Abstract
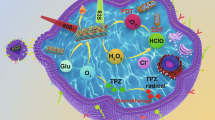
Artificial enzyme cascade systems with confinement effect are highly important in synthetic biology and biomedicine. Herein, a framework nucleic acid-based confined enzyme cascade (FNA-CEC) for synergistic cancer therapy in vivo was developed. The FNA-CEC consisted of glucose oxidase and horseradish peroxidase precisely assembled on an addressable DNA tetrahedron scaffold within few nanometers. Glucose oxidase (GOx) can trigger efficient glucose depletion for tumor starvation therapy, and increase the local concentration of H2O2in situ for enhanced downstream horseradish peroxidase (HRP)-activated prodrug therapy. Due to the spatial-confinement on DNA tetrahedron scaffold, the efficiency of intermediate metabolites transportation between the enzyme cascades was improved. Moreover, FNA-CEC was applied for efficient synergistic cancer therapy in vitro and in vivo. As a simple and efficient approach, the FNA-CEC is expected to expand the toolbox of technologies in synthetic biology and biomedicine.

Similar content being viewed by others
References
Küchler A, Yoshimoto M, Luginbühl S, Mavelli F, Walde P. Nat Nanotechnol, 2016, 11: 409–420
Good MC, Zalatan JG, Lim WA. Science, 2011, 332: 680–686
Fu J, Yang YR, Johnson-Buck A, Liu M, Liu Y, Walter NG, Woodbury NW, Yan H. Nat Nanotech, 2014, 9: 531–536
Yang Y, Zhu W, Cheng L, Cai R, Yi X, He J, Pan X, Yang L, Yang K, Liu Z, Tan W, Chen M. Biomaterials, 2020, 246: 119971
Delebecque CJ, Lindner AB, Silver PA, Aldaye FA. Science, 2011, 333: 470–474
Yang Y, He J, Zhu W, Pan X, Yazd HS, Cui C, Yang L, Li X, Li L, Cheng L, Feng L, Wang R, Liu Z, Chen M, Tan W. Theranostics, 2020, 10: 4030–4041
Wang ZG, Ding B. Acc Chem Res, 2014, 47: 1654–1662
Wen ZB, Peng X, Yang ZZ, Zhuo Y, Chai YQ, Liang WB, Yuan R. Chem Commun, 2019, 55: 13414–13417
Wang H, Li C, Liu X, Zhou X, Wang F. Chem Sci, 2018, 9: 5842–5849
Ge Z, Gu H, Li Q, Fan C. J Am Chem Soc, 2018, 140: 17808–17819
Liu M, Zhang Q, Li Z, Gu J, Brennan JD, Li Y. Nat Commun, 2016, 7: 12074
Dong Y, Dong C, Wan F, Yang J, Zhang C. Sci China Chem, 2015, 58: 1515–1523
Li J, Qiu L, Xie S, Zhang J, Zhang L, Liu H, Li J, Zhang X, Tan W. Sci China Chem, 2018, 61: 497–504
Zhao Y, Zuo X, Li Q, Chen F, Chen Y-R, Deng J, Han D, Hao C, Huang F, Huang Y, Ke G, Kuang H, Li F, Li J, Li M, Li N, Lin Z, Liu D, Liu J, Liu L, Liu X, Lu C, Luo F, Mao X, Sun J, Tang B, Wang F, Wang J, Wang L, Wang S, Wu L, Wu Z-S, Xia F, Xu C, Yang Y, Yuan B-F, Yuan Q, Zhang C, Zhu Z, Yang C, Zhang X-B, Yang H, Tan W, Fan C. Sci China Chem, 2020, doi: https://doi.org/10.1007/s11426-020-9864-7
Chen Y, Ke G, Ma Y, Zhu Z, Liu M, Liu Y, Yan H, Yang CJ. J Am Chem Soc, 2018, 140: 8990–8996
Erkelenz M, Kuo CH, Niemeyer CM. J Am Chem Soc, 2011, 133: 16111–16118
Fu J, Liu M, Liu Y, Woodbury NW, Yan H. J Am Chem Soc, 2012, 134: 5516–5519
Ngo TA, Nakata E, Saimura M, Morii T. J Am Chem Soc, 2016, 138: 3012–3021
Sun L, Gao Y, Xu Y, Chao J, Liu H, Wang L, Li D, Fan C. J Am Chem Soc, 2017, 139: 17525–17532
Fu Y, Zeng D, Chao J, Jin Y, Zhang Z, Liu H, Li D, Ma H, Huang Q, Gothelf KV, Fan C. J Am Chem Soc, 2013, 135: 696–702
Wilner OI, Weizmann Y, Gill R, Lioubashevski O, Freeman R, Willner I. Nat Nanotech, 2009, 4: 249–254
Xin L, Zhou C, Yang Z, Liu D. Small, 2013, 9: 3088–3091
Fejerskov B, Jarlstad Olesen MT, Zelikin AN. Adv Drug Deliver Rev, 2017, 118: 24–34
Greco O, Folkes LK, Wardman P, Tozer GM, Dachs GU. Cancer Gene Ther, 2000, 7: 1414–1420
Fu LH, Qi C, Lin J, Huang P. Chem Soc Rev, 2018, 47: 6454–6472
Vander Heiden MG, Cantley LC, Thompson CB. Science, 2009, 324: 1029–1033
Lin M, Wang J, Zhou G, Wang J, Wu N, Lu J, Gao J, Chen X, Shi J, Zuo X, Fan C. Angew Chem Int Ed, 2015, 54: 2151–2155
Xiang Y, Lu Y. Nat Chem, 2011, 3: 697–703
Liu Q, Ge Z, Mao X, Zhou G, Zuo X, Shen J, Shi J, Li J, Wang L, Chen X, Fan C. Angew Chem Int Ed, 2018, 57: 7131–7135
Liu L, Rong Q, Ke G, Zhang M, Li J, Li Y, Liu Y, Chen M, Zhang XB. Anal Chem, 2019, 91: 3675–3680
Mottley C, Mason RP. J Biol Chem, 1986, 261: 16860–16864
Shen S, Zhu C, Huo D, Yang M, Xue J, Xia Y. Angew Chem Int Ed, 2017, 56: 8801–8804
Li M, Wang C, Di Z, Li H, Zhang J, Xue W, Zhao M, Zhang K, Zhao Y, Li L. Angew Chem Int Ed, 2019, 58: 1350–1354
Knott GJ, Doudna JA. Science, 2018, 361: 866–869
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21705038, 21705043, 21890744, 21705037, 21521063), the National Key R&D Program of China (2019YFA0210100), Natural Science Foundation of Hunan Province (2018JJ3029, 2018JJ3092), China Postdoctoral Science Foundation (2020M672470), and the Open Funding Project of Shandong Key Laboratory of Biochemical Analysis (QUSTHX201808).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kong, G., Zhang, M., Xiong, M. et al. Framework nucleic acid-based confined enzyme cascade for efficient synergistic cancer therapy in vivo. Sci. China Chem. 64, 660–665 (2021). https://doi.org/10.1007/s11426-020-9927-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-020-9927-9