Abstract
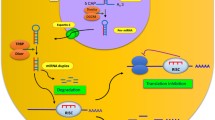
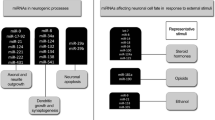
MicroRNAs play important roles in post-transcriptional regulation of gene expression by inhibiting protein translation and/or promoting mRNA degradation. Importantly, biogenesis of microRNAs displays specific temporal and spatial profiles in distinct cell and tissue types and hence affects a broad spectrum of biological functions in normal cell growth and tumor development. Recent discoveries have revealed sophisticated mechanisms that control microRNA production and homeostasis in response to developmental and extracellular signals. Moreover, a link between dysregulation of microRNAs and human brain disorders has become increasingly evident. In this review, we focus on recent advances in understanding the regulation of microRNA biogenesis and function in neuronal and glial development in the mammalian brain, and dysregulation of the microRNA pathway in neurodevelopmental and neurodegenerative diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bushati N, Cohen S M. microRNA functions. Annu Rev Cell Dev Biol, 2007, 23: 175–205
Kim V N, Han J, Siomi M C. Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol, 2009, 10: 126–139
Flynt A S, Lai E C. Biological principles of microRNA-mediated regulation: shared themes amid diversity. Nat Rev Genet, 2008, 9: 831–842
Ruby J G, Jan C H, Bartel D P. Intronic microRNA precursors that bypass Drosha processing. Nature, 2007, 448: 83–86
Berezikov E, Chung W J, Willis J, et al. Mammalian mirtron genes. Mol Cell, 2007, 28: 328–336
Okamura K, Hagen J W, Duan H, et al. The mirtron pathway generates microRNA-class regulatory RNAs in Drosophila. Cell, 2007, 130: 89–100
Bogerd H P, Karnowski H W, Cai X, et al. A mammalian herpesvirus uses noncanonical expression and processing mechanisms to generate viral MicroRNAs. Mol Cell, 2010, 37: 135–142
Viswanathan S R, Daley G Q, Gregory R I. Selective blockade of microRNA processing by Lin28. Science, 2008, 320: 97–100
Michlewski G, Caceres J F. Antagonistic role of hnRNP A1 and KSRP in the regulation of let-7a biogenesis. Nat Struct Mol Biol, 2010, 17: 1011–1018
Brennecke J, Stark A, Russell R B, et al. Principles of microRNA-target recognition. PLoS Biol, 2005, 3: e85
Shen W F, Hu Y L, Uttarwar L, et al. MicroRNA-126 regulates HOXA9 by binding to the homeobox. Mol Cell Biol, 2008, 28: 4609–4619
Forman J J, Legesse-Miller A, Coller H A. A search for conserved sequences in coding regions reveals that the let-7 microRNA targets Dicer within its coding sequence. Proc Natl Acad Sci USA, 2008, 105: 14879–14884
Duursma A M, Kedde M, Schrier M, et al. miR-148 targets human DNMT3b protein coding region. RNA, 2008, 14: 872–877
Miranda K C, Huynh T, Tay Y, et al. A pattern-based method for the identification of MicroRNA binding sites and their corresponding heteroduplexes. Cell, 2006, 126: 1203–1217
Eulalio A, Huntzinger E, Izaurralde E. Getting to the root of miRNA-mediated gene silencing. Cell, 2008, 132: 9–14
Chen P S, Su J L, Cha S T, et al. miR-107 promotes tumor progression by targeting the let-7 microRNA in mice and humans. J Clin Invest, 2011, 121: 3442–3455
Tang R, Li L, Zhu D, et al. Mouse miRNA-709 directly regulates miRNA-15a/16-1 biogenesis at the posttranscriptional level in the nucleus: evidence for a microRNA hierarchy system. Cell Res, 2011, in press
Bhattacharyya S N, Habermacher R, Martine U, et al. Relief of microRNA-mediated translational repression in human cells subjected to stress. Cell, 2006, 125: 1111–1124
Kim H H, Kuwano Y, Srikantan S, et al. HuR recruits let-7/RISC to repress c-Myc expression. Genes Dev, 2009, 23: 1743–1748
Lagos-Quintana M, Rauhut R, Yalcin A, et al. Identification of tissue-specific microRNAs from mouse. Curr Biol, 2002, 12: 735–739
Bak M, Silahtaroglu A, Moller M, et al. microRNA expression in the adult mouse central nervous system. RNA, 2008, 14: 432–444
Miska E A, Alvarez-Saavedra E, Townsend M, et al. Microarray analysis of microRNA expression in the developing mammalian brain. Genome Biol, 2004, 5: R68
Lau P, Verrier J D, Nielsen J A, et al. Identification of dynamically regulated microRNA and mRNA networks in developing oligodendrocytes. J Neurosci, 2008, 28: 11720–11730
Dugas J C, Cuellar T L, Scholze A, et al. Dicer1 and miR-219 Are required for normal oligodendrocyte differentiation and myelination. Neuron, 2010, 65: 597–611
Zhao X, He X, Han X, et al. MicroRNA-mediated control of oligodendrocyte differentiation. Neuron, 2010, 65: 612–626
Fiore R, Khudayberdiev S, Christensen M, et al. Mef2-mediated transcription of the miR379-410 cluster regulates activity-dependent dendritogenesis by fine-tuning Pumilio2 protein levels. EMBO J, 2009, 28: 697–710
Kye M J, Liu T, Levy S F, et al. Somatodendritic microRNAs identified by laser capture and multiplex RT-PCR. RNA, 2007, 13: 1224–1234
Lugli G, Torvik V I, Larson J, et al. Expression of microRNAs and their precursors in synaptic fractions of adult mouse forebrain. J Neurochem, 2008, 106: 650–661
Bernstein E, Kim S Y, Carmell M A, et al. Dicer is essential for mouse development. Nat Genet, 2003, 35: 215–217
De Pietri T D, Pulvers J N, Haffner C, et al. miRNAs are essential for survival and differentiation of newborn neurons but not for expansion of neural progenitors during early neurogenesis in the mouse embryonic neocortex. Development, 2008, 135: 3911–3921
Kawase-Koga Y, Otaegi G, Sun T. Different timings of Dicer deletion affect neurogenesis and gliogenesis in the developing mouse central nervous system. Dev Dyn, 2009, 238: 2800–2812
Kim J, Inoue K, Ishii J, et al. A microRNA feedback circuit in midbrain dopamine neurons. Science, 2007, 317: 1220–1224
Schaefer A, O’Carroll D, Tan C L, et al. Cerebellar neurodegeneration in the absence of microRNAs. J Exp Med, 2007, 204: 1553–1558
Davis T H, Cuellar T L, Koch S M, et al. Conditional loss of Dicer disrupts cellular and tissue morphogenesis in the cortex and hippocampus. J Neurosci, 2008, 28: 4322–4330
Stark K L, Xu B, Bagchi A, et al. Altered brain microRNA biogenesis contributes to phenotypic deficits in a 22q11-deletion mouse model. Nat Genet, 2008, 40: 751–760
Fenelon K, Mukai J, Xu B, et al. Deficiency of Dgcr8, a gene disrupted by the 22q11.2 microdeletion, results in altered short-term plasticity in the prefrontal cortex. Proc Natl Acad Sci USA, 2011, 108: 4447–4452
Schofield C M, Hsu R, Barker A J, et al. Monoallelic deletion of the microRNA biogenesis gene Dgcr8 produces deficits in the development of excitatory synaptic transmission in the prefrontal cortex. Neural Dev, 2011, 6: 11
Baumann N, Pham-Dinh D. Biology of oligodendrocyte and myelin in the mammalian central nervous system. Physiol Rev, 2001, 81: 871–927
Shin D, Shin J Y, McManus M T, et al. Dicer ablation in oligodendrocytes provokes neuronal impairment in mice. Ann Neurol, 2009, 66: 843–857
Pereira J A, Baumann R, Norrmen C, et al. Dicer in Schwann cells is required for myelination and axonal integrity. J Neurosci, 2010, 30: 6763–6775
Yun B, Anderegg A, Menichella D, et al. MicroRNA-deficient Schwann cells display congenital hypomyelination. J Neurosci, 2010, 30: 7722–7728
Tao J, Wu H, Lin Q, et al. Deletion of astroglial Dicer causes non-cell-autonomous neuronal dysfunction and degeneration. J Neurosci, 2011, 31: 8306–8319
Makeyev E V, Zhang J, Carrasco M A, et al. The MicroRNA miR-124 promotes neuronal differentiation by triggering brain-specific alternative pre-mRNA splicing. Mol Cell, 2007, 27: 435–448
Yoo A S, Staahl B T, Chen L, et al. MicroRNA-mediated switching of chromatin-remodelling complexes in neural development. Nature, 2009, 460: 642–646
Yoo A S, Sun A X, Li L, et al. MicroRNA-mediated conversion of human fibroblasts to neurons. Nature, 2011, 476: 228–231
Lugli G, Larson J, Martone M E, et al. Dicer and eIF2c are enriched at postsynaptic densities in adult mouse brain and are modified by neuronal activity in a calpain-dependent manner. J Neurochem, 2005, 94: 896–905
Siegel G, Obernosterer G, Fiore R, et al. A functional screen implicates microRNA-138-dependent regulation of the depalmitoylation enzyme APT1 in dendritic spine morphogenesis. Nat Cell Biol, 2009, 11: 705–716
Cuellar T L, Davis T H, Nelson P T, et al. Dicer loss in striatal neurons produces behavioral and neuroanatomical phenotypes in the absence of neurodegeneration. Proc Natl Acad Sci USA, 2008, 105: 5614–5619
Haramati S, Chapnik E, Sztainberg Y, et al. miRNA malfunction causes spinal motor neuron disease. Proc Natl Acad Sci USA, 2010, 107: 13111–13116
Kawase-Koga Y, Low R, Otaegi G, et al. RNAase-III enzyme Dicer maintains signaling pathways for differentiation and survival in mouse cortical neural stem cells. J Cell Sci, 2010, 123: 586–594
Hebert S S, Papadopoulou A S, Smith P, et al. Genetic ablation of Dicer in adult forebrain neurons results in abnormal tau hyperphosphorylation and neurodegeneration. Hum Mol Genet, 2010, 19: 3959–3969
Konopka W, Kiryk A, Novak M, et al. MicroRNA loss enhances learning and memory in mice. J Neurosci, 2010, 30: 14835–14842
Damiani D, Alexander J J, O’Rourke J R, et al. Dicer inactivation leads to progressive functional and structural degeneration of the mouse retina. J Neurosci, 2008, 28: 4878–4887
Georgi S A, Reh T A. Dicer is required for the transition from early to late progenitor state in the developing mouse retina. J Neurosci, 2010, 30: 4048–4061
Iida A, Shinoe T, Baba Y, et al. Dicer plays essential roles for retinal development by regulation of survival and differentiation. Invest Ophthalmol Vis Sci, 2011, 52: 3008–3017
Friedman L M, Dror A A, Mor E, et al. MicroRNAs are essential for development and function of inner ear hair cells in vertebrates. Proc Natl Acad Sci USA, 2009, 106: 7915–7920
Choi P S, Zakhary L, Choi W Y, et al. Members of the miRNA-200 family regulate olfactory neurogenesis. Neuron, 2008, 57: 41–55
Zheng K, Li H, Zhu Y, et al. MicroRNAs are essential for the developmental switch from neurogenesis to gliogenesis in the developing spinal cord. J Neurosci, 2010, 30: 8245–8250
Budde H, Schmitt S, Fitzner D, et al. Control of oligodendroglial cell number by the miR-17-92 cluster. Development, 2010, 137: 2127–2132
Vo N, Klein M E, Varlamova O, et al. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc Natl Acad Sci USA, 2005, 102: 16426–16431
Obernosterer G, Leuschner P J, Alenius M, et al. Post-transcriptional regulation of microRNA expression. RNA, 2006, 12: 1161–1167
Kedde M, Agami R. Interplay between microRNAs and RNA-binding proteins determines developmental processes. Cell Cycle, 2008, 7: 899–903
Muddashetty R S, Nalavadi V C, Gross C, et al. Reversible inhibition of PSD-95 mRNA translation by miR-125a, FMRP phosphorylation, and mGluR signaling. Mol Cell, 2011, 42: 673–688
Wayman G A, Davare M, Ando H, et al. An activity-regulated microRNA controls dendritic plasticity by down-regulating p250GAP. Proc Natl Acad Sci USA, 2008, 105: 9093–9098
Schratt G M, Tuebing F, Nigh E A, et al. A brain-specific microRNA regulates dendritic spine development. Nature, 2006, 439: 283–289
Lau P, Verrier J D, Nielsen J A, et al. Identification of dynamically regulated microRNA and mRNA networks in developing oligodendrocytes. J Neurosci, 2008, 28: 11720–11730
Hart I K, Richardson W D, Raff M C. PDGF increases the expression of Fos and Jun in newly formed oligodendrocytes that have become resistant to the mitogenic effect of PDGF. Glia, 1992, 6: 310–313
Kleihues P, Louis D N, Scheithauer B W. World Health Organization Classification of Tumours of the Nervous System. 2000
Roussel M F, Hatten M E. Cerebellum development and medulloblastoma. Curr Top Dev Biol, 2011, 94: 235–282
Turner J D, Williamson R, Almefty K K, et al. The many roles of microRNAs in brain tumor biology. Neurosurg Focus, 2010, 28: E3
Chen Y, Liu W, Chao T, et al. MicroRNA-21 down-regulates the expression of tumor suppressor PDCD4 in human glioblastoma cell T98G. Cancer Lett, 2008, 272: 197–205
Papagiannakopoulos T, Shapiro A, Kosik K S. MicroRNA-21 targets a network of key tumor-suppressive pathways in glioblastoma cells. Cancer Res, 2008, 68: 8164–8172
Gabriely G, Wurdinger T, Kesari S, et al. MicroRNA 21 promotes glioma invasion by targeting matrix metalloproteinase regulators. Mol Cell Biol, 2008, 28: 5369–5380
Li Y, Li W, Yang Y, et al. MicroRNA-21 targets LRRFIP1 and contributes to VM-26 resistance in glioblastoma multiforme. Brain Res, 2009, 1286: 13–18
Kwak H J, Kim Y J, Chun K R, et al. Downregulation of Spry2 by miR-21 triggers malignancy in human gliomas. Oncogene, 2011, 30: 2433–2442
Grunder E, D’Ambrosio R, Fiaschetti G, et al. MicroRNA-21 suppression impedes medulloblastoma cell migration. Eur J Cancer, 2011, in press
Zhang C Z, Zhang J X, Zhang A L, et al. MiR-221 and miR-222 target PUMA to induce cell survival in glioblastoma. Mol Cancer, 2010, 9: 229
Quintavalle C, Garofalo M, Zanca C, et al. miR-221/222 overexpression in human glioblastoma increases invasiveness by targeting the protein phosphate PTPmu. Oncogene, 2011, in press
Gillies J K, Lorimer I A. Regulation of p27Kip1 by miRNA 221/222 in glioblastoma. Cell Cycle, 2007, 6: 2005–2009
Ciafre S A, Galardi S, Mangiola A, et al. Extensive modulation of a set of microRNAs in primary glioblastoma. Biochem Biophys Res Commun, 2005, 334: 1351–1358
Lukiw W J, Cui J G, Li Y Y, et al. Up-regulation of micro-RNA-221 (miRNA-221; chr Xp11.3) and caspase-3 accompanies down-regulation of the survivin-1 homolog BIRC1 (NAIP) in glioblastoma multiforme (GBM). J Neurooncol, 2009, 91: 27–32
Ernst A, Campos B, Meier J, et al. De-repression of CTGF via the miR-17-92 cluster upon differentiation of human glioblastoma spheroid cultures. Oncogene, 2010, 29: 3411–3422
Uziel T, Karginov F V, Xie S, et al. The miR-17∼92 cluster collaborates with the Sonic Hedgehog pathway in medulloblastoma. Proc Natl Acad Sci USA, 2009, 106: 2812–2817
Northcott P A, Fernandez-L A, Hagan J P, et al. The miR-17/92 polycistron is up-regulated in sonic hedgehog-driven medulloblastomas and induced by N-myc in sonic hedgehog-treated cerebellar neural precursors. Cancer Res, 2009, 69: 3249–3255
Sasayama T, Nishihara M, Kondoh T, et al. MicroRNA-10b is overexpressed in malignant glioma and associated with tumor invasive factors, uPAR and RhoC. Int J Cancer, 2009, 125: 1407–1413
Sun L, Yan W, Wang Y, et al. MicroRNA-10b induces glioma cell invasion by modulating MMP-14 and uPAR expression via HOXD10. Brain Res, 2011, 1389: 9–18
Gabriely G, Yi M, Narayan R S, et al. Human glioma growth is controlled by microRNA-10b. Cancer Res, 2011, 71: 3563–3572
Huse J T, Brennan C, Hambardzumyan D, et al. The PTEN-regulating microRNA miR-26a is amplified in high-grade glioma and facilitates gliomagenesis in vivo. Genes Dev, 2009, 23: 1327–1337
Kim H, Huang W, Jiang X, et al. Integrative genome analysis reveals an oncomir/oncogene cluster regulating glioblastoma survivorship. Proc Natl Acad Sci USA, 2010, 107: 2183–2188
Weeraratne S D, Amani V, Neiss A, et al. miR-34a confers chemosensitivity through modulation of MAGE-A and p53 in medulloblastoma. Neuro Oncol, 2011, 13: 165–175
Luan S, Sun L, Huang F. MicroRNA-34a: a novel tumor suppressor in p53-mutant glioma cell line U251. Arch Med Res, 2010, 41: 67–74
Li Y, Guessous F, Zhang Y, et al. MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res, 2009, 69: 7569–7576
Guessous F, Zhang Y, Kofman A, et al. microRNA-34a is tumor suppressive in brain tumors and glioma stem cells. Cell Cycle, 2010, 9: 1031–1036
Yang G, Zhang R, Chen X, et al. MiR-106a inhibits glioma cell growth by targeting E2F1 independent of p53 status. J Mol Med (Berl), 2011, 89: 1037–1050
Pierson J, Hostager B, Fan R, et al. Regulation of cyclin dependent kinase 6 by microRNA 124 in medulloblastoma. J Neurooncol, 2008, 90: 1–7
Li K K, Pang J C, Ching A K, et al. miR-124 is frequently down-regulated in medulloblastoma and is a negative regulator of SLC16A1. Hum Pathol, 2009, 40: 1234–1243
Silber J, Lim D A, Petritsch C, et al. miR-124 and miR-137 inhibit proliferation of glioblastoma multiforme cells and induce differentiation of brain tumor stem cells. BMC Med, 2008, 6: 14
Zhang Y, Chao T, Li R, et al. MicroRNA-128 inhibits glioma cells proliferation by targeting transcription factor E2F3a. J Mol Med (Berl), 2009, 87: 43–51
Cui J G, Zhao Y, Sethi P, et al. Micro-RNA-128 (miRNA-128) down-regulation in glioblastoma targets ARP5 (ANGPTL6), Bmi-1 and E2F-3a, key regulators of brain cell proliferation. J Neurooncol, 2010, 98: 297–304
Venkataraman S, Alimova I, Fan R, et al. MicroRNA 128a increases intracellular ROS level by targeting Bmi-1 and inhibits medulloblastoma cancer cell growth by promoting senescence. PLoS ONE, 2010, 5: e10748
Xia H F, He T Z, Liu C M, et al. MiR-125b expression affects the proliferation and apoptosis of human glioma cells by targeting Bmf. Cell Physiol Biochem, 2009, 23: 347–358
Shi L, Zhang J, Pan T, et al. MiR-125b is critical for the suppression of human U251 glioma stem cell proliferation. Brain Res, 2010, 1312: 120–126
Garzia L, Andolfo I, Cusanelli E, et al. MicroRNA-199b-5p impairs cancer stem cells through negative regulation of HES1 in medulloblastoma. PLoS ONE, 2009, 4: e4998
Ciafre S A, Galardi S, Mangiola A, et al. Extensive modulation of a set of microRNAs in primary glioblastoma. Biochem Biophys Res Commun, 2005, 334: 1351–1358
Ferretti E, De Smaele E, Po A, et al. MicroRNA profiling in human medulloblastoma. Int J Cancer, 2009, 124: 568–577
Corsten M F, Miranda R, Kasmieh R, et al. MicroRNA-21 knockdown disrupts glioma growth in vivo and displays synergistic cytotoxicity with neural precursor cell delivered S-TRAIL in human gliomas. Cancer Res, 2007, 67: 8994–9000
Zhou X, Ren Y, Moore L, et al. Downregulation of miR-21 inhibits EGFR pathway and suppresses the growth of human glioblastoma cells independent of PTEN status. Lab Invest, 2010, 90: 144–155
Ren Y, Zhou X, Mei M, et al. MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer, 2010, 10: 27
Zhou X, Zhang J, Jia Q, et al. Reduction of miR-21 induces glioma cell apoptosis via activating caspase 9 and 3. Oncol Rep, 2010, 24: 195–201
Fineberg S K, Kosik K S, Davidson B L. MicroRNAs potentiate neural development. Neuron, 2009, 64: 303–309
Jin P, Zarnescu D C, Ceman S, et al. Biochemical and genetic interaction between the fragile X mental retardation protein and the microRNA pathway. Nat Neurosci, 2004, 7: 113–117
Edbauer D, Neilson J R, Foster K A, et al. Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron, 2010, 65: 373–384
Xu X L, Li Y, Wang F, et al. The steady-state level of the nervous-system-specific microRNA-124a is regulated by dFMR1 in Drosophila. J Neurosci, 2008, 28: 11883–11889
Samaco R C, Neul J L. Complexities of Rett syndrome and MeCP2. J Neurosci, 2011, 31: 7951–7959
Klein M E, Lioy D T, Ma L, et al. Homeostatic regulation of MeCP2 expression by a CREB-induced microRNA. Nat Neurosci, 2007, 10: 1513–1514
Urdinguio R G, Fernandez A F, Lopez-Nieva P, et al. Disrupted microRNA expression caused by Mecp2 loss in a mouse model of Rett syndrome. Epigenetics, 2010, 5: 656–663
Wu H, Tao J, Chen P J, et al. Genome-wide analysis reveals methyl-CpG-binding protein 2-dependent regulation of microRNAs in a mouse model of Rett syndrome. Proc Natl Acad Sci USA, 2010, 107: 18161–18166
Lees A J, Hardy J, Revesz T. Parkinson’s disease. Lancet, 2009, 373: 2055–2066
Junn E, Lee K W, Jeong B S, et al. Repression of alpha-synuclein expression and toxicity by microRNA-7. Proc Natl Acad Sci USA, 2009, 106: 13052–13057
Doxakis E. Post-transcriptional regulation of alpha-synuclein expression by mir-7 and mir-153. J Biol Chem, 2010, 285: 12726–12734
Cogswell J P, Ward J, Taylor I A, et al. Identification of miRNA changes in Alzheimer’s disease brain and CSF yields putative biomarkers and insights into disease pathways. J Alzheimers Dis, 2008, 14: 27–41
Wang W X, Rajeev B W, Stromberg A J, et al. The expression of microRNA miR-107 decreases early in Alzheimer’s disease and may accelerate disease progression through regulation of beta-site amyloid precursor protein-cleaving enzyme 1. J Neurosci, 2008, 28: 1213–1223
Hebert S S, Horre K, Nicolai L, et al. Loss of microRNA cluster miR-29a/b-1 in sporadic Alzheimer’s disease correlates with increased BACE1/beta-secretase expression. Proc Natl Acad Sci USA, 2008, 105: 6415–6420
Boissonneault V, Plante I, Rivest S, et al. MicroRNA-298 and microRNA-328 regulate expression of mouse beta-amyloid precursor protein-converting enzyme 1. J Biol Chem, 2009, 284: 1971–1981
Hebert S S, Horre K, Nicolai L, et al. MicroRNA regulation of Alzheimer’s Amyloid precursor protein expression. Neurobiol Dis, 2009, 33: 422–428
Vilardo E, Barbato C, Ciotti M, et al. MicroRNA-101 regulates amyloid precursor protein expression in hippocampal neurons. J Biol Chem, 2010, 285: 18344–18351
Liu W, Liu C, Zhu J, et al. MicroRNA-16 targets amyloid precursor protein to potentially modulate Alzheimer’s-associated pathogenesis in SAMP8 mice. Neurobiol Aging, 2010
Long J M, Lahiri D K. MicroRNA-101 downregulates Alzheimer’s amyloid-beta precursor protein levels in human cell cultures and is differentially expressed. Biochem Biophys Res Commun, 2011, 404: 889–895
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Feng, W., Feng, Y. MicroRNAs in neural cell development and brain diseases. Sci. China Life Sci. 54, 1103–1112 (2011). https://doi.org/10.1007/s11427-011-4249-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4249-8