Abstract
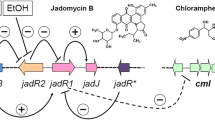
The biosynthesis of antibiotics is controlled by cascade regulation involving cluster-situated regulators (CSRs) and pleiotropic regulators. Three CSRs have been identified in the jadomycin biosynthetic gene cluster, including one OmpR-type activator (JadR1) and two TetR-like repressors (JadR* and JadR2). To examine their interactions in jadomycin biosynthesis, a series of mutants were generated and tested for jadomycin production. We noticed that jadomycin production in the jadR*-jadR2 double mutant was increased dramatically compared with either single mutant. Transcriptional analysis showed that jadR* and jadR2 act synergistically to repress jadomycin production by inhibiting the transcription of jadR1. Furthermore, jadR* and jadR2 reciprocally inhibit each other. The complex interactions among these three CSRs may provide clues for the activation of the jadomycin gene cluster, which would otherwise remain silent without stimulation from stress signals.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Liu G, Chater K F, Govind C, et al. Molecular regualtion of antibiotic biosynthesis in Streptomyces. Microbiol Mol Biol R, 2013, 17: 112–143
Arias P, Fernández-Moreno M A, Malpartida F. Characterization of the pathway-specific positive transcriptional regulator for actinorhodin biosynthesis in Streptomyces coelicolor A3(2) as a DNA-binding protein. J Bacteriol, 1999, 181: 6958–6968
Liu G, Tian Y Q, Yang H H, et al. A pathway-specific transcriptional regulatory gene for nikkomycin biosynthesis in Streptomyces ansochromogenes that also influences colony development. Mol Microbiol, 2005, 55: 1855–1866
Li R, Liu G, Xie Z J, et al. PolY, a transcriptional regulator with ATPase activity, directly activates transcription of polR in polyoxin biosynthesis in Streptomyces cacaoi. Mol Microbiol, 2010, 75: 349–364
Li R, Xie Z J, Tian Y Q, et al. polR, a pathway-specific transcriptional gene, positively controls polyoxin biosynthesis in Streptomyces cacaoi subsp. asoensis. Microbiol, 2009, 155: 1819–1831
Ajithkumar V, Karuppasamy K, Prasad R. Regulation of daunorubicin biosynthesis in Streptomyces peucetius feed-forward and feedback transcriptional control. J Basic Microbiol, 2013, 53: 1–9
Cundliffe E. Control of tylosin biosynthesis in Streptomyces fradiae. J Microbiol Biotechnol, 2008, 18: 1485–1491
Doull J L, Ayer S W, Singh A K, et al. Production of a novel polyketide antibiotic, jadomycin B, by Streptomyces venezuelae following heat shock. J Antibiot (Tokyo), 1993, 46: 869–871
Doull J L, Singh A K, Hoare M, et al. Conditions for the production of jadomycin B by Streptomyces venezuelae ISP5230: effects of heat shock, ethanol treatment and phage infection. J Ind Microbiol, 1994, 13: 120–125
Zheng J T, Rix U, Zhao L X, et al. Cytotoxic activities of new jadomycin derivatives. J Antibiot (Tokyo), 2005, 58: 405–408
Jakeman D L, Bandi S, Graham C L, et al. Antimicrobial activities of jadomycin B and structurally related analogues. Antimicrob Agents Chemother, 2009, 53: 1245–1247
Fan K Q, Zhang X, Liu H C, et al, Evaluation of the cytotoxic activity of new jadomycin derivatives reveals the potential to improve its selectivity against tumor cells. J Antibiot (Tokyo), 2012a, 65: 449–452
Wang L Q, Tian X Y, Wang J, et al. Autoregulation of antibiotic biosynthesis by binding of the end product to an atypical response regulator. Proc Natl Acad Sci USA, 2009, 106: 8617–8622
Xu G M, Wang J, Wang L Q, et al. “Pseudo” ?-butyrolactone receptors respond to antibiotic signals to coordinate antibiotic biosynthesis. J Biol Chem 2010, 285: 27440–27448
Yang K Q, Han L, He J Y, et al. A repressor-response regulator gene pair controlling jadomycin B production in Streptomyces venezuelae ISP5230. Gene, 2001, 279: 165–173
Jakeman D L, Graham C L, Young W, et al. Culture conditions improving the production of jadomycin B. J Ind Microbiol Biotechnol, 2006, 33: 767–772
Bierman M, Logan R, O’Brien K, et al. Plasmid cloning vectors for the conjugal transfer of DNA from Escherichia coli to Streptomyces spp. Gene, 1992, 116: 43–49
Kieser T, Bibb M J, Buttner M J, et al. Practical Streptomyces Genetics. Norwich: The John Innes Foundation, 2000
Livak K J, Schmittgen T D. Analysis of relative gene expression data using real-time quantitative PCR and the
 method. Methods, 2001, 25: 402–408
method. Methods, 2001, 25: 402–408Yang H H, Wang L Q, Xie Z J, et al. The tyrosine degradation gene hppD is transcriptionally activated by HpdA and repressed by HpdR in Streptomyces coelicolor, while hpdA is negatively autoregulated and repressed by HpdR. Mol Microbiol, 2007, 65: 1064–1077
Gottelt M, Kol S, Gomez-Escribano J P, et al. Deletion of a regulatory gene within the cpk gene cluster reveals novel antibacterial activity in Streptomyces coelicolor A3(2). Microbiol, 2010, 156: 2343–2353
O’Rourke S, Wietzorrek A, Fowler K, et al. Extracellular signalling, translational control, two repressors and an activator all contribute to the regulation of methylenomycin production in Streptomyces coelicolor. Mol Microbiol, 2009, 71: 763–778
Wang J, Wang W S, Wang L Q, et al. A novel role of’ pseudo’ γ-butyrolactone receptors in controlling γ-butyrolactone biosynthesis in Streptomyces. Mol Microbiol, 2011, 82: 236–250
Xie P F, Sheng Y, Ito T, et al. Transcriptional regulation and increased production of asukamycin in engineered Streptomyces nodosus subsp. asukaensis strains. Appl Microbiol Biotechnol, 2012, 96: 451–460
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Zhang, Y., Zou, Z., Niu, G. et al. jadR* and jadR2 act synergistically to repress jadomycin biosynthesis. Sci. China Life Sci. 56, 584–590 (2013). https://doi.org/10.1007/s11427-013-4508-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-013-4508-y




 method. Methods, 2001, 25: 402–408
method. Methods, 2001, 25: 402–408