Abstract
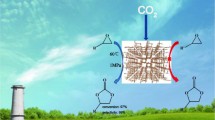
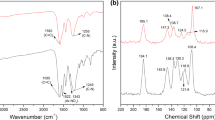
Covalent organic frameworks (COFs) are a potential platform for carbon dioxide (CO2) conversion owing to their periodic permanent porosity, adjustable structure, and chemical stability. For good catalytic performance in CO2 conversion, collaborative multifunctions should be strategically integrated into the catalytic system design and construction. In this study, a four-in-one high-efficiency catalyst was synthesized and tested for CO2 cycloaddition to form cyclic carbonate. The obtained Tp-MPB-Br-COF had a high nitrogen content, which enhanced its CO2 affinity through substantial Lewis acid-base or dipole-quadrupole interactions; moreover, the acid (protons transferring from oxygen (–OH) to nitrogen (–NH)), hydrogen bond donor (hydroxyl group), and Br− (nucleophile group) served as three active sites, further improving the catalyst activity. These results provide a basis for designing efficient and stable CO2-conversion catalysts.

Similar content being viewed by others
References
Luderer G, Vrontisi Z, Bertram C, Edelenbosch OY, Pietzcker RC, Rogelj J, De Boer HS, Drouet L, Emmerling J, Fricko O, Fujimori S, Havlík P, Iyer G, Keramidas K, Kitous A, Pehl M, Krey V, Riahi K, Saveyn B, Tavoni M, Van Vuuren DP, Kriegler E. Nat Clim Change, 2018, 8: 626–633
Al-Mamoori A, Krishnamurthy A, Rownaghi AA, Rezaei F. Energy Technol, 2017, 5: 834–849
Matthews HD, Gillett NP, Stott PA, Zickfeld K. Nature, 2009, 459: 829–832
Zhang Q, Yang C, Guan A, Kan M, Zheng G. Nanoscale, 2022, 14: 10268–10285
Hui W, Xu XY, Mao FF, Shi L, Wang HJ. Sustain Energy Fuels, 2022, 6: 3208–3219
Yao Q, Shi Y, Wang Y, Zhu X, Yuan D, Yao Y. Asian J Org Chem, 2022, 11: e202200106
Saptal VB, Bhanage BM. Curr Opin Green Sustain Chem, 2017, 3: 1–10
Zhao Y, Huang H, Zhu H, Zhong C. Microporous Mesoporous Mater, 2022, 329: 111526
Zhang Y, Lan X, Yan F, He X, Wang J, Ricardez-Sandoval L, Chen L, Bai G. Green Chem, 2022, 24: 930–940
Wu J, Ma S, Cui J, Yang Z, Zhang J. Nanomaterials, 2022, 12: 3088
Hao Y, Yan X, Chang T, Liu X, Kang L, Zhu Z, Panchal B, Qin S. Sustain Energy Fuels, 2022, 6: 121–127
Du YR, Yang X, Wang YF, Guan PX, Wang R, Xu BH. Mol Catal, 2022, 520: 112164
Liang S, Liu H, Jiang T, Song J, Yang G, Han B. Chem Commun, 2011, 47: 2131–2133
Ema T, Miyazaki Y, Shimonishi J, Maeda C, Hasegawa J. J Am Chem Soc, 2014, 136: 15270–15279
North M, Pasquale R. Angew Chem Int Ed, 2008, 48: 2946–2948
Dai WL, Yin SF, Guo R, Luo SL, Du X, Au CT. Catal Lett, 2010, 136: 35–44
Cao JP, Xue YS, Li NF, Gong JJ, Kang RK, Xu Y. J Am Chem Soc, 2019, 141: 19487–19497
Caló V, Nacci A, Monopoli A, Fanizzi A. Org Lett, 2002, 4: 2561–2563
Chen Y, Mu T. Green Chem, 2019, 21: 2544–2574
Roshan KR, Palissery RA, Kathalikkattil AC, Babu R, Mathai G, Lee HS, Park DW. Catal Sci Technol, 2016, 6: 3997–4004
Sun J, Cheng W, Yang Z, Wang J, Xu T, Xin J, Zhang S. Green Chem, 2014, 16: 3071–3078
Du Y, Yang H, Whiteley JM, Wan S, Jin Y, Lee S-, Zhang W. Angew Chem Int Ed, 2016, 55: 1737–1741
Baldwin LA, Crowe JW, Pyles DA, McGrier PL. J Am Chem Soc, 2016, 138: 15134–15137
Pramudya Y, Mendoza-Cortes JL. J Am Chem Soc, 2016, 138: 15204–15213
Huang N, Krishna R, Jiang D. J Am Chem Soc, 2015, 137: 7079–7082
Lin S, Diercks CS, Zhang YB, Kornienko N, Nichols EM, Zhao Y, Paris AR, Kim D, Yang P, Yaghi OM, Chang CJ. Science, 2015, 349: 1208–1213
Ding SY, Gao J, Wang Q, Zhang Y, Song WG, Su CY, Wang W. J Am Chem Soc, 2011, 133: 19816–19822
Vyas VS, Haase F, Stegbauer L, Savasci G, Podjaski F, Ochsenfeld C, Lotsch BV. Nat Commun, 2015, 6: 8508
Sun Q, Aguila B, Perman J, Nguyen N, Ma S. J Am Chem Soc, 2016, 138: 15790–15796
Wang X, Han X, Zhang J, Wu X, Liu Y, Cui Y. J Am Chem Soc, 2016, 138: 12332–12335
Sun Q, Tang Y, Aguila B, Wang S, Xiao F-, Thallapally PK, Al-Enizi AM, Nafady A, Ma S. Angew Chem Int Ed, 2019, 58: 8670–8675
Bhadra M, Kandambeth S, Sahoo MK, Addicoat M, Balaraman E, Banerjee R. J Am Chem Soc, 2019, 141: 6152–6156
Geng K, He T, Liu R, Dalapati S, Tan KT, Li Z, Tao S, Gong Y, Jiang Q, Jiang D. Chem Rev, 2020, 120: 8814–8933
Diercks C, Kalmutzki M, Yaghi O. Molecules, 2017, 22: 1575
Liu R, Tan KT, Gong Y, Chen Y, Li Z, Xie S, He T, Lu Z, Yang H, Jiang D. Chem Soc Rev, 2021, 50: 120–242
Lohse MS, Bein T. Adv Funct Mater, 2018, 28: 1705553
Liang RR, Jiang SY, A RH, Zhao X. Chem Soc Rev, 2020, 49: 3920–3951
Li J, Wang J, Wu Z, Tao S, Jiang D. Angew Chem Int Ed, 2021, 60: 12918–12923
Wu Z, He Y, Liu L, Wang J, Xu Q, Zhang XM, Li J. ACS Appl Mater Interfaces, 2022, 14: 43861–43867
Yang X, Jin Y, Yu B, Gong L, Liu W, Liu X, Chen X, Wang K, Jiang J. Sci China Chem, 2022, 65: 1291–1298
Liu H, Yan X, Chen W, Xie Z, Li S, Chen W, Zhang T, Xing G, Chen L. Sci China Chem, 2021, 64: 827–833
Wu Z, Huang X, Li X, Hai G, Li B, Wang G. Sci China Chem, 2021, 64: 1964–1969
Tong Y, Cheng R, Dong H, Liu B. J Porous Mater, 2022, 29: 1253–1263
Zhang Y, Yang DH, Qiao S, Han BH. Langmuir, 2021, 37: 10330–10339
Li Y, Zhang J, Zuo K, Li Z, Wang Y, Hu H, Zeng C, Xu H, Wang B, Gao Y. Catalysts, 2021, 11: 1133
Li Y, Song X, Zhang G, Chen W, Wang L, Liu Y, Chen L. Sci China Mater, 2021, 65: 1377–1382
Peng J, Deng Y. New J Chem, 2001, 25: 639–641
Su Q, Qi Y, Yao X, Cheng W, Dong L, Chen S, Zhang S. Green Chem, 2018, 20: 3232–3241
Cui C, Sa R, Hong Z, Zhong H, Wang R. ChemSusChem, 2020, 13: 180–187
Ding LG, Yao BJ, Jiang WL, Li JT, Fu QJ, Li YA, Liu ZH, Ma JP, Dong YB. Inorg Chem, 2017, 56: 2337–2344
Yan Q, Liang H, Wang S, Hu H, Su X, Xiao S, Xu H, Jing X, Lu F, Gao Y. Molecules, 2022, 27: 6204
Du YR, Ding GR, Wang YF, Xu BH, Zhang SJ. Green Chem, 2021, 23: 2411–2419
Luo R, Liu X, Chen M, Liu B, Fang Y. ChemSusChem, 2022, 13: 3945–3966
Yang Y, Chen K, Liu X, Chen M, Xu W, Liu B, Ji H, Fang Y. J Mater Chem A, 2021, 9: 20941–20956
Wang KY, Yang Q, Chung TS, Rajagopalan R. Chem Eng Sci, 2009, 64: 1577–1584
Thomas OD, Soo KJWY, Peckham TJ, Kulkarni MP, Holdcroft S. J Am Chem Soc, 2012, 134: 10753–10756
He C, Si D, Huang Y, Cao R. Angew Chem Int Ed, 2022, 61: e202207478
Kang DW, Kang M, Yun H, Park H, Hong CS. Adv Funct Mater, 2021, 31: 2100083
Lin X, Varcoe JR, Poynton SD, Liang X, Ong AL, Ran J, Li Y, Xu T. J Mater Chem A, 2013, 1: 7262–7269
Ito E, Oji H, Araki T, Oichi K, Ishii H, Ouchi Y, Ohta T, Kosugi N, Maruyama Y, Naito T, Inabe T, Seki K. J Am Chem Soc, 1997, 119: 6336–6344
Liu TT, Xu R, Yi JD, Liang J, Wang XS, Shi PC, Huang YB, Cao R. ChemCatChem, 2018, 10: 2036–2040
Huang K, Zhang JY, Liu F, Dai S. ACS Catal, 2018, 8: 9079–9102
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21805173, 52273208), Shanxi Agricultural University (SXBYKY2022078, 2021BQ120), Shanxi Scholarship Council of China (2022-004), and the Natural Science Foundation of Shanxi Province (202203021211289).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at chem.scichina.com and link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic Supplementary Information (ESI)
11426_2023_1754_MOESM1_ESM.pdf
C2-Phenyl-Substituted-Benzimidazolium-Based Covalent Organic Framework as Efficient Catalyst for CO2 Conversion without Solvents, Metals, and Cocatalysts
Rights and permissions
About this article
Cite this article
Wu, Z., Wang, J., Liu, L. et al. C2-phenyl-substituted benzimidazolium-based covalent organic framework as efficient catalyst for CO2 conversion without solvents, metals, and cocatalysts. Sci. China Chem. 67, 551–557 (2024). https://doi.org/10.1007/s11426-023-1754-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1754-5