Abstract
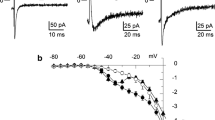
The purpose of the present study was to further study the characteristics and regulation of spontaneous transient outward currents (STOCs) in freshly isolated porcine coronary artery smooth muscle cells (ASMCs). STOCs were recorded using the perforated whole-cell patch-clamp configuration. STOCs were voltage-dependent and superimposed stochastically onto whole-cell Ca2+-activated-K+ (BKCa) currents. Charybdotoxin (ChTX, 200 nmol/L), a selective blocker of BKCa channels, completely inhibited STOCs within 10 min. STOCs activity was greatly suppressed when extracellular Ca2+ concentration decreased from 1.8 mmol/L to 200 nmol/L, further removal of Ca2+ abolished STOCs activity. Ca2+ ionophore A23187 (10 μmol/L) increased STOCs activity significantly. Verapamil (20 μmol/L) and CdCl2 (200 μmol/L), two kinds of organic L-type voltage-dependent Ca2+ channels (L-VDCCs) antagonists, had little effect on STOCs. In addition, the ryanodine receptors (RyRs) agonist caffeine (5 mmol/L) significantly activated STOCs. Application of ryanodine (50 μmol/L) to block RyRs abolished STOCs, subsequent washout of ryanodine or application of caffeine failed to reproduce STOCs activity. Inhibition of inositol 1,4,5-trisphosphate receptors (IP3Rs) by 2APB (40 μmol/L) greatly suppressed the activity of STOCs, application of caffeine (5 mmol/L) in the presence of 2APB caused a burst of outward currents followed by inhibition of STOCs. These results suggest that STOCs in porcine coronary ASMCs are mediated by BKCa channels. Extracellular Ca2+ is essential for STOCs activity, while Ca2+ entry through L-VDCCs has little effect on STOCs. Intracellular Ca2+ release induced by RyRs is responsible for the regulation of STOCs, whereas IP3Rs might also be involved.
Similar content being viewed by others
References
Ledoux J, Werner M E, Brayden J E, et al. Calcium-activated potassium channels and the regulation of vascular tone. Physiology, 2006, 21(1): 69–78
Wellman G C, Nelson M T. Signaling between SR and plasmalemma in smooth muscle: Sparks and the activation of Ca2+-sensitive ion channels. Cell Calcium, 2003, 34(3): 211–229
Jaggar J H, Porter V A, Lederer W J, et al. Calcium sparks in smooth muscle. Am J Physiol Cell Physiol, 2000, 278(2): C235–C256
Yang Y, Zeng X R, Liu Z F, et al. Characterization of whole-cell IKCa in freshly isolated artery vascular smooth muscle cells from porcine coronary arteries. J Luzhou Med College (in Chinese), 2003, 26(6):473–476
Chen J (translator). Lectures about patch-clamp techniques: Whole-cell recording and perforated patch-clamp. Chin J Neuroanatomy (in Chinese), 1995, 11(1): 87–94
Yang Y Y, Yang Y, Zeng X R, et al. Effects of tetramethylpyrazine on large-conductance Ca2+-activated potassium channels in porcine coronary artery smooth muscle cells. Acta Physiol Sin, 2006, 58(1):83–89
Fill M, Copello J A. Ryanodine receptor calcium release channels. Physiol Rev, 2002, 82: 893–922
Wu J, Kamimura N, Takeo T, et al. 2-Aminoethoxydiphenyl borate modulates kinetics of intracellular Ca2+ signals mediated by inositol 1,4,5-trisphosphate-sensitive Ca2+ stores in single pancreatic acinar cells mouse. Mol Pharmacol, 2000, 58: 1368–1374
Cai F, Li P Y, Yang Y, et al. Characteristic of spontaneous transient outward potassium currents in vascular smooth muscle cells of porcine coronary artery. Acta Physiol Sin, 2007, 59(1): 27–34
Jaggar J H, Stevenson A S, Nelson M T. Voltage dependence of Ca2+ sparks in intact cerebral arteries. Am J Physiol Cell Physiol, 1998, 274: C1755–C1761
Zhuge R, Fogarty K E, Baker S P, et al. Ca2+ spark sites in smooth muscle cells are numerous and differ in number of ryanodine receptors, large-conductance K+ channels, and coupling ratio between them. Am J Physiol Cell Physiol, 2004, 287: C1577–C1588
McCarron J G, Bradley K N, Muir T C. Ca2+ signaling and Ca2+-activated K+ channels in smooth muscle. Novartis Found Symp, 2002, 246: 52–64
Bychkov R, Gollasch M, Ried C, et al. Regulation of spontaneous transient outward potassium currents in human coronary arteries. Circulation, 1997, 95: 503–510
Collier M L, Ji G, Wang Y X, et al. Calcium-induced calcium release in smooth muscle: Loose coupling between the action potential and calcium release. J General Physiol, 2000, 115(5): 653–662
Sharma M R, Jeyakumar L H, Fleischer S, et al. Three-dimensional structure of ryanodine receptor isoform three in two conformational states as visualized by cryo-electron microscopy. J Biol Chem, 2000, 275(13): 9485–9491
Pan B X, Zhao G L, Huang X L, et al. Calcium mobilization is required for peroxynitrite-mediated enhancement of spontaneous transient outward currents in arteriolar smooth muscle cells. Free Radic Biol Med, 2004, 37(6): 823–838
Kong I D, Koh S D, Sanders K M. Purinergic activation of spontaneous transient outward currents in guinea pig taenia colonic myocytes. Am J Physiol Cell Physiol, 2000, 278(2): C352–C362
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Grant No. 30370527)
Rights and permissions
About this article
Cite this article
Li, P., Zeng, X., Yang, Y. et al. Role of calcium mobilization in the regulation of spontaneous transient outward currents in porcine coronary artery myocytes. SCI CHINA SER C 50, 660–668 (2007). https://doi.org/10.1007/s11427-007-0064-7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11427-007-0064-7