Abstract
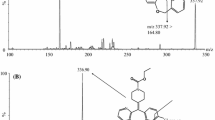
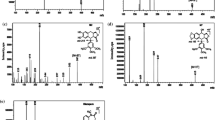
A new liquid chromatography-mass spectrometry method for the determination of carbamazepine, clonazepam, alprazolam, estazolam and phenytoin in human plasma has been developed by using diazepam as an internal standard. Chromatographic separation was performed on a Zorbax SB-C18 column (30 mm × 2.1 mm, 3.5 μm) with a mobile phase consisting of methanol and aqueous 25 mM ammonium acetate using gradient elution. A diethyl ether extraction method was used for the extraction of five anti-epilepsy drugs. The final extract was injected for analysis by LC-MS/MS. The method was validated within the concentration range of 50–5000 ng mL−1 for five anti-epilepsy drugs. The precision of the assay (RSD%) was less than 10% at all concentration levels within the tested range. The method recoveries for all samples were more than 90%. The results indicate that the method is specific, sensitive and accurate, and suitable to study the pharmacokinetics, to adjust the dosage for individual administration, and to monitor the drug-concentration and drug abuse of the five anti-epilepsy drugs.
Similar content being viewed by others
References
Schmidt D. Drug treatment of epilepsy: Options and limitations. Epilepsy Behav, 2009, 15: 56–65
Sánchez MB, Herranz JL, Leno C, Arteaga R, Oterino A, Valdizán EM, Nicolás JM, Adín J, Armijo JA. Genetic factors associated with drug-resistance of epilepsy: Relevance of stratification by patient age and aetiology of epilepsy. Seizure, 2010, 19: 93–101
Jabbaribar F, Mortazavi SA, Jouyban A. Determination of phenytoin and its major metabolite in human serum by MEKC. J Chromatogr Sci, 2009, 47: 877–880
Ali AAP, Kamiar AP. Antinuclear antibodies in children with epilepsy treated by carbamazepine. Epilepsy Res, 2008, 80: 229–230
Badiu CL. Sensitivity of thalamic GABAergic currents to clonazepam does not differ between control and genetic absence epilepsy rats. Brain Res, 2004, 1026: 261–266
Lane SD, Gowin JL. GABAergic modulation of human social interaction in a prisoner’s dilemma model by acute administration of alprazolam. Behav Pharmacol, 2009, 20: 657–661
Miura M, Otani K, Ohkubo T. Identification of human cytochrome P450 enzymes involved in the formation of 4-hydroxyestazolam from estazolam. Xenobiotica, 2005, 35: 455–465
Allqvist A, Wennerholm A, Svensson JO, Rajaa AM. Simultaneous quantification of alprazolam, 4- and α-hydroxyalprazolam in plasma samples using liquid chromatography mass spectrometry. J Chromatogr B, 2005, 814: 127–131
van Rooyen GF, Badenhorst D, Swart KJ, Hundt HKL, Scanes T, Hundt AF. Determination of carbamazepine and carbamazepine 10,11-epoxide in human plasma by tandem liquid chromatographymass spectrometry with electro spray ionization. J Chromatogr B, 2002, 769: 1–7
Quintela O, Lopez P, Bermejo AM, Lopez-Rivadulla M, Determination of methadone, 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine and alprazolam in human plasma by liquid chromatography-electro spray ionization mass spectrometry. J Chromatogr B, 2006, 834: 188–194
Zhu Y, Chiang H, Wulster-Radcliffe H, Hilt R, Wong P, Kissinger CB, Kissinger PT. Liquid chromatography/tandem mass spectrometry for the determination of carbamazepine and its main metabolite in rat plasma utilizing an automated blood sampling system. J Pharm Biomed Anal, 2005, 38: 119–125
Bares IF, Pehourcq F, Jarry C. Development of a rapid RP-HPLC method for the determination of clonazepam in human plasma. J Pharmaceut Biomed Anal, 2004, 36: 865–869
Kelly JF, Greenblatt DJ. Rapid and sensitive gas chromatographic determination of estazolam. J Chromatogr B, 1993, 621: 102–104
Liu YZ, Zhou SX, Zeng L. Simultaneous determination of concentration of 5 anti-epilepsy drugs in plasma by solid phase extraction RP-HPLC method. Central South Pharmacy, 2008, 6: 686–688
Xu RN, Fan L, Rieser MJ. Recent advances in high throughput quantitative bioanalysis by LC-MS/MS. J Pharm Biom Anal, 2007, 44: 342
Ma SG, Zhu MS. Recent advances in applications of liquid chromatography-tandem mass spectrometry to the analysis of reactive drug metabolites. Chem-Biol Inter, 2009, 179: 25–37
Vanhoutte K, Dongen WV, Esmans EL. On-line nanoscale liquid chromatography nano-electrospray mass spectrometry: Effect of the mobile phase composition and the electrospray tip design on the performance of a nanoflow TM electrospray probe. Rapid Commun Mass Spect, 1998, 12: 15–24
Garcia MC. The effect of the mobile phase additives on sensitivity in the analysis of peptides and proteins by high-performance liquid chromatography-electrospray mass spectrometry. J Chromatogr B, 2005, 825: 111–123
Rochfort SJ, Trenerry CV, Imsic M, Panozzo J, Jones R Class targeted metabolomics: ESI ion trap screening methods for glucosinolates based on MSn fragmentation. Phytochemistry, 2008, 69: 1671–1679
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Q., Stefanie, W., Luo, S. et al. Simultaneous determination of five anti-epilepsy drugs in human plasma using liquid chromatography-mass spectrometry. Sci. China Chem. 53, 2373–2378 (2010). https://doi.org/10.1007/s11426-010-4009-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-010-4009-9