Abstract
Intramolecular N-H…X (X = F, Cl, Br, and I) hydrogen bonding patterns of aromatic amides in the solid state are summarized. It is revealed that the key for the formation of this kind of weak intramolecular hydrogen bonding in X-ray crystal structures is to suppress the competition of strong intermolecular N-H…O-C hydrogen bonding of the amide unit. For amides with identical backbones, the bonding capacity of halogen atoms as hydrogen bonding acceptors is in the order of F>Cl>Br>I, which is in accordance with their electronegativity strength. Generally, the five-membered hydrogen bonding is easier to form than the six-membered one.
Similar content being viewed by others
References
Arunan E, Desiraju GR, Klein RA, Sadlej J, Scheiner S, Alkorta I, Clary DC, Crabtree RH, Dannenberg JJ, Hobza P, Kjaergaard HG, Legon AC, Mennucci B, Nesbitt DJ. Definition of the hydrogen bond (IUPAC recommendations 2011). Pure Appl Chem, 2011, 83: 1637–1641
Jeffrey GA. An Introduction to Hydrogen Bonding. Oxford University Press, Oxford, 1997
Pauling L. The Nature of the Chemical Bond. 3rd ed. Cornell University Press: Ithaca, New York, 1960
Dunitz JD. Organic fluorine: Odd man out. ChemBioChem, 2004, 5: 614–621
Dunitz JD, Taylor R. Organic fluorine hardly ever accepts hydrogen bonds. Chem Eur J, 1997, 3: 89–98
Manjunatha GNR, Vasantha KMV, Row GTN, Suryaprakash N. N-H…F hydrogen bonds in fluorinated benzanilides: NMR and DFT study. Phys Chem Chem Phys, 2010, 12: 13232–13237
Legon AC. Tilden lecture. The properties of hydrogen-bonded dimers from rotational spectroscopy. Chem Soc Rev, 1990, 19: 197–237
Liu Z, Remsing RC, Liu D, Moyna G, Pophristic V. Hydrogen bonding in ortho-substituted arylamides: The influence of protic solvents. J Phys Chem B, 2009, 113: 7041–7044
Frey JA, Leist R, Leutwyler S. Hydrogen bonding of the nucleobase mimic 2-pyridone to fluorobenzenes: An ab initio investigation. J Phys Chem A, 2006, 110: 4188–4195
Koller AN, Bozilovic J, Engels JW, Gohlke H. Aromatic N versus aromatic F: Bioisosterism discovered in RNA base pairing interactions leads to a novel class of universal base analogs. Nucleic Acids Res, 2010, 38: 3133–3146
Toth G, Bowers SG, Truong AP, Probst G. The role and significance of unconventional hydrogen bonds in small molecule recognition by biological receptors of pharmaceutical relevance. Curr Pharm Des, 2007, 13: 3476–3493
Desiraju GR, Steiner T. The weak hydrogen bond. In: Structural Chemistry and Biology. Oxford University Press, New York, 2001
Biradha K. Crystal engineering: From weak hydrogen bonds to co-ordination bonds. CrystEngComm, 2003, 5: 374–384
Desiraju GR. C-H…O and other weak hydrogen bonds. From crystal engineering to virtual screening. Chem Commun, 2005, 2995-3001
Zhu YY, Wang GT, Li ZT. Molecular recognition with linear molecules as receptors. Curr Org Chem, 2011, 15: 1266–1292
Zhao X, Li ZT. Hydrogen bonded aryl amide and hydrazide oligomers: A new generation of preorganized soft frameworks. Chem Commun, 2010, 46: 1601–1616
Saraogi I, Hamilton AD. Recent advances in the development of aryl-based foldamers. Chem Soc Rev, 2009, 38: 1726–1743
Li ZT, Hou JL, Li C. Peptide mimics by linear arylamides: A structural and functional diversity test. Acc Chem Res, 2008, 41: 1343–1353
Li ZT, Hou JL, Li C, Yi HP. Shape-persistent aromatic amide oligomers: New tools for supramolecular chemistry. Chem Asian J, 2006, 1: 766–778
Huc I. Aromatic oligoamide foldamers. Eur J Org Chem, 2004, 17-29
Gan Q, Wang Y, Jiang H. Aromatic oligoamide foldamers: A paradigm for structure-property relationship. Curr Org Chem, 2011, 15: 1293–1301
Tew GN, Scott RW, Klein ML, DeGrado WF. De Novo design of antimicrobial polymers, foldamers, and small molecules: From discovery to practical applications. Acc Chem Res, 2010, 43: 30–39
Li X, Wu YD, Yang D. α-Aminoxy acids: New possibilities from foldamers to anion receptors and channels. Acc Chem Res, 2008, 41: 1428–1438
Bautista AD, Craig CJ, Harker EA, Schepartz A. Sophistication of foldamer form and function in vitro and in vivo. Curr Opin Chem Biol, 2007, 11: 685–692
Howard JAK, Roy VJ, O’Hagan D, Smith GT. How good is fluorine as a hydrogen bond acceptor? Tetrahedron, 1996, 52: 12613–12622
Barbarich TJ, Rithner CD, Miller SM, Anderson OP, Strauss SS. Significant inter- and intramolecular O-H…FC hydrogen bonding. J Am Chem Soc, 1999, 121: 4280–4281
Buyukgungor O, Odabasoglu M. 2-Fluoroanilinium N-(2-fluorophenyl)oxamate. Acta Crystallogr E, 2008, 64: o808–o808
Li C, Ren SF, Hou JL, Yi HP, Zhu SZ, Jiang XK, Li ZT. F…HN hydrogen bonding driven foldamers: Efficient receptors for dialkylammonium ions. Angew Chem Int Ed, 2005, 44: 5725–5729
Chopra D, Row TNG. Dimorphic forms in a non-centrosymmetric environment from a prochiral molecule: Cooperative interplay of strong hydrogen bonds and weak intermolecular interactions. Cryst Growth Des, 2005, 5: 1679–1681
Lewis RJ, Camilleri P, Kirby AJ, Marby CA, Slawin AA, Williams DJ. Structure-activity studies on amides inhibiting photosystem. II. J Chem Soc Perkin Trans 2, 1991, 1625–1630
Kato Y, Sakurai K. The crystal structure of o-fluorobenzamide. Bull Chem Soc Jpn, 1982, 55: 1643–1644
Zhao X, Wang XZ, Jiang XK, Chen YQ, Li ZT, Chen GJ. Hydrazide-based quadruply hydrogen-bonded heterodimers. Structure, assembling selectivity, and supramolecular substitution. J Am Chem Soc, 2003, 125: 15128–15139
Chopra D, Row TNG. Evaluation of the interchangeability of C-H and C-F groups: Insights from crystal packing in a series of isomeric fluorinated benzanilides. CrystEngComm, 2008, 10: 54–67
Kampf M, Richter R, Hennig L, Eidner A, Baldamus J, Kirmse R. Synthese, strukturen, NMR-und EPR-spektroskopische untersuchungen an übergangsmetallkomplexen monofluorsubstituierter acylselenoharnstoffliganden. Z Anorg Allg Chem, 2004, 630: 2677–2686
Chisholm G, Kennedy AR, Beaton L, Brook E. Structural motifs in acetoacetanilides: The effect of a fluorine substituent. Acta Crystallogr C, 2002, 58: o645–o648
Gong B. Crescent oligoamides: From acyclic “macrocycles” to folding nanotubes. Chem Eur J, 2001, 7: 4337–4342
Zhu YY, Wu J, Li C, Zhu J, Hou JL, Li CZ, Jiang XK, Li ZT. F…H-N and MeO…H-N hydrogen-bonding in the solid states of aromatic amides and hydrazides: A comparison study. Cryst Growth Des, 2007, 7: 1490–1496
Zhu S, Nieger M, Daniels J, Felder T, Kossev I, Schmidt T, Sokolowski M, Vögtle F, Schalley CA. Conformational flexibility of tetralactam macrocycles and their intermolecular hydrogen-bonding patterns in the solid state. Chem Eur J, 2009, 15: 5040–5046
Guo D, Nichol GS, Cain JP, Yalkowsky SH. 2-[N-(2,4-Difluorophenyl)carbamoyl]-3,4,5,6-tetrafluorobenzoic acid. Acta Crystallogr E, 2009, 65: o2644
Wan X, Ma Z, Li B, Zhang K, Cao S, Zhang S, Shi Z. Highly selective C-H functionalization/halogenation of acetanilide. J Am Chem Soc, 2006, 128: 7416–7417
Gowda BT, Svoboda I, Fuess H. N-(2,4-Dichlorophenyl)acetamide. Acta Crystallogr E, 2007, 63: o3308
Zhu YY, Yi HP, Li C, Jiang XK, Li ZT. The N-H…X (X = Cl, Br, and I) hydrogen-bonding pattern in aromatic amides: A crystallographic and 1H NMR study. Cryst Growth Des, 2008, 8: 1294–1300
Gowda BT, Foro S, Fuess H. 2-Chloro-N-(2-chlorophenyl)acetamide. Acta Crystallogr E, 2007, 63: o4611
Gowda BT, Paulus H, Fuess H. Structural studies on substituted N-(phenyl)-2,2-dichloroacetamides, 2/4-XC6H4·NHCO·CHCl2 (X = H, Cl or CH3). Z Naturforsch A, 2001, 56: 386–394
Gowda BT, Paulus H, Fuess H. Effect of substituent on crystal structures of N-(substituted-phenyl)-2,2,2-trichloroacetamides, 2/3/4-XC6H4·NHCO·CCl3 (X = Cl, NO2 or CH3). Z Naturforsch A, 2000, 55: 711–720
Gowda BT, Foro S, Suchetan PA, Fuess H, Terao H. 2-Bromo-N-(2-chlorophenyl)acetamide. Acta Crystallogr E, 2009, 65: o1998
Gowda BT, Foro S, Suchetan PA, Fuess H. 2,2,2-Tribromo-N-(2-chlorophenyl)acetamide. Acta Crystallogr E, 2010, 66: o386
Kashino S, Iwamoto T, Yamamoto E, Shiraga T. Crystal structures of acrylamide derivatives: N-t-Butylacrylamide, N-t-butylmethacrylamide, dimorphs of N-phenylmethacrylamide, and N-o-chlorophe-nylmethacrylamide. Bull Chem Soc Jpn, 1994, 67: 1226–1231
Gowda BT, Sowmya BP, Kozisek J, Tokarcik M, Fuess H. N-(2-Chlorophenyl)benzamide. Acta Crystallogr E 2007, 63: o2906
Saeed A, Khera RA, Gotoh K, Ishida H. 4-Chloro-N-(2-chlorophenyl)benzamide. Acta Crystallogr E, 2008, 64: o1934
Gowda BT, Foro S, Sowmya BP, Fuess H. N-(2-Chlorophenyl)-2-methylbenzamide. Acta Crystallogr E, 2008, 64: o1421
Rodrigues VZ, Tokarcik M, Gowda BT, Kozisek J. N-(2-Chlorophenyl)-3-methylbenzamide. Acta Crystallogr E, 2010, 66: o891
Gowda BT, Tokarcik M, Kozisek J, Sowmya BP, Fuess H. N-(2,4-Dichlorophenyl)benzamide. Acta Crystallogr E, 2008, 64: o950
Gowda BT, Foro S, Sowmya BP, Fuess H. 2-Chloro-N-(2-chlorophenyl)benzamide. Acta Crystallogr E, 2007, 63: o3789
Gowda BT, Foro S, Sowmya BP, Fuess H. 2-Chloro-N-(2,3-dichlorophenyl)benzamide. Acta Crystallogr E, 2008, 64, o1342
Sindt AC, Mackay MF. Flukicides. II. Crystal structures of the triclinic and monoclinic polymorphs of 3,3′,5,5′,6-pentachloro-2′-hydroxysalicylanilide. Acta Crystallogr B, 1979, 35: 2103–2108
Khan ZA, Khan KM, Anjum S. 2-(2-Chlorobenzamido)benzoic acid. Acta Crystallogr E 2007, 63: o4103
Kato Y, Takaki Y, Sakurai K. Polymorphism and disordered structures of o-chlorobenzamide. Acta Crystallogr B, 1974, 30: 2683–2687
Etter MC. Encoding and decoding hydrogen-bond patterns of organic compounds. Acc Chem Res, 1990, 23: 120–126
Arslan H, Florke U, Kulcu N, Binzet G. The molecular structure and vibrational spectra of 2-chloro-N-(diethylcarbamothioyl)benzamide by Hartree-Fock and density functional methods Spectrochim Acta, A 2007, 68: 1347–1353
Binzet G, Arslan H, Florke U, Kulcu N, Duran N. Synthesis, Characterization and antimicrobial activities of transition metal complexes of N,N-dialkyl-N-(2-chlorobenzoyl)thiourea derivatives. J Coord Chem, 2006, 59: 1395–1406
Binzet G, Florke U, Kulcu N, Arslan H. Crystal structure of 3-(2-chlorobenzoyl)-1,1-diphenylthiourea, (C6H5)2N(CS)(NH)(CO)-(C6H4Cl). Z Kristallogr-New Cryst Struct, 2004, 219: 395–397
Caleta I, Cincic D, Karminski-Zamola G, Kaitner B. The synthesis and structure of two novel N-(benzothiazol-2-yl)benzamides. J Chem Cryst, 2008, 38: 775–780
Ronaldson V, Storey JMD, Harrison WTA. N-(2-Bromophenyl) acetamide. Acta Crystallogr E, 2005, 61: o3156–o3158
Ronaldson V, Storey JMD, Harrison WTA. N-(2-Bromophenyl) propionamide. Acta Crystallogr E, 2005, 61: o3200–o3202
Huang JY, Xu W. N-(2-Bromophenyl)-2-phenylpropanamide. Acta Crystallogr E 2006, 62: o2651–o2652
Liu DQ, Yan FY. (E)-N-(2-Bromophenyl)-3-(2-chloro-3,3,3-trifluoroprop-1-enyl)-2,2-dimethylcyclopropanecarboxamide. Acta Crystallogr E, 2007, 63: o4202
Venkatachalam TK, Zheng Y, Ghosh S, Uckun FM. Structural influence on the intermolecular/intramolecular hydrogen bonding in solid state of substituted leflunomides: Evidence by X-ray crystal structure. J Mol Struct, 2005, 743: 103–115
Percival D, Storey JMD, Harrison WTA. N-(2-Bromophenyl)pyridine-3-carboxamide. Acta Crystallogr E, 2007, 63: o1851–o1852
Izumi T, Okamoto N. The crystal structure of o-bromobenzamide. Mem Chubu Inst Technol, 1972, 8: 139–142
Zhu YY, Jiang L, Li ZT. Intramolecular six-membered N-H…Br and N-H…I hydrogen bonding in aromatic amides in the absence of competing interactions. CrystEngComm, 2009, 11: 235–238
Narayana B, Sunil K, Yathirajan HS, Sarojini BK, Bolte M. 2-Bromo-N′-[(E)-(4-fluorophenyl)methylene]-5-methoxybenzohydraz ide monohydrate. Acta Crystallogr E, 2007, 63: o2948
Bowie AL Jr, Hughes CC, Trauner D. Concise synthesis of (±)-rhazinilam through direct coupling. Org Lett, 2005, 7: 5207–5209
Cicak H, Dakovic M, Mihalic Z, Pavlovic G, Racane L, Tralic-Kulenovic V. Hydrogen and halogen bonding patterns and π-π aromatic interactions of some 6,7-disubstituted 1,3-benzothiazoles studied by X-ray diffraction and DFT calculations. J Mol Struct, 2010, 975: 115–127
Wardell JL, Skakle JMS, Low JN, Glidewell C. Contrasting three-dimensional framework structures in the isomeric pair 2-iodo-N-(2-nitrophenyl)benzamide and N-(2-iodophenyl)-2-nitrobenzamide. Acta Crystallogr C, 2005, 61: o634–o638
Demartin F, Filippini G, Gavezzotti A, Rizzato S. X-ray diffraction and packing analysis on vintage crystals: Wilhelm Koerner′s nitrobenzene derivatives from the school of agricultural sciences in milano. Acta Crystallogr B, 2004, 60: 609–620
Wardell JL, Low JN, Skakle JMS, Glidewell C. Isomeric N-(iodophenyl)nitrobenzamides form different three-dimensional framework structures. Acta Crystallogr B 2006, 62: 931–943
Glidewell C, Low JN, Skakle JMS, Wardell JL. Bilayers in 4-iodo-N-(2-iodo-4-nitrophenyl)benzamide generated by a combination of an N-H…O=C hydrogen bond and two independent iodo-nitro interactions. Acta Crystallogr C, 2003, 59: o636–o637
Garden SJ, Wardell JL, Low JN, Skakle JMS, Glidewell C. 2-Iodo-4-nitro-N-(trifluoroacetyl)aniline: Sheets built from iodo-nitro and nitro-nitro interactions. Acta Crystallogr E, 2006, 62: o3762–o3764
Nakata K, Tateno T, Sakurai K. The crystal structure of o-iodobenzamide, C7H6NOI. Mem Osaka Kyoiku Univ, 1976, 25: 61–68
Balavoine F, Madec D, Mioskowski C. Highly regioselective palladium-catalyzed condensation of terminal acetylenes with 2,5-diiodobenzoic acid. Tetrahedron Lett, 1999, 40: 8351–8354
Garden SJ, Glidewell C, Low JN, Skakle JMS, Wardell JL. 2-Iodo-N-(4-nitrophenyl)benzamide forms hydrogen-bonded sheets of R4 4(24) rings. Acta Crystallogr C, 2005, 61: o450–o451
Gan Q, Ferrand Y, Bao C, Kauffmann B, Grélard A, Jiang H, Huc I. Helix-rod host-guest complexes with shuttling rates much faster than disassembly. Science, 2011, 331: 1172–1175
Gan Q, Li F, Li G, Kauffmann B, Xiang J, Huc I, Jiang H. Heteromeric double helix formation by cross-hybridization of chloro-and fluoro-substituted quinoline oligoamides. Chem Commun, 2010, 46: 297–299
Gan Q, Bao C, Kauffmann C, Grélard A, Xiang J, Liu S, Huc I, Jiang H, Quadruple and double helices of 8-fluoroquinoline oligoamides. Angew Chem Int Ed, 2008, 47: 1715–1718
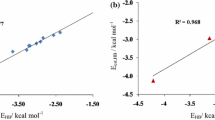
Zhao GJ, Han KL, Hydrogen bonding in the electronic excited state. Acc Chem Res, 2012, 45: 404–413
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, D., Wang, J., Zhang, D. et al. N-H…X (X = F, Cl, Br, and I) hydrogen bonding in aromatic amide derivatives in crystal structures. Sci. China Chem. 55, 2018–2026 (2012). https://doi.org/10.1007/s11426-012-4716-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-012-4716-5