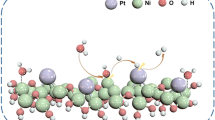
Abstract
The kinetics of hydrogen oxidation reaction (HOR) declines with orders of magnitude when the electrolyte varies from acid to base. Therefore, unveiling the mechanism of pH-dependent HOR and narrowing the acid-base kinetic gap are indispensable and challenging. Here, the HOR behaviors of palladium phosphides and their counterpart (PdP2/C, Pd5P2/C, Pd3P/C, and Pd/C) in the whole pH region (from pH 1 to 13) are explored. Unexpectedly, there are non-monotonous relationships between their HOR kinetics and varied pHs, showing distinct inflection-point behaviors (inflection points and acid-base kinetic gaps). We find the inflection-point behaviors can be explained by the discrepant role of pH-dependent hydroxyl binding energy (OHBE) and hydrogen binding energy (HBE) induced HOR kinetics under the entire pH range. We further reveal that the strengthened OHBE is responsible for the earlier appearance of the inflection point and much narrower acid-base kinetic gap. These findings are conducive to understanding the mechanism of the pH-targeted HOR process, and provide a new strategy for rational designing advanced HOR electrocatalysts under alkaline electrolyte.

Similar content being viewed by others
References
Yang Y, Li P, Zheng X, Sun W, Dou SX, Ma T, Pan H. Chem Soc Rev, 2022, 51: 9620–9693
Yan QQ, Yin P, Liang HW. ACS Mater Lett, 2021, 3: 1197–1212
Zhang Y, Li G, Zhao Z, Han L, Feng Y, Liu S, Xu B, Liao H, Lu G, Xin HL, Huang X. Adv Mater, 2021, 33: 2105049
Sheng W, Gasteiger HA, Shao-Horn Y. J Electrochem Soc, 2010, 157: B1529
Durst J, Siebel A, Simon C, Hasché F, Herranz J, Gasteiger HA. Energy Environ Sci, 2014, 7: 2255–2260
Strmcnik D, Uchimura M, Wang C, Subbaraman R, Danilovic N, van der Vliet D, Paulikas AP, Stamenkovic VR, Markovic NM. Nat Chem, 2013, 5: 300–306
Men Y, Wu D, Hu Y, Li L, Li P, Jia S, Wang J, Cheng G, Chen S, Luo W. Angew Chem Int Ed, 2023, 62: e202217976
Yao ZC, Tang T, Jiang Z, Wang L, Hu JS, Wan LJ. ACS Nano, 2022, 16: 5153–5183
Luo H, Wang K, Lin F, Lv F, Zhou J, Zhang W, Wang D, Zhang W, Zhang Q, Gu L, Luo M, Guo S. Adv Mater, 2023, 35: 2211854
Huang G, Li Y, Tao L, Huang Z, Kong Z, Xie C, Du S, Wang T, Wu Y, Liu Q, Zhang D, Lin J, Li M, Wang J, Zhang J, Lu S, Cheng Y, Wang S. Angew Chem Int Ed, 2023, 62: e202215177
Yang X, Wang Y, Wang X, Mei B, Luo E, Li Y, Meng Q, Jin Z, Jiang Z, Liu C, Ge J, Xing W. Angew Chem Int Ed, 2021, 60: 26177–26183
Zheng J, Sheng W, Zhuang Z, Xu B, Yan Y. Sci Adv, 2016, 2: e1501602
Wang B, Huang H, Huang M, Yan P, Isimjan TT, Yang X. Sci China Chem, 2020, 63: 841–849
Sheng W, Myint MNZ, Chen JG, Yan Y. Energy Environ Sci, 2013, 6: 1509–1512
Ledezma-Yanez I, Wallace WDZ, Sebastián-Pascual P, Climent V, Feliu JM, Koper MTM. Nat Energy, 2017, 2: 17031
Dekel DR. Curr Opin Electrochem, 2018, 12: 182–188
Subbaraman R, Tripkovic D, Strmcnik D, Chang KC, Uchimura M, Paulikas AP, Stamenkovic V, Markovic NM. Science, 2011, 334: 1256–1260
Strmcnik D, Lopes PP, Genorio B, Stamenkovic VR, Markovic NM. Nano Energy, 2016, 29: 29–36
Zhang X, Li Z, Sun X, Wei L, Niu H, Chen S, Chen Q, Wang C, Zheng F. ACS Mater Lett, 2022, 4: 2097–2105
Li J, Ghoshal S, Bates MK, Miller TE, Davies V, Stavitski E, Attenkofer K, Mukerjee S, Ma ZF, Jia Q. Angew Chem Int Ed, 2017, 56: 15594–15598
Wang Y, Wang X, Ze H, Zhang X, Radjenovic PM, Zhang Y, Dong J, Tian Z, Li J. Angew Chem Int Ed, 2021, 60: 5708–5711
McCrum IT, Koper MTM. Nat Energy, 2020, 5: 891–899
Men Y, Su X, Li P, Tan Y, Ge C, Jia S, Li L, Wang J, Cheng G, Zhuang L, Chen S, Luo W. J Am Chem Soc, 2022, 144: 12661–12672
Kim J, Jung H, Jung SM, Hwang J, Kim DY, Lee N, Kim KS, Kwon H, Kim YT, Han JW, Kim JK. J Am Chem Soc, 2021, 143: 1399–1408
Su L, Jin Y, Gong D, Ge X, Zhang W, Fan X, Luo W. Angew Chem Int Ed, 2023, 62: e202215585
Zhou P, Li N, Chao Y, Zhang W, Lv F, Wang K, Yang W, Gao P, Guo S. Angew Chem Int Ed, 2019, 58: 14184–14188
Henkes AE, Vasquez Y, Schaak RE. J Am Chem Soc, 2007, 129: 1896–1897
Guo Z, Wang R, Guo Y, Jiang J, Wang Z, Li W, Zhang M. ACS Catal, 2022, 12: 15193–15206
Jin C, Li J, Zhang K, Habibullah K, Xia G, Wu C, Wang Y, Cen W, Chen Y, Yan Y, Chen Y. Nano Energy, 2022, 99: 107360
Luo F, Zhang Q, Yu X, Xiao S, Ling Y, Hu H, Guo L, Yang Z, Huang L, Cai W, Cheng H. Angew Chem Int Ed, 2018, 57: 14862–14867
Zhang G, Wang A, Niu L, Gao W, Hu W, Liu Z, Wang R, Chen J. Adv Energy Mater, 2022, 12: 2103511
Liu Y, Ding J, Li F, Su X, Zhang Q, Guan G, Hu F, Zhang J, Wang Q, Jiang Y, Liu B, Yang HB. Adv Mater, 2023, 35: 2207114
Zhou S, Jang H, Qin Q, Hou L, Kim MG, Liu S, Liu X, Cho J. Angew Chem Int Ed, 2022, 61: e202212196
Ohyama J, Sato T, Yamamoto Y, Arai S, Satsuma A. J Am Chem Soc, 2013, 135: 8016–8021
Janssen A, Nguyen QN, Xia Y. Angew Chem Int Ed, 2021, 60: 12192–12203
Shen T, Chen S, Zhang C, Hu Y, Ma E, Yang Y, Hu J, Wang D. Adv Funct Mater, 2021, 32: 2107672
Zhou Y, Xie Z, Jiang J, Wang J, Song X, He Q, Ding W, Wei Z. Nat Catal, 2020, 3: 454–462
Rebollar L, Intikhab S, Oliveira NJ, Yan Y, Xu B, McCrum IT, Snyder JD, Tang MH. ACS Catal, 2020, 10: 14747–14762
Zhu S, Qin X, Xiao F, Yang S, Xu Y, Tan Z, Li J, Yan J, Chen Q, Chen M, Shao M. Nat Catal, 2021, 4: 711–718
Kim H, Yoo JM, Chung DY, Kim Y, Jung M, Bootharaju MS, Kim J, Koo S, Shin H, Na G, Mun BS, Kwak JH, Sung YE, Hyeon T. ACS Nano, 2022, 16: 16529–16538
Zhang J, Zhang L, Liu J, Zhong C, Tu Y, Li P, Du L, Chen S, Cui Z. Nat Commun, 2022, 13: 5497
Duan Y, Yu ZY, Yang L, Zheng LR, Zhang CT, Yang XT, Gao FY, Zhang XL, Yu X, Liu R, Ding HH, Gu C, Zheng XS, Shi L, Jiang J, Zhu JF, Gao MR, Yu SH. Nat Commun, 2020, 11: 4789
Jiang J, Liao J, Tao S, Najam T, Ding W, Wang H, Wei Z. Electrochim Acta, 2020, 333: 135444
Wang M, Yang H, Shi J, Chen Y, Zhou Y, Wang L, Di S, Zhao X, Zhong J, Cheng T, Zhou W, Li Y. Angew Chem Int Ed, 2021, 60: 5771–5777
Mao J, He CT, Pei J, Liu Y, Li J, Chen W, He D, Wang D, Li Y. Nano Lett, 2020, 20: 3442–3448
Liu H, Fu J, Li H, Sun J, Liu X, Qiu Y, Peng X, Liu Y, Bao H, Zhuo L, Cao R, Zhang S, Luo J. Appl Catal B-Environ, 2022, 306: 121029
Zhang Z, Ni L, Liu H, Zhao ZL, Yuan XZ, Li H. Sci China Chem, 2022, 65: 611–618
Wang Z, Shen K, Chen L, Li Y. Sci China Chem, 2022, 65: 619–629
Hu Q, Gao K, Wang X, Zheng H, Cao J, Mi L, Huo Q, Yang H, Liu J, He C. Nat Commun, 2022, 13: 3958
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2021YFB4001200), the National Natural Science Foundation of China (22272121, 21972107), and the Natural Science Foundation of Hubei Province (2020CFA095). The numerical calculations in this paper have been done on the supercomputing system in the Supercomputing Center of Wuhan University. We thank the Core Facility of Wuhan University for the measurements of ICP-AES and XPS. We also thank the Core Research Facilities of the College of Chemistry and Molecular Sciences of Wuhan University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information
The supporting information is available online at chem.scichina.com and link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supporting Information
Rights and permissions
About this article
Cite this article
Su, L., Jin, Y., Fan, X. et al. pH-Dependent binding energy-induced inflection-point behaviors for pH-universal hydrogen oxidation reaction. Sci. China Chem. 66, 3262–3268 (2023). https://doi.org/10.1007/s11426-023-1709-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1709-0