Abstact
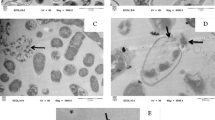
The effects of melittin on growth and bacteriostasis of four pathogens were extensively investigated using scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The results indicated that the melittin had a marked bacteriostatic effect on the four pathogenic bacteria. Among these, E. cacotowora was influenced most powerfully and quickly, the yeast and F. fulva were the second, and the S. aureus was inhibited by a low concentration but was killed by a high concentration. It was observed in the experiments that melittin killed pathogenic bacteria in three ways. One was by pore formation. After integrating melittin into the plasma membrane, a vacuole was formed then penetrated, resulting in bacterial content leakage. The vacuole also experienced plasmolysis and the growing cavity destroyed the membrane. A third effect was the formation of vacuoles in the cells which induced the pycnosis of the cytoplasm resulting in a cell death. The mechanism of melittin bacteriostasis was the result of integrating melittin with phospholipod double layers of the plasmalemma and the endomembranes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Boman H G, Agerbeth B, Boman A. Mechanisms of action on Escherichia coli of cecropinPI and PR-39, two antibacterial peptides from pig intestine. Infect Immun, 1993, 61: 2978–2984, 1:CAS:528:DyaK3sXlsFyitLc%3D, 8514403
Fennell J F, Shipman W H, Cole L J. Antibacterial action of a bee venom fraction (melittin) against a penicillin-resistant staphylococcus and other microorganisms. Res Dev Tech Rep, 1967, 1967: 1–13
Zasloff M. Antimicrobial peptides of multicellular organisms. Nature, 2002, 415: 389–395, 1:CAS:528:DC%2BD38XhtVCgsLs%3D, 10.1038/415389a, 11807545
Matsuzaki K. Why and how are peptide-lipid interactions utilized for self-defense? Magainins and tachyplesins as archetypes. Biochim Biophys Acta, 1999, 1462: 1–10, 1:CAS:528:DyaK1MXnslymuro%3D, 10.1016/S0005-2736(99)00197-2, 10590299
Yang L, Weiss T M, Lehrer R I, et al. Crystallization of antimicrobial pores in membranes: magainin and protegrin. Biophys J, 2000, 79: 2002–2009, 1:CAS:528:DC%2BD3cXnt12ht7s%3D, 10.1016/S0006-3495(00)76448-4, 11023904
Westerhoff H V, Juretic D, Hendler R W, et al. Magainins and the disruption of membrane-linked free-energy transduction. Proc Natl Acad Sci USA, 1989, 86: 6597–6601, 1:CAS:528:DyaL1MXmt1Onsb0%3D, 10.1073/pnas.86.17.6597, 2671997
Bierbaum G, Sahl H G. Induction of autolysis of Staphylocci by the basic peptide antibiotics pep5 and nisin and their influence on the activity of autolytic enzymes. Arch Microbiol, 1985, 141: 249–254, 1:CAS:528:DyaL2MXktlCktL8%3D, 10.1007/BF00408067, 4004448
Kragol G. The antibacterial peptide pyrrhocoricin inhibits the ATPase actions of DnaK and prevents chaperone-assisted protein folding. Biochem, 2001, 40: 3016–3026, 1:CAS:528:DC%2BD3MXhtVygurs%3D, 10.1021/bi002656a
Pan L Z, Na J, Xing Z, et al. Inhibiting effect of melittin on pathogens of crops.Chinese Sci Bull, 2007, 52: 639–644, 1:CAS:528:DC%2BD2sXisV2nsrk%3D, 10.1007/s11434-007-0117-0
Tang X M, Tai S S. ESEM observation for biological samples. J Chinese electr micros soc, 2001, 20: 217–223
Yang L, Weiss T M, Lehrer R I. Crystallization of antimicrobial pores in membranes:magainin and protegrin. Biophys J, 2000, 79: 2002–2009, 1:CAS:528:DC%2BD3cXnt12ht7s%3D, 10.1016/S0006-3495(00)76448-4, 11023904
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wang, G., Na, J., Pan, L. et al. Ultrastructural observation on sterilization of melittin. Sci. China Life Sci. 54, 166–170 (2011). https://doi.org/10.1007/s11427-010-4118-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-010-4118-x