009). GOAT links dietary lipids with the endocrine control of energy balance. Nat Med 15, 741–745.
Kirchner, H., Heppner, K.M., Holland, J., Kabra, D., Tschop, M.H., and Pfluger, P.T. (2013). Ablation of ghrelin O-acyltransferase does not improve glucose intolerance or body adiposity in mice on a leptin-deficient ob/ob background. PLoS One 8, e61822.
Kojima, M., Hosoda, H., Date, Y., Nakazato, M., Matsuo, H., and Kangawa, K. (1999). Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402, 656–660.
Landgren, S., Simms, J.A., Hyytia, P., Engel, J.A., Bartlett, S.E., and Jerlhag, E. (2012). Ghrelin receptor (GHS-R1A) antagonism suppresses both operant alcohol self-administration and high alcohol consumption in rats. Addict Biol 17, 86–94.
Lau, O.D., Kundu, T.K., Soccio, R.E., Ait- Si-Ali, S., Khalil, E.M., Vassilev, A., Wolffe, A.P., Nakatani, Y., Roeder, R.G., and Cole, P.A. (2000). HATs off: selective synthetic inhibitors of the histone acetyltransferases p300 and PCAF. Mol Cell 5, 589–595.
Lawrence, C.B., Snape, A.C., Baudoin, F.M., and Luckman, S.M. (2002). Acute central ghrelin and GH secretagogues induce feeding and activate brain appetite centers. Endocrinology 143, 155–162.
Lee, H.M., Wang, G., Englander, E.W., Kojima, M., and Greeley, G.H. (2002). Ghrelin, a new gastrointestinal endocrine peptide that stimulates insulin secretion: enteric distribution, ontogeny, influence of endocrine, and dietary manipulations. Endocrinology 143, 185–190.
Levin, F., Edholm, T., Schmidt, P.T., Gryback, P., Jacobsson, H., Degerblad, M., Hoybye, C., Holst, J.J., Rehfeld, J.F., Hellstrom, P.M., and Naslund, E. (2006). Ghrelin stimulates gastric emptying and hunger in normal-weight humans. J Clin Endocrinol Metab 91, 3296–3302.
Li, Z., Xu, G., Qin, Y., Zhang, C., Tang, H., Yin, Y., Xiang, X., Li, Y., Zhao, J., Mulholland, M., and Zhang, W. (2014). Ghrelin promotes hepatic lipogenesis by activation of mTOR-PPARgamma signaling pathway. Proc Natl Acad Sci USA 111, 13163–13168.
Lim, C.T., Kola, B., and Korbonits, M. (2011). The ghrelin/GOAT/GHS-R system and energy metabolism. Rev Endocr Metab Disord 12, 173–186.
Lin, L., Saha, P.K., Ma, X., Henshaw, I.O., Shao, L., Chang, B.H., Buras, E.D., Tong, Q., Chan, L., McGuinness, O.P., and Sun, Y. (2011). Ablation of ghrelin receptor reduces adiposity and improves insulin sensitivity during aging by regulating fat metabolism in white and brown adipose tissues. Aging Cell 10, 996–1010.
Longo, K.A., Charoenthongtrakul, S., Giuliana, D.J., Govek, E.K., McDonagh, T., Qi, Y., DiStefano, P.S., and Geddes, B.J. (2008). Improved insulin sensitivity and metabolic flexibility in ghrelin receptor knockout mice. Regul Pept 150, 55–61.
Mela, D.J., and Sacchetti, D.A. (1991). Sensory preferences for fats: relationships with diet and body composition. Am J Clin Nutr 53, 908–915.
Molfino, A., Gioia, G., and Muscaritoli, M. (2013). The hunger hormone ghrelin in cachexia. Exp Opin Biol Ther 13, 465–468.
Nakazato, M., Murakami, N., Date, Y., Kojima, M., Matsuo, H., Kangawa, K., and Matsukura, S. (2001). A role for ghrelin in the central regulation of feeding. Nature 409, 194–198.
Ohgusu, H., Shirouzu, K., Nakamura, Y., Nakashima, Y., Ida, T., Sato, T., and Kojima, M. (2009). Ghrelin O-acyltransferase (GOAT) has a preference for n-hexanoyl-CoA over n-octanoyl-CoA as an acyl donor. Biochem. Biophys Res Commun 386, 153–158.
Ohgusu, H., Takahashi, T., and Kojima, M. (2012). Enzymatic characterization of GOAT, ghrelin O-acyltransferase. Methods Enzymol 514, 147–163.
Patterson, M., Bloom, S.R., and Gardiner, J.V. (2011). Ghrelin and appetite control in humans—potential application in the treatment of obesity. Peptides 32, 2290–2294.
Perello, M., Sakata, I., Birnbaum, S., Chuang, J.C., Osborne-Lawrence, S., Rovinsky, S.A., Woloszyn, J., Yanagisawa, M., Lutter, M., and Zigman, J.M. (2010). Ghrelin increases the rewarding value of high-fat diet in an orexin-dependent manner. Biol Psychiatry 67, 880–886.
Petrova, E., Rios-Esteves, J., Ouerfelli, O., Glickman, J.F., and Resh, M.D. (2013). Inhibitors of Hedgehog acyltransferase block Sonic Hedgehog signaling. Nat Chem Biol 9, 247–249.
Qi, Y., Longo, K.A., Giuliana, D.J., Gagne, S., McDonagh, T., Govek, E., Nolan, A., Zou, C., Morgan, K., Hixon, J., Saunders, J.O., Distefano, P.S., and Geddes, B.J. (2011). Characterization of the insulin sensitivity of ghrelin receptor K Omice using glycemic clamps. BMC Physiol 11, 1.
Reimer, R.A., Maurer, A.D., Lau, D.C., and Auer, R.N. (2010). Long-term dietary restriction influences plasma ghrelin and GOAT mRNA level in rats. Physiol Behav 99, 605–610.
Shiiya, T., Nakazato, M., Mizuta, M., Date, Y., Mondal, M.S., Tanaka, M., Nozoe, S., Hosoda, H., Kangawa, K., and Matsukura, S. (2002). Plasma ghrelin levels in lean and obese humans and the effect of glucose on ghrelin secretion. J Clin Endocrinol Metab 87, 240–244.
Shimbara, T., Mondal, M.S., Kawagoe, T., Toshinai, K., Koda, S., Yamaguchi, H., Date, Y., and Nakazato, M. (2004). Central administration of ghrelin preferentially enhances fat ingestion. Neurosci Lett 369, 75–79.
Shin, Y.K., Martin, B., Kim, W., White, C.M., Ji, S., Sun, Y., Smith, R.G., Sevigny, J., Tschop, M.H., Maudsley, S., and Egan, J.M. (2010). Ghrelin is produced in taste cells and ghrelin receptor null mice show reduced taste responsivity to salty (NaCl) and sour (citric acid) tastants. PLoS One 5, e12729.
Skibicka, K.P., Hansson, C., Egecioglu, E., and Dickson, S.L. (2012). Role of ghrelin in food reward: impact of ghrelin on sucrose self-administration and mesolimbic dopamine and acetylcholine receptor gene expression. Addict Biol 17, 95–107.
Smith, R.G., Sun, Y., Betancourt, L., and Asnicar, M. (2004). Growth hormone secretagogues: prospects and potential pitfalls. Best Pract. Res Clin Endocrinol Metab 18, 333–347.
Srinivas, S.R., Gopal, E., Zhuang, L., Itagaki, S., Martin, P.M., Fei, Y.J., Ganapathy, V., and Prasad, P.D. (2005). Cloning and functional identification of slc5a12 as a sodium-coupled low-affinity transporter for monocarboxylates (SMCT2). Biochem J 392, 655–664.
Stengel, A., Goebel, M., Wang, L., Tache, Y., Sachs, G., and Lambrecht, N.W. (2010). Differential distribution of ghrelin-O-acyltransferase (GOAT) immunoreactive cells in the mouse and rat gastric oxyntic mucosa. Biochem Biophys Res Commun 392, 67–71.
Stevanovic, D., Trajkovic, V., Muller-Luhlhoff, S., Brandt, E., Abplanalp, W., Bumke-Vogt, C., Liehl, B., Wiedmer, P., Janjetovic, K., Starcevic, V., Pfeiffer, A.F., Al-Hasani, H., Tschop, M.H., and Castaneda, T.R. (2013). Ghrelin-induced food intake and adiposity depend on central mTORC1/S6K1 signaling. Mol Cell Endocrinol 381, 280–290.
Sun, Y., Asnicar, M., Saha, P.K., Chan, L., and Smith, R.G. (2006). Ablation of ghrelin improves the diabetic but not obese phenotype of ob/ob mice. Cell Metab 3, 379–386.
Szentirmai, E., Kapas, L., Sun, Y., Smith, R.G., and Krueger, J.M. (2009). The preproghrelin gene is required for the normal integration of thermoregulation and sleep in mice. Proc Natl Acad Sci USA 106, 14069–14074.
Tack, J., Depoortere, I., Bisschops, R., Delporte, C., Coulie, B., Meulemans, A., Janssens, J., and Peeters, T. (2006). Influence of ghrelin on interdigestive gastrointestinal motility in humans. Gut 55, 327–333.
Takada, R., Satomi, Y., Kurata, T., Ueno, N., Norioka, S., Kondoh, H., Takao, T., and Takada, S. (2006). Monounsaturated fatty acid modification of Wnt protein: its role in Wnt secretion. Dev Cell 11, 791–801.
Taylor, M.S., Ruch, T.R., Hsiao, P.Y., Hwang, Y., Zhang, P., Dai, L., Huang, C.R., Berndsen, C.E., Kim, M.S., Pandey, A., Wolberger, C., Marmorstein, R., Machamer, C., Boeke, J.D., and Cole, P.A. (2013). Architectural organization of the metabolic regulatory enzyme ghrelin O-acyltransferase. J Biol Chem 288, 32211–32228.
Teramae, H., Yoshikawa, T., Inoue, R., Ushida, K., Takebe, K., Nio-Kobayashi, J., and Iwanaga, T. (2010). The cellular expression of SMCT2 and its comparison with other transporters for monocarboxylates in the mouse digestive tract. Biomed Res 31, 239–249.
Teubner, B.J., Garretson, J.T., Hwang, Y., Cole, P.A., and Bartness, T.J. (2013). Inhibition of ghrelin O-acyltransferase attenuates food deprivation-induced increases in ingestive behavior. Horm Behav 63, 667–673
Theander-Carrillo, C., Wiedmer, P., Cettour-Rose, P., Nogueiras, R., Perez-Tilve, D., Pfluger, P., Castaneda, T.R., Muzzin, P., Schurmann, A., Szanto, I., Tschop, M.H., and Rohner-Jeanrenaud, F. (2006). Ghrelin action in the brain controls adipocyte metabolism. J Clin Invest 116, 1983–1993.
Trudel, L., Tomasetto, C., Rio, M.C., Bouin, M., Plourde, V., Eberling, P., and Poitras, P. (2002). Ghrelin/motilin-related peptide is a potent prokinetic to reverse gastric postoperative ileus in rat. Am J Physiol Gastrointest Liver Physiol 282, G948–952.
Tschop, M., Smiley, D.L., and Heiman, M.L. (2000). Ghrelin induces adiposity in rodents. Nature 407, 908–913.
Tsubone, T., Masaki, T., Katsuragi, I., Tanaka, K., Kakuma, T., and Yoshimatsu, H. (2005). Ghrelin regulates adiposity in white adipose tissue and UCP1 mRNA expression in brown adipose tissue in mice. Regul. Peptides 130, 97–103.
Vaughan, A.M., and Oram, J.F. (2005). ABCG1 redistributes cell cholesterol to domains removable by high density lipoprotein but not by lipid-depleted apolipoproteins. J Biol Chem 280, 30150–30157.
Wiedmer, P., Strasser, F., Horvath, T.L., Blum, D., Dimarchi, R., Lutz, T., Schurmann, A., Joost, H.G., Tschop, M.H., and Tong, J. (2011). Ghrelin-induced hypothermia: a physiological basis but no clinical risk. Physiol Behav 105, 43–51.
Xu, G., Li, Y., An, W., Li, S., Guan, Y., Wang, N., Tang, C., Wang, X., Zhu, Y., Li, X., Mulholland, M.W., and Zhang, W. (2009). Gastric mammalian target of rapamycin signaling regulates ghrelin production and food intake. Endocrinology 150, 3637–3644.
Xu, G., Wang, Z., Li, Y., Li, Z., Tang, H., Zhao, J., Xiang, X., Ding, L., Ma, L., Yuan, F., Fei, J., Wang, W., Wang, N., Guan, Y., Tang, C., Mulholland, M., and Zhang, W. (2012). Ghrelin contributes to derangements of glucose metabolism induced by rapamycin in mice. Diabetologia 55, 1813–1823.
Yang, J., Brown, M.S., Liang, G., Grishin, N.V., and Goldstein, J.L. (2008a). Identification of the acyltransferase that octanoylates ghrelin, an appetite-stimulating peptide hormone. Cell 132, 387–396.
Yang, J., Zhao, T.J., Goldstein, J.L., and Brown, M.S. (2008b). Inhibition of ghrelin O-acyltransferase (GOAT) by octanoylated pentapeptides. Proc Natl Acad Sci USA 105, 10750–10755.
Zhao, T.J., Liang, G., Li, R.L., Xie, X., Sleeman, M.W., Murphy, A.J., Valenzuela, D.M., Yancopoulos, G.D., Goldstein, J.L., and Brown, M.S. (2010). Ghrelin O-acyltransferase (GOAT) is essential for growth hormone-mediated survival of calorie-restricted mice. Proc Natl Acad Sci USA 107, 7467–7472.
Zhu, X., Cao, Y., Voogd, K., and Steiner, D.F. (2006). On the processing of proghrelin to ghrelin. J Biol Chem 281, 38867–38870.
Zigman, J.M., Nakano, Y., Coppari, R., Balthasar, N., Marcus, J.N., Lee, C.E., Jones, J.E., Deysher, A.E., Waxman, A.R., White, R.D., Williams, T.D., Lachey, J.L., Seeley, R.J., Lowell, B.B., and Elmquist, J.K. (2005). Mice lacking ghrelin receptors resist the development of diet-induced obesity. J Clin Invest 115, 3564–3572.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Li, Z., Mulholland, M. & Zhang, W. Ghrelin O-acyltransferase (GOAT) and energy metabolism. Sci. China Life Sci. 59, 281–291 (2016). https://doi.org/10.1007/s11427-015-4973-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4973-6