Abstract
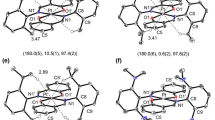
Isolation of E-1,2-bis(4-bromophenyl)-1,2-diphenyl-ethylene from the E/Z isomer mixture obtained by a McMurry coupling reaction and reaction of this isomer with imidazole followed by N-alkylation with nBuBr and anion exchange yielded the bisimidazolium tetraphenylethylene (TPE) derivative H2-E-1(PF6)2. The reaction of H2-E-1(PF6)2 with Ag2O yielded the di-nuclear metallarectangle [Ag2(E-1)2](PF6)2 where the two bis-NHC donors E-1 bridge two silver atoms. Irradiation of [Ag2(E-1)2](PF6)2 leads to E/Z isomerization of the di-NHC ligand and formation of Z-1 in the mononuclear complex [Ag(Z-1)]PF6. Demetallation of the di-NHC ligand with NH4Cl/NH4PF6 yielded bisimidazolium salt H2-Z-1(PF6)2. The unique isomerization of the E-TPE derivative into its Z-isomer via metal complex formation/irradiation/demetallation cannot be achieved by irradiation of the individual imidazolium salt. The emissive properties of the TPE complexes [Ag2(E-1)2](PF6)2 and [Ag(Z-1)]PF6 have been investigated.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
28 November 2021
The article uploaded incorrectly as non open access. This has been change into open access article.
References
Luo J, Xie Z, Lam JWY, Cheng L, Chen H, Qiu C, Kwok HS, Zhan X, Liu Y, Zhu D, Tang BZ. Chem Commun, 2001: 1740–1741
Mei J, Leung NLC, Kwok RTK, Lam JWY, Tang BZ. Chem Rev, 2015, 115: 11718–11940
Ding D, Li K, Liu B, Tang BZ. Acc Chem Res, 2013, 46: 2441–2453
Hong Y, Lam JWY, Tang BZ. Chem Soc Rev, 2011, 40: 5361–5388
Mei J, Hong Y, Lam JWY, Qin A, Tang Y, Tang BZ. Adv Mater, 2014, 26: 5429–5479
Zhao GJ, Han KL, Lei YB, Dou YS. J Chem Phys, 2007, 127: 094307
Xiong JB, Yuan YX, Wang L, Sun JP, Qiao WG, Zhang HC, Duan M, Han H, Zhang S, Zheng YS. Org Lett, 2018, 20: 373–376
Kokado K, Machida T, Iwasa T, Taketsugu T, Sada K. J Phys Chem C, 2018, 122: 245–251
Prlj A, Došlić N, Corminboeuf C. Phys Chem Chem Phys, 2016, 18: 11606–11609
Yang Z, Qin W, Leung NLC, Arseneault M, Lam JWY, Liang G, Sung HHY, Williams ID, Tang BZ. J Mater Chem C, 2016, 4: 99–107
Liu Y, Ye X, Liu G, Lv Y, Zhang X, Chen S, Lam JWY, Kwok HS, Tao X, Tang BZ. J Mater Chem C, 2014, 2: 1004–1009
He L, Liu X, Liang J, Cong Y, Weng Z, Bu W. Chem Commun, 2015, 51: 7148–7151
Garg K, Ganapathi E, Rajakannu P, Ravikanth M. Phys Chem Chem Phys, 2015, 17: 19465–19473
Xie N, Liu Y, Hu R, Leung NLC, Arseneault M, Tang BZ. Isr J Chem, 2014, 54: 958–966
Wang Z, Cheng X, Qin A, Zhang H, Sun JZ, Tang BZ. J Phys Chem B, 2018, 122: 2165–2176
Le Bras L, Adamo C, Perrier A. J Phys Chem C, 2017, 121: 25603–25616
Ghosh KR, Saha SK, Gao JP, Wang ZY. Chem Commun, 2014, 50: 716–718
Hurlock MJ, Kan Y, Lécrivain T, Lapka J, Nash KL, Zhang Q. Cryst Growth Des, 2018, 18: 6197–6203
Ye X, Liu Y, Lv Y, Liu G, Zheng X, Han Q, Jackson KA, Tao X. Angew Chem Int Ed, 2015, 54: 7976–7980
Xu J, Zhang C, Wang X, Jiang J, Wang F. Acta Chim Sin, 2017, 75: 473–478
Zhou SY, Wan HB, Zhou F, Gu PY, Xu QF, Lu JM. Chin J Polym Sci, 2019, 37: 302–326
Xie Y, Li Z. Chem Asian J, 2019, 14: 2524–2541
Hu YB, Lam JWY, Tang BZ. Chin J Polym Sci, 2019, 37: 289–301
Tian W, Lin T, Chen H, Wang W. ACS Appl Mater Interfaces, 2019, 11: 6302–6314
Jia J, Wen J. Tetrahedron Lett, 2020, 61: 151577
Lu Z, Lu S, Cheng Y, Qin Y, Yang S, Liu X, Fan W, Zheng L, Zhang H. J PhotoChem PhotoBiol A-Chem, 2020, 392: 112357
Chen H, Fan Y, Yu X, Semetey V, Trépout S, Li MH. ACS Nano, 2021, 15: 884–893
Rouillon J, Blahut J, Jean M, Albalat M, Vanthuyne N, Lesage A, Ali LMA, Hadj-Kaddour K, Onofre M, Gary-Bobo M, Micouin G, Banyasz A, Le Bahers T, Andraud C, Monnereau C. ACS Appl Mater Interfaces, 2020, 12: 55157–55168
Sun N, Su K, Zhou Z, Wang D, Fery A, Lissel F, Zhao X, Chen C. Macromolecules, 2020, 53: 10117–10127
Wang J, Mei J, Hu R, Sun JZ, Qin A, Tang BZ. J Am Chem Soc, 2012, 134: 9956–9966
Fang X, Zhang YM, Chang K, Liu Z, Su X, Chen H, Zhang SXA, Liu Y, Wu C. Chem Mater, 2016, 28: 6628–6636
Peng HQ, Zheng X, Han T, Kwok RTK, Lam JWY, Huang X, Tang BZ. J Am Chem Soc, 2017, 139: 10150–10156
Zhang CJ, Feng G, Xu S, Zhu Z, Lu X, Wu J, Liu B. Angew Chem Int Ed, 2016, 55: 6192–6196
Hu R, Lam JWY, Liu J, Sung HHY, Williams ID, Yue Z, Wong KS, Yuen MMF, Tang BZ. Polym Chem, 2012, 3: 1481–1489
Yu WH, Chen C, Hu P, Wang BQ, Redshaw C, Zhao KQ. RSC Adv, 2013, 3: 14099–14105
Tseng NW, Liu J, Ng JCY, Lam JWY, Sung HHY, Williams ID, Tang BZ. Chem Sci, 2012, 3: 493–497
Rit A, Pape T, Hahn FE. J Am Chem Soc, 2010, 132: 4572–4573
Han YF, Jin GX, Hahn FE. J Am Chem Soc, 2013, 135: 9263–9266
Segarra C, Guisado-Barrios G, Hahn FE, Peris E. Organometallics, 2014, 33: 5077–5080
Sinha N, Hahn FE. Acc Chem Res, 2017, 50: 2167–2184
Gan MM, Liu JQ, Zhang L, Wang YY, Hahn FE, Han YF. Chem Rev, 2018, 118: 9587–9641
Sun LY, Sinha N, Yan T, Wang YS, Tan TTY, Yu L, Han YF, Hahn FE. Angew Chem Int Ed, 2018, 57: 5161–5165
Martínez-Agramunt V, Eder T, Darmandeh H, Guisado-Barrios G, Peris E. Angew Chem Int Ed, 2019, 58: 5682–5686
Ibáñez S, Peris E. Angew Chem Int Ed, 2019, 58: 6693–6697
Ibáñez S, Poyatos M, Peris E. Acc Chem Res, 2020, 53: 1401–1413
Sinha N, Tan TTY, Peris E, Hahn FE. Angew Chem Int Ed, 2017, 56: 7393–7397
Wang YS, Feng T, Wang YY, Hahn FE, Han YF. Angew Chem Int Ed, 2018, 57: 15767–15771
Li Y, Yang T, Li N, Bai S, Li X, Ma LL, Wang K, Zhang Y, Han YF. CCS Chem, 2021, 3: 957–968
Li Y, Yu JG, Ma LL, Li M, An YY, Han YF. Sci China Chem, 2021, 64: 701–718
Sinha N, Stegemann L, Tan TTY, Doltsinis NL, Strassert CA, Hahn FE. Angew Chem Int Ed, 2017, 56: 2785–2789
Li Y, An YY, Fan JZ, Liu XX, Li X, Hahn FE, Wang YY, Han YF. Angew Chem Int Ed, 2020, 59: 10073–10080
Haldar R, Diring S, Samanta PK, Muth M, Clancy W, Mazel A, Schlabach S, Kirschhöfer F, Brenner-Weiß G, Pati SK, Odobel F, Wöll C. Angew Chem Int Ed, 2018, 57: 13662–13665
Zhang Z, Zhao Z, Wu L, Lu S, Ling S, Li G, Xu L, Ma L, Hou Y, Wang X, Li X, He G, Wang K, Zou B, Zhang M. J Am Chem Soc, 2020, 142: 2592–2600
Yan X, Cook TR, Wang P, Huang F, Stang PJ. Nat Chem, 2015, 7: 342–348
Acknowledgements
This work was supported by the National Natural Science Fund for Distinguished Young Scholars of China (22025107), the National Youth Top-notch Talent Support Program of China, and Key Science and Technology Innovation Team of Shaanxi Province (2019TD-007, 2019JLZ-02). The FM&EM International Joint Laboratory of Northwest University is gratefully acknowledged. FEH gratefully acknowledges financial support from the DFG (SFB 858, IRTG 2027).
Funding
Funding note Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provided a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Liu, XX., Li, Y., Li, X. et al. Photoinduced E to Z isomerization of tetraphenylethylene derivatives within organometallic supramolecular assemblies. Sci. China Chem. 64, 1709–1715 (2021). https://doi.org/10.1007/s11426-021-1041-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-021-1041-8