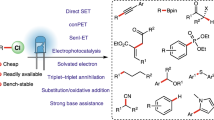
Abstract
Alkyl chlorides are abundant and easily accessible starting materials. However, due to the high reduction potentials associated with unactivated alkyl chlorides, achieving their single electron reduction remains a persistent challenge. This challenge has spurred the exploration of efficient activation methods to overcome this issue. In recent years, photocatalysis has emerged as a mild and potent tool for the single electron reduction of unactivated alkyl chlorides, opening up new possibilities in this field. Considering the rapid advancements in this area, a comprehensive review that provides a conceptual understanding of this emerging field, with a specific focus on reaction design and catalytic mechanisms, would be timely and highly valuable. Hence, we present an overview of various synthetic techniques for photoinduced single electron reduction of unactivated alkyl chlorides. Furthermore, we also discuss the limitations of the present methods and future directions that lie ahead in this field.

Similar content being viewed by others
References
Clayden J, Greeves N, Warren S. Organic Chemistry. Oxford: Oxford University Press, 2012
Gribble GW. Acc Chem Res, 1998, 31: 141–152
Alonso F, Beletskaya IP, Yus M. Chem Rev, 2002, 102: 4009–4092
Vechorkin O, Barmaz D, Proust V, Hu X. J Am Chem Soc, 2009, 131: 12078–12079
Lu Z, Fu G. Angew Chem Int Ed, 2010, 49: 6676–6678
Lu Z, Wilsily A, Fu GC. J Am Chem Soc, 2011, 133: 8154–8157
Sakai HA, Liu W, Le C, MacMillan DWC. J Am Chem Soc, 2020, 142: 11691–11697
Cybularczyk-Cecotka M, Szczepanik J, Giedyk M. Nat Catal, 2020, 3: 872–886
Frisch AC, Beller M. Angew Chem Int Ed, 2005, 44: 674–688
Krische MJ. Radicals in synthesis III. In: Topics in Current Chemistry. Berlin: Springer, 2012. 320
Luo YR. Comprehensive Handbook of Chemical Bond Energies. Boca Raton: CRC Press, 2007
Stephenson CRJ, Yoon TP, MacMillan DWC, Eds. Visible Light Photocatalysis in Organic Chemistry. Weinheim: Wiley-VCH, 2018
Prier CK, Rankic DA, MacMillan DWC. Chem Rev, 2013, 113: 5322–5363
Garbarino S, Ravelli D, Protti S, Basso A. Angew Chem Int Ed, 2016, 55: 15476–15484
Marzo L, Pagire SK, Reiser O, König B. Angew Chem Int Ed, 2018, 57: 10034–10072
Crisenza GEM, Mazzarella D, Melchiorre P. J Am Chem Soc, 2020, 142: 5461–5476
Melchiorre P. Chem Rev, 2022, 122: 1483–1484
Silvi M, Melchiorre P. Nature, 2018, 554: 41–49
Koike T, Akita M. Trends Chem, 2021, 3: 416–427
Liao LL, Song L, Yan SS, Ye JH, Yu DG. Trends Chem, 2022, 4: 512–527
Chen Y, Lu LQ, Yu DG, Zhu CJ, Xiao WJ. Sci China Chem, 2019, 62: 24–57
Wang P, Zhao Q, Xiao W, Chen J. Green Synthesis Catal, 2020, 1: 42–51
Nicewicz DA, MacMillan DWC. Science, 2008, 322: 77–80
Rono LJ, Yayla HG, Wang DY, Armstrong MF, Knowles RR. J Am Chem Soc, 2013, 135: 17735–17738
Murphy JJ, Bastida D, Paria S, Fagnoni M, Melchiorre P. Nature, 2016, 532: 218–222
Proctor RSJ, Davis HJ, Phipps RJ. Science, 2018, 360: 419–422
Jurczyk J, Lux MC, Adpressa D, Kim SF, Lam Y, Yeung CS, Sarpong R. Science, 2021, 373: 1004–1012
Huang M, Zhang L, Pan T, Luo S. Science, 2022, 375: 869–874
Berger M, Ma D, Baumgartner Y, Wong THF, Melchiorre P. Nat Catal, 2023, 6: 332–338
Dai L, Xia Z, Gao Y, Gao Z, Ye S. Angew Chem Int Ed, 2019, 58: 18124–18130
Dai L, Guo J, Huang Q, Lu Y. Sci Adv, 2022, 8: eadd2574
Du J, Skubi KL, Schultz DM, Yoon TP. Science, 2014, 344: 392–396
Tellis JC, Primer DN, Molander GA. Science, 2014, 345: 433–436
Zuo Z, Cong H, Li W, Choi J, Fu GC, MacMillan DWC. J Am Chem Soc, 2016, 138: 1832–1835
Kainz QM, Matier CD, Bartoszewicz A, Zultanski SL, Peters JC, Fu GC. Science, 2016, 351: 681–684
Chen C, Peters JC, Fu GC. Nature, 2021, 596: 250–256
Jiang X, Xiong W, Deng S, Lu FD, Jia Y, Yang Q, Xue LY, Qi X, Tunge JA, Lu LQ, Xiao WJ. Nat Catal, 2022, 5: 788–797
Xiong W, Jiang X, Wang WC, Cheng Y, Lu LQ, Gao K, Xiao WJ. J Am Chem Soc, 2023, 145: 7983–7991
Guo J, Shen ZA, Zhou X, Dai L, Lu Y. Sci China Chem, 2023, 66: 127–132
Chen KQ, Sheng H, Liu Q, Shao PL, Chen XY. Sci China Chem, 2021, 64: 7–16
Crespi S, Fagnoni M. Chem Rev, 2020, 120: 9790–9833
Gál B, Bucher C, Burns NZ. Marine Drugs, 2016, 14: 206
Ertl P, Schuhmann T. J Nat Prod, 2019, 82: 1258–1263
Constantin T, Górski B, Tilby MJ, Chelli S, Juliá F, Llaveria J, Gillen KJ, Zipse H, Lakhdar S, Leonori D. Science, 2022, 377: 1323–1328
Juliá F, Constantin T, Leonori D. Chem Rev, 2022, 122: 2292–2352
Chen JJ, Huang HM. Tetrahedron Lett, 2022, 102: 153945
Matsubara R, Yabuta T, Md Idros U, Hayashi M, Ema F, Kobori Y, Sakata K. J Org Chem, 2018, 83: 9381–9390
Matsubara R, Shimada T, Kobori Y, Yabuta T, Osakai T, Hayashi M. Chem Asian J, 2016, 11: 2006–2010
Griffin GW, Horn KA. J Am Chem Soc, 1987, 109: 4919–4926
Thakar N, Polder N, Djanashvili K, Vanbekkum H, Kapteijn F, Moulijn J. J Catal, 2007, 246: 344–350
Simmons EM, Hartwig JF. Angew Chem Int Ed, 2012, 51: 3066–3072
Konermann L, Pan J, Liu YH. Chem Soc Rev, 2011, 40: 1224–1234
Allen PH, Hickey MJ, Kingston LP, Wilkinson DJ. Labelled Comp Radiopharmac, 2010, 53: 731–738
Atzrodt J, Derdau V, Kerr WJ, Reid M. Angew Chem Int Ed, 2018, 57: 1758–1784
Pirali T, Serafini M, Cargnin S, Genazzani AA. J Med Chem, 2019, 62: 5276–5297
Li Y, Ye Z, Lin YM, Liu Y, Zhang Y, Gong L. Nat Commun, 2021, 12: 2894
Wei D, Li X, Shen L, Ding Y, Liang K, Xia C. Org Chem Front, 2021, 8: 6364–6370
Liang K, Liu Q, Shen L, Li X, Wei D, Zheng L, Xia C. Chem Sci, 2020, 11: 6996–7002
Wu S, Schiel F, Melchiorre P. Angew Chem Int Ed, 2023, 62: e202306364
Ghosh I, Ghosh T, Bardagi JI, König B. Science, 2014, 346: 725–728
Giedyk M, Narobe R, Weiß S, Touraud D, Kunz W, König B. Nat Catal, 2020, 3: 40–47
Kerzig C, Guo X, Wenger OS. J Am Chem Soc, 2019, 141: 2122–2127
Creutz SE, Lotito KJ, Fu GC, Peters JC. Science, 2012, 338: 647–651
Ratani TS, Bachman S, Fu GC, Peters JC. J Am Chem Soc, 2015, 137: 13902–13907
Parasram M, Chuentragool P, Sarkar D, Gevorgyan V. J Am Chem Soc, 2016, 138: 6340–6343
Chuentragool P, Kurandina D, Gevorgyan V. Angew Chem Int Ed, 2019, 58: 11586–11598
Lee GS, Kim D, Hong SH. Nat Commun, 2021, 12: 991
Muralirajan K, Kancherla R, Gimnkhan A, Rueping M. Org Lett, 2021, 23: 6905–6910
Zuo Z, Ahneman DT, Chu L, Terrett JA, Doyle AG, MacMillan DWC. Science, 2014, 345: 437–440
Chan AY, Perry IB, Bissonnette NB, Buksh BF, Edwards GA, Frye LI, Garry OL, Lavagnino MN, Li BX, Liang Y, Mao E, Millet A, Oakley JV, Reed NL, Sakai HA, Seath CP, MacMillan DWC. Chem Rev, 2022, 122: 1485–1542
Claros M, Ungeheuer F, Franco F, Martin-Diaconescu V, Casitas A, Lloret-Fillol J. Angew Chem Int Ed, 2019, 58: 4869–4874
Claros M, Casitas A, Lloret-Fillol J. Synlett, 2019, 30: 1496–1507
Aragón J, Sun S, Pascual D, Jaworski S, Lloret-Fillol J. Angew Chem Int Ed, 2022, 61: e202114365
Chen L, Kametani Y, Imamura K, Abe T, Shiota Y, Yoshizawa K, Hisaeda Y, Shimakoshi H. Chem Commun, 2019, 55: 13070–13073
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22001248), the Fundamental Research Funds for the Central Universities, and the University of the Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Dai, L., Zhang, ZF. & Chen, XY. Reduction of unactivated alkyl chlorides enabled by light-induced single electron transfer. Sci. China Chem. 67, 471–481 (2024). https://doi.org/10.1007/s11426-023-1787-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1787-3