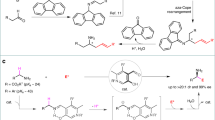
Abstract
This article demonstrates a catalytic method for C(aryl)—C(alkyl) bond functionalization of N-sulfonyl amines. A cobalt(III) catalyst was used to cleave the C—C bond via β-carbon elimination, providing a metallacycle intermediate. Subsequent allylation, amidation, or alkenylation of the intermediate led to divergent conversions in the presence of diverse coupling partners. when the coupling partner was a diene, an insertion-type functionalization was realized with an exclusive 1,3-regioselectivity, in which both of the fragments derived from N-sulfonyl amines were utilized.

Similar content being viewed by others
References
Wang B, Perea MA, Sarpong R. Angew Chem Int Ed, 2020, 59: 18898–18919
Nairoukh Z, Cormier M, Marek I. Nat Rev Chem, 2017, 1: 0035
Murakami M, Ishida N. J Am Chem Soc, 2016, 138: 13759–13769
Lutz MDR, Morandi B. Chem Rev, 2021, 121: 300–326
O’Reilly ME, Dutta S, Veige AS. Chem Rev, 2016, 116: 8105–8145
Satoh T, Miura M. Top Organomet Chem, 2005, 14: 1–20
For selected reviews on C—C bond activation, see: (a)Sokolova OO, Bower JF. Chem Rev, 2021, 121: 80–109
Nogi K, Yorimitsu H. Chem Rev, 2021, 121: 345–364
Lu H, Yu TY, Xu PF, Wei H. Chem Rev, 2021, 121: 365–411
Li JF, Luan YX, Ye M. Sci China Chem, 2021, 64: 1923–1937
Xia Y, Dong G. Nat Rev Chem, 2020, 4: 600–614
Song F, Gou T, Wang BQ, Shi ZJ. Chem Soc Rev, 2018, 47: 7078–7115
Fumagalli G, Stanton S, Bower JF. Chem Rev, 2017, 117: 9404–9432
Chen PH, Billett BA, Tsukamoto T, Dong G. ACS Catal, 2017, 7: 1340–1360
Kim DS, Park WJ, Jun CH. Chem Rev, 2017, 117: 8977–9015
Souillart L, Cramer N. Chem Rev, 2015, 115: 9410–9464
Chen F, Wang T, Jiao N. Chem Rev, 2014, 114: 8613–8661
For recent examples on C—C bond activation, see: (a) Cao J, Wu H, Wang Q, Zhu J. Nat Chem, 2021, 13: 671–676
Zhou X, Xu Y, Dong G. Nat Catal, 2021, 4: 703–710
Guo JH, Liu Y, Lin XC, Tang TM, Wang BQ, Hu P, Zhao KQ, Song F, Shi ZJ. Angew Chem Intl Edit, 2021, 60: 19079–19084
Lu H, Zhao TT, Bai JH, Ye D, Xu PF, Wei H. Angew Chem Int Ed, 2020, 59: 23537–23543
Pannilawithana N, Yi CS. ACS Catal, 2020, 10: 5852–5861
Li H, Ma B, Liu QS, Wang ML, Wang ZY, Xu H, Li LJ, Wang X, Dai HX. Angew Chem Int Ed, 2020, 59: 14388–14393
Morioka T, Nishizawa A, Furukawa T, Tobisu M, Chatani N. J Am Chem Soc, 2017, 139: 1416–1419
Lutz MDR, Gasser VCM, Morandi B. Chem, 2021, 7: 1108–1119
Qiu Y, Scheremetjew A, Ackermann L. J Am Chem Soc, 2019, 141: 2731–2738
Smits G, Audic B, Wodrich MD, Corminboeuf C, Cramer N. Chem Sci, 2017, 8: 7174–7179
Li H, Li Y, Zhang XS, Chen K, Wang X, Shi ZJ. J Am Chem Soc, 2011, 133: 15244–15247
Zhao P, Incarvito CD, Hartwig JF. J Am Chem Soc, 2006, 128: 3124–3125
For selected examples of β-C elimination reactions beyond alcohols, see: Tan G, Das M, Maisuls I, Strassert CA, Glorius F. Angew Chem Int Ed, 2021, 60: 15650–15655
Onodera S, Togashi R, Ishikawa S, Kochi T, Kakiuchi F. J Am Chem Soc, 2020, 142: 7345–7349
Onodera S, Ishikawa S, Kochi T, Kakiuchi F. J Am Chem Soc, 2018, 140: 9788–9792
Deng R, Xi J, Li Q, Gu Z. Chem, 2019, 5: 1834–1846
Bour JR, Green JC, Winton VJ, Johnson JB. J Org Chem, 2013, 78: 1665–1669
Terao Y, Wakui H, Satoh T, Miura M, Nomura M. J Am Chem Soc, 2001, 123: 10407–10408
Huang W, Meng F. Angew Chem Int Ed, 2021, 60: 2694–2698
Ozkal E, Cacherat B, Morandi B. ACS Catal, 2015, 5: 6458–6462
Wang H, Choi I, Rogge T, Kaplaneris N, Ackermann L. Nat Catal, 2018, 1: 993–1001
Wang H, Kaplaneris N, Ackermann L. Cell Rep Phys Sci, 2020, 1: 100178
Kondo T, Kodoi K, Nishinaga E, Okada T, Morisaki Y, Watanabe Y, Mitsudo T. J Am Chem Soc, 1998, 120: 5587–5588
Shao Y, Zhang F, Zhang J, Zhou X. Angew Chem Int Ed, 2016, 55: 11485–11489
Ye J, Limouni A, Zaichuk S, Lautens M. Angew Chem Int Ed, 2015, 54: 3116–3120
Sugiishi T, Kimura A, Nakamura H. J Am Chem Soc, 2010, 132: 5332–5333
Tsuritani T, Yamamoto Y, Kawasaki M, Mase T. Org Lett, 2009, 11: 1043–1045
Fan C, Lv XY, Xiao LJ, Xie JH, Zhou QL. J Am Chem Soc, 2019, 141: 2889–2893
Li L, Liu YC, Shi H. J Am Chem Soc, 2021, 143: 4154–4161
Yan XB, Li L, Wu WQ, Xu L, Li K, Liu YC, Shi H. Nat Commun, 2021, 12: 5881
Marcoux JF, Wagaw S, Buchwald SL. J Org Chem, 1997, 62: 1568–1569
Hummel JR, Boerth JA, Ellman JA. Chem Rev, 2017, 117: 9163–9227
For a review on Co-catalyzed activation of aryl C—O bonds, see: Su B, Cao ZC, Shi ZJ. Acc Chem Res, 2015, 48: 886–896
For selected examples of directed C—C bond activation, see: Zhu J, Zhang R, Dong G. Nat Chem, 2021, 13: 836–842
Yu TY, Xu WH, Lu H, Wei H. Chem Sci, 2020, 11: 12336–12340
Zhu J, Chen PH, Lu G, Liu P, Dong G. J Am Chem Soc, 2019, 141: 18630–18640
Zhao TT, Xu WH, Zheng ZJ, Xu PF, Wei H. J Am Chem Soc, 2018, 140: 586–589
Moselage M, Li J, Kramm F, Ackermann L. Angew Chem Int Ed, 2017, 56: 5341–5344
Yoshino T, Matsunaga S. Adv Synth Catal, 2017, 359: 1245–1262
Moselage M, Li J, Ackermann L. ACS Catal, 2016, 6: 498–525
Zhang SS, Wu JQ, Lao YX, Liu XG, Liu Y, Lv WX, Tan DH, Zeng YF, Wang H. Org Lett, 2014, 16: 6412–6415
Zhang SS, Wu JQ, Liu X, Wang H. ACS Catal, 2015, 5: 210–214
For addition of C-Carbonyl or C-CN bonds to unsaturated bonds, see: (a) Ito Y, Nakatani S, Shiraki R, Kodama T, Tobisu M. J Am Chem Soc, 2022, 144: 662–666
Denton EH, Lee YH, Roediger S, Boehm P, Fellert M, Morandi B. Angew Chem Int Ed, 2021, 60: 23435–23443
Nakao Y. Bull Chem Soc Jpn, 2012, 85: 731–745
Wentzel MT, Reddy VJ, Hyster TK, Douglas CJ. Angew Chem Int Ed, 2009, 48: 6121–6123
For ring expansion of non-strained unsaturated compounds, see: Zhang R, Xia Y, Dong G. Angew Chem Int Ed, 2021, 60: 20476–20482
Xia Y, Ochi S, Dong G. J Am Chem Soc, 2019, 141: 13038–13042
Herraiz AG, Cramer N. ACS Catal, 2021, 11: 11938–11944
Dongbang S, Ellman JA. Angew Chem Int Ed, 2021, 60: 2135–2139
Dongbang S, Shen Z, Ellman JA. Angew Chem Int Ed, 2019, 58: 12590–12594
Yang J, Ji DW, Hu YC, Min XT, Zhou X, Chen QA. Chem Sci, 2019, 10: 9560–9564
Boerth JA, Maity S, Williams SK, Mercado BQ, Ellman JA. Nat Catal, 2018, 1: 673–679
Boerth JA, Ellman JA. Angew Chem Int Ed, 2017, 56: 9976–9980
Boerth JA, Hummel JR, Ellman JA. Angew Chem Int Ed, 2016, 55: 12650–12654
Acknowledgements
This work was supported by the “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2022SDXHDX0006) and the Leading Innovative and Entrepreneur Team Introduction Program of Zhejiang (2020R01004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at http://chem.scichina.com and http://link.springer.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Rights and permissions
About this article
Cite this article
Xu, L., Shi, H. Cobalt-catalyzed divergent functionalization of N-sulfonyl amines via β-carbon elimination. Sci. China Chem. 65, 2214–2218 (2022). https://doi.org/10.1007/s11426-022-1251-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-022-1251-4