Abstract
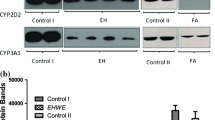
This study explored the effects of cucurbitacin E (CuE), a bioactive compound from Cucurbitaceae, on the metabolism/pharmacokinetic of tolbutamide, a model CYP2C9/11 probe substrate, and hepatic CYP2C11 expression in rats. Liquid chromatography-(tandem) mass spectrometry (LC-MS/MS) assay was used to detect tolbutamide as well as 4-hydroxytolbutamide, and then successfully applied to the pharmacokinetic study of tolbutamide in rats. The effect of CuE on CYP2C11 expression was determined by western blot. CuE (1.25–100 μmol L−1) competitively inhibited tolbutamide 4-hydroxylation (CYP2C11) activity only in concentration-dependent manner with a K i value of 55.5 μmol L−1 in vitro. In whole animal studies, no significant difference in metabolism/pharmacokinetic of tolbutamide was found for the single pretreatment groups. In contrast, multiple pretreatments of CuE (200 μg kg−1 d−1, 3 d, i.p.) significantly decreased tolbutamide clearance (CL) by 25% and prolonged plasma half-time (T 1/2) by 37%. Moreover, CuE treatment (50–200 μg kg−1 d−1, i.p.) for 3 d did not affect CYP2C11 expression. These findings demonstrated that CuE competitively inhibited the metabolism of CYP2C11 substrates but had no effect on rat CYP2C11 expression. This study may provide a useful reference for the reasonable and safe use of herbal or natural products containing CuE to avoid unnecessary drug-drug interactions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Guengerich FP. Cytochrome P450: what have we learned and what are the future issues? Drug Metab Rev, 2004, 36: 159–197
Wang X. Evaluation of Cytochrome P450 enzymes in drug interactions. In: Wu J, ed. Cytochrome P450 Enzymes: Biochemistry, Pharmacology and Health Implications. New York: Nova Science Publishers, 2014
Zhou S, Gao Y, Jiang W, Huang M, Xu A, Paxton JW. Interactions of herbs with cytochrome P450. Drug Metab Rev, 2003, 35: 35–98
Bhandari P, Kumar N, Singh B, Kaul VK. Cucurbitacins from Bacopa monnieri. Phytochemistry, 2007, 68: 1248–1254
Abbas S, Vincourt JB, Habib L, Netter P, Greige-Gerges H, Magdalou J. The ucurbitacins E, Dand I: investigation of their cytotoxicity toward human chondrosarcoma SW1353 cell line and their biotransformation in man liver. Toxicol Lett, 2013, 216: 189–199
Abdelwahab SI, Hassan LE, Sirat HM, Yagi SM, Koko WS, Mohan S, Taha MM, Ahmad S, Chuen CS, Narrima P, Rais MM, Hadi AH. Anti-inflammatory activities of cucurbitacin E isolated from Citrullus lanatus var. citroides: role of reactive nitrogen species and cyclooxygenase enzyme inhibition. Fitoterapia, 2011, 82: 1190–1197
Qiao J, Xu LH, He J, Ouyang DY, He XH. Cucurbitacin E exhibits anti-inflammatory effect in RAW 264.7 cells via suppression of NF-kappaB nuclear translocation. Inflamm Res, 2013, 62: 461–469
Sun C, Zhang M, Shan X, Zhou X, Yang J, Wang Y, Li-Ling J, Deng Y. Inhibitory effect of cucurbitacin E on pancreatic cancer cells growth via STAT3 signaling. J Cancer Res Clin Oncol, 2010, 136: 603–610
Dong Y, Lu B, Zhang X, Zhang J, Lai L, Li D, Wu Y, Song Y, Luo J, Pang X, Yi Z, Liu M. Cucurbitacin E, a tetracyclic triterpenes compound from Chinese medicine, inhibits tumor angiogenesis through VEGFR2-mediated Jak2-STAT3 signaling pathway. Carcinogenesis, 2010, 31: 2097–2104
Li Y, Wang R, Ma E, Deng Y, Wang X, Xiao J, Jing Y. The induction of G2/M cell-cycle arrest and apoptosis by cucurbitacin E is associated with increased phosphorylation of eIF2alpha in leukemia cells. Anticancer Drugs, 2010, 21: 389–400
Zhang T, Li J, Dong Y, Zhai D, Lai L, Dai F, Deng H, Chen Y, Liu M, Yi Z. Cucurbitacin E inhibits breast tumor metastasis by suppressing cell migration and invasion. Breast Cancer Res Treat, 2012, 135: 445–458
Lan T, Wang L, Xu Q, Liu W, Jin H, Mao W, Wang X. Growth inhibitory effect of Cucurbitacin E on breast cancer cells. Int J Clin Exp Pathol, 2013, 6: 1799–1805
Attard E, Brincat MP, Cuschieri A. Immunomodulatory activity of cucurbitacin E isolated from Ecballium elaterium. Fitoterapia, 2005, 76: 439–441
China Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China (Part One). Beijing: China Medical Science Press, 2010
Ding T, Zhang Y, Chen A, Tang Y, Liu M, Wang X. Effects of Cucurbitacin E, a tetracyclic triterpene compound from Cucurbitaceae, on the pharmacokinetics and pharmacodynamics of warfarin in Rats. Basic Clin Pharmacol Toxicol, 2015, 116: 385–389
Jensen BP, Chin PK, Roberts RL, Begg EJ. Influence of adult age on the total and free clearance and protein binding of (R)- and (S)-warfarin. Br J Clin Pharmacol, 2012, 74: 797–805
Kaminsky LS, Zhang ZY. Human P450 metabolism of warfarin. Pharmacol Ther, 1997, 73: 67–74
Wu WW, Yeung JH. Inhibition of warfarin hydroxylation by major tanshinones of Danshen (Salvia miltiorrhiza) in the rat in vitro and in vivo. Phytomedicine, 2010, 17: 219–226
Brown RE, Jarvis KL, Hyland KJ. Protein measurement using bicinchoninic acid: elimination of interfering substances. Anal Biochem, 1989, 180: 136–139
Schenkman, JB, Jansson I. Spectral analysis of cytochrome P450. In: Phillips IR, Stephard EA, eds. Methods in Molecular Biology, vol. 107: Cytochrome P450 Protocols. Totowa: Human Press, 1998
Wang X, Lee WY, Or PM, Yeung JH. Pharmacokinetic interaction studies of tanshinones with tolbutamide, a model CYP2C11 probe substrate, using liver microsomes, primary hepatocytes and in vivo in the rat. Phytomedicine, 2010, 17: 203–211
Sun M, Tang Y, Ding T, Liu M, Wang X. Inhibitory effects of celastrol on rat liver cytochrome P450 1A2, 2C11, 2D6, 2E1 and 3A2 activity. Fitoterapia, 2014, 92: 1–8
Chen A, Qin X, Tang Y, Liu M, Wang X. Evaluation of enzyme inhibition kinetics in drug-drug interactions. Chem Biol Interact, 2014, 222: 133–134
Sun M, Ding TG, Tang Y, Liu MY, Wang X. Cucurbitacin E exhibits inhibitory effects on CYP2C, 3A activities in rat and human liver in vitro. Acta Pharmacol Sin, 2013, 34: 29–29
Ouellet D, Bramson C, Roman D, Remmers AE, Randinitis E, Milton A, Gardner M. Effects of three cytochrome P450 inhibitors, ketoconazole, fluconazole, and paroxetine, on the pharmacokinetics of lasofoxifene. Br J Clin Pharmacol, 2007, 63: 59–66
Tateishi T, Asoh M, Nakura H, Watanabe M, Tanaka M, Kumai T, Kobayashi S. Carbamazepine induces multiple cytochrome P450 subfamilies in rats. Chem Biol Interact, 1999, 117: 257–268
Rettie AE, Jones JP. Clinical and toxicological relevance of CYP2C9: drug-drug interactions and pharmacogenetics. Annu Rev Pharmacol Toxicol, 2005, 45: 477–494
Yeung JH, Or PM. Polysaccharide peptides from Coriolus versicolor competitively inhibit tolbutamide 4-hydroxylation in specific human CYP2C9 isoform and pooled human liver microsomes. Phytomedicine, 2011, 18: 1170–1175
Kakkar T, Boxenbaum H, Mayersohn M. Estimation of Ki in a competitive enzyme-inhibition model: comparisons among three methods of data analysis. Drug Metab Dispos, 1999, 27: 756–762
Wang X, Lee WY, Or PM, Yeung JH. Effects of major tanshinones isolated from Danshen (Salvia miltiorrhiza) on rat CYP1A2 expression and metabolism of model CYP1A2 probe substrates. Phytomedicine, 2009, 16: 712–725
Wang X, Yeung JH. Effects of the aqueous extract from Salvia miltiorrhiza Bunge on caffeine pharmacokinetics and liver microsomal CYP1A2 activity in humans and rats. J Pharm Pharmacol, 2010, 62: 1077–1083
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81301908), and the Science and Technology Commission of Shanghai Municipality (13ZR1412600, 14DZ2270100).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Lu, J., Ding, T., Qin, X. et al. In vitro and in vivo evaluation of cucurbitacin E on rat hepatic CYP2C11 expression and activity using LC-MS/MS. Sci. China Life Sci. 60, 215–224 (2017). https://doi.org/10.1007/s11427-015-4911-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-015-4911-7