Abstract
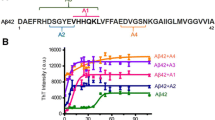
β-amyloid (Aβ) and copper play important roles in the pathogenesis of Alzheimer’s disease (AD). However, the behavioral correlativity and molecular mechanisms of Aβ and copper toxicity have been investigated less often. In the present study, we investigated the interaction and toxicity of Aβ1–42 and copper in the Aβ1–42 transgenic Caenorhabditis elegans worm model CL2006. Our data show that the paralysis behavior of CL2006 worms significantly deteriorated after exposure to 10−3 mol L−1 copper ions. However, the paralysis behavior was dramatically attenuated with exposure to 10−4 mol L−1 copper ions. The exogenous copper treatment also partially changed the homeostatic balance of zinc, manganese, and iron. Our data suggest that the accumulation of reactive oxygen species (ROS) was responsible for the paralysis induced by Aβ and copper in CL2006. The ROS generation induced by Aβ and copper appear to be through sod-1, prdx-2, skn-1, hsp-60 and hsp-16.2 genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mattson M P. Pathways towards and away from Alzheimer’s disease. Nature, 2004, 430: 631–639 15295589, 10.1038/nature02621, 1:CAS:528:DC%2BD2cXmt1Gis7o%3D
Goedert M, Spillantini M G. A century of Alzheimer’s disease. Science, 2006, 314: 777–781 17082447, 10.1126/science.1132814, 1:CAS:528:DC%2BD28XhtFKit73K
Bush A I. The metallobiology of Alzheimer’s disease. Trends Neurosci, 2003, 26: 207–214 12689772, 10.1016/S0166-2236(03)00067-5, 1:CAS:528:DC%2BD3sXis12nsb8%3D
Bush A I, Curtain C C. Twenty years of metallo-neurobiology: Where to now? Eur Biophys J, 2007, 37: 241–245 17994233, 10.1007/s00249-007-0228-1
Cater M A, McInnes K T, Li Q X, et al. Intracellular copper deficiency increases amyloid-β secretion by diverse mechanisms. Biochem J, 2008, 412: 141–152 18248325, 10.1042/BJ20080103, 1:CAS:528:DC%2BD1cXltVykurg%3D
Crouch P J, Hung L W, Adlard P A, et al. Increasing Cu bioavailability inhibits Aβ oligomers and tau phosphorylation. Proc Natl Acad Sci USA, 2009, 106: 381–386 19122148, 10.1073/pnas.0809057106, 1:CAS:528:DC%2BD1MXhtVels7Y%3D
Markesbery W R. Oxidative stress hypothesis in Alzheimer’s disease. Free Radical Bio Med, 2007, 23: 134–147 10.1016/S0891-5849(96)00629-6
Sparks D L, Schreurs B G. Trace amounts of copper in water induce β-amyloid plaques and learning deficits in a rabbit model of Alzheimer’s disease. Proc Natl Acad Sci USA, 2003, 100: 11065–11069 12920183, 10.1073/pnas.1832769100, 1:CAS:528:DC%2BD3sXnslynu7s%3D
Phinney A L, Drisaldi B, Schmidt S D, et al. In vivo reduction of amyloid-β by a mutant copper transporter. Proc Natl Acad Sci USA, 2003, 100: 14193–14198 14617772, 10.1073/pnas.2332851100, 1:CAS:528:DC%2BD3sXpsFGitbk%3D
Bayer T A, Schäfer S, Simons A, et al. Dietary Cu stabilizes brain superoxide dismutase 1 activity and reduces amyloid Aβ production in APP23 transgenic mice. Proc Natl Acad Sci USA, 2003, 100: 14187–14192 14617773, 10.1073/pnas.2332818100, 1:CAS:528:DC%2BD3sXpsFGitbg%3D
Link C D. Invertebrate models of Alzheimer’s disease. Genes Brain Behav, 2005, 4: 147–156 15810903, 10.1111/j.1601-183X.2004.00105.x, 1:STN:280:DC%2BD2M7nvFSqtQ%3D%3D
Link C D. C. elegans models of age-associated neurodegenerative diseases: Lessons from transgenic worm models of Alzheimer’s disease. Exp Gerontol, 2006, 41: 1007–1013 16930903, 10.1016/j.exger.2006.06.059, 1:CAS:528:DC%2BD28Xht1KrsLrE
Brenner S. The genetics of Caenorhabditis elegans. Genetics, 1974, 77: 71–94 4366476, 1:STN:280:DyaE2c3ntFWlsw%3D%3D
Feng Z, Li W, Ward A, et al. A C. elegans model of nicotine-dependent behavior: Regulation by TRP-family channels. Cell, 2006, 127: 621–633 17081982, 10.1016/j.cell.2006.09.035, 1:CAS:528:DC%2BD28Xht1Wgtb%2FF
Cohen E, Bieschke J, Perciavalle R M, et al. Opposing activities protect against age-onset proteotoxicity. Science, 2006, 313: 1604–1610 16902091, 10.1126/science.1124646, 1:CAS:528:DC%2BD28XpsVOktbk%3D
Gao Y X, Liu N Q, Chen C Y, et al. Mapping technique for biodistribution of elements in a model organism, Caenorhabditis elegans, after exposure to copper nanoparticles with microbeam synchrotron radiation X-ray fluorescence. J Anal At Spectrom, 2008, 23: 1121–1124 10.1039/b802338g, 1:CAS:528:DC%2BD1cXptVCnu78%3D
Schulz T J, Zarse K, Voigt A, et al. Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab, 2007, 6: 280–293 17908557, 10.1016/j.cmet.2007.08.011, 1:CAS:528:DC%2BD2sXht1Sjsr3J
Hoogewijs D, Houthoofd K, Matthijssens F, et al. Selection and validation of a set of reliable reference genes for quantitative sod gene expression analysis in C. elegans. BMC Mol Biol, 2008, 9: 9 18211699, 10.1186/1471-2199-9-9
Praticò D. Oxidative stress hypothesis in Alzheimer’s disease: A reappraisal. Trends Pharmacol Sci, 2008, 29: 609–615 18838179, 10.1016/j.tips.2008.09.001
Giglio A M, Hunter T, Bannister J V, et al. The copper/zinc superoxide dismutase gene of Caenorhabditis elegans. Biochem Mol Biol Int, 1994, 33: 41–44 8081212, 1:CAS:528:DyaK2cXlt1Kmsbw%3D
Hunter T, Bannister W H, Hunter G J. Cloning, expression, and characterization of two manganese superoxide dismutases from Caenorhabditis elegans. J Biol Chem, 1997, 272: 28652–28659 9353332, 10.1074/jbc.272.45.28652, 1:CAS:528:DyaK2sXntlCqsb0%3D
Taub J, Lau J F, Ma C, et al. A cytosolic catalase is needed to extend adult lifespan in C. elegans daf-C and clk-1 mutants. Nature, 1999, 399: 162–166 10335847, 10.1038/20208, 1:CAS:528:DyaK1MXjsVOhtLs%3D
Isermann K, Liebau E, Roeder T, et al. A peroxiredoxin specifically expressed in two types of pharyngeal neurons is required for normal growth and egg production in Caenorhabditis elegans. J Mol Biol, 2004, 338: 745–755 15099742, 10.1016/j.jmb.2004.03.021, 1:CAS:528:DC%2BD2cXjtFOhu7s%3D
Simonetta S H, Romanowski A, Minniti A N, et al. Circadian stress tolerance in adult Caenorhabditis elegans. J Comp Physiol A Neuroethol Sens Neural Behav Physiol, 2008, 194: 821–828 18648821, 10.1007/s00359-008-0353-z, 1:CAS:528:DC%2BD1cXhtVahu77F
An J H, Vranas K, Lucke M, et al. Regulation of the Caenorhabditis elegans oxidative stress defense protein SKN-1 by glycogen synthase kinase-3. Proc Natl Acad Sci USA, 2005, 102: 16275–16280 16251270, 10.1073/pnas.0508105102, 1:CAS:528:DC%2BD2MXht1Cgur3F
Steinkraus K A, Smith E D, Davis C, et al. Dietary restriction suppresses proteotoxicity and enhances longevity by an hsf-1-dependent mechanism in Caenorhabditis elegans. Aging Cell, 2008, 7: 394–404 18331616, 10.1111/j.1474-9726.2008.00385.x, 1:CAS:528:DC%2BD1cXntFWnt7w%3D
Benedetti C, Haynes C M, Yang Y, et al. Ubiquitin-like protein 5 positively regulates chaperone gene expression in the mitochondrial unfolded protein response. Genetics, 2006, 174: 229–239 16816413, 10.1534/genetics.106.061580, 1:CAS:528:DC%2BD28XhtFGgsrnE
Hong M, Kwon J Y, Shim J, et al. Differential hypoxia response of hsp-16 genes in the nematode. J Mol Biol, 2004, 344: 369–381 15522291, 10.1016/j.jmb.2004.09.077, 1:CAS:528:DC%2BD2cXptFOns74%3D
Good P F, Werner P, Hsu A, et al. Evidence for neuronal oxidative damage in Alzheimer’s disease. Am J Pathol, 1996, 149: 21 8686745, 1:STN:280:DyaK283nsFGntw%3D%3D
Markesbery W R, Carney J M. Oxidative alterations in Alzheimer’s disease. Brain Pathology, 1999, 9: 133–146 9989456, 10.1111/j.1750-3639.1999.tb00215.x, 1:CAS:528:DyaK1MXht1Cmsbw%3D
Smith M A, Rottkamp C A, Nunomura A, et al. Oxidative stress in Alzheimer’s disease. Biochim Biophys Acta-Mol Basis Dis, 2000, 1502: 139–144 1:CAS:528:DC%2BD3cXkvFSgur8%3D
Zhu X, Raina A K, Perry G, et al. Alzheimer’s disease: The two-hit hypothesis. Lancet Neurol, 2004, 3: 219–226 15039034, 10.1016/S1474-4422(04)00707-0, 1:CAS:528:DC%2BD2cXjvFyisbo%3D
Perry G, Nunomura A, Hirai K, et al. Is oxidative damage the fundamental pathogenic mechanism of Alzheimer’s and other neurodegenerative diseases? Free Radical Bio Med, 2002, 11: 1475–1479 10.1016/S0891-5849(02)01113-9
Lovell M A, Robertson J D, Teesdale W J, et al. Copper, iron and zinc in Alzheimer’s disease senile plaques. J Neurol Sci, 1998, 158: 47–52 9667777, 10.1016/S0022-510X(98)00092-6, 1:CAS:528:DyaK1cXjsVentbo%3D
Huang X, Cuajungco M P, Atwood C S, et al. Cu(II) potentiation of Alzheimer Aβ neurotoxicity. Correlation with cell-free hydrogen peroxide production and metal reduction. J Biol Chem, 1999, 274: 37111–37116 1:CAS:528:DC%2BD3cXpsFSi
Quinn J F, Crane S, Harris C, et al. Copper in Alzheimer’s disease: Too much or too little? Expert Rev Neurother, 2009, 9: 631–637 19402774, 10.1586/ern.09.27, 1:CAS:528:DC%2BD1MXlt1ClsLg%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Luo, Y., Zhang, J., Liu, N. et al. Copper ions influence the toxicity of β-amyloid(1–42) in a concentration-dependent manner in a Caenorhabditis elegans model of Alzheimer’s disease. Sci. China Life Sci. 54, 527–534 (2011). https://doi.org/10.1007/s11427-011-4180-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4180-z