Abstract
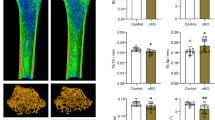
(–)-epigallocatechin-3-O-gallate (EGCG) is a bioactive polyphenol in green tea. Previous studies have demonstrated the beneficial effects of EGCG on muscle mass and muscle atrophy. In the current study, we investigated the mechanisms underlying effect of EGCG on muscle atrophy. It was demonstrated that EGCG suppressed muscle-specific ubiquitin ligase, muscle RING Finger 1 (MuRF1) expression through 67-kDa laminin receptor (67LR). Previous studies have shown that eriodictyol potentiates the anti-tumor activities of EGCG by amplifying 67LR signaling. Therefore, we investigated the effects of EGCG and eriodictyol on the MuRF1 expression in C2C12 myotubes. The combined treatment of EGCG and eriodictyol significantly suppressed MuRF1 expression in dexamethasone-treated C2C12 myotubes. Tail suspension was maintained for 10 consecutive days using C57BL6/J mice, and during this time EGCG and eriodictyol were orally administered. In the gastrocnemius muscle, the muscle mass loss was inhibited by the combination of EGCG and eriodictyol. Therefore, EGCG may prevent muscle atrophy by inducing 67LR signaling and eriodictyol amplifies this pathway.




Similar content being viewed by others
References
Phillips SM, Glover EI, Rennie MJ (2009) Alterations of protein turnover underlying disuse atrophy in human skeletal muscle. J Appl Physiol 107(3):645–654
Evans WJ (1127S) Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr 91(4):1123S–1127S
Goodpaster BH, Park SW, Harris TB, Kritchevsky SB, Nevitt M, Schwartz AV, Simonsick EM, Tylavsky FA, Visser M, Newman AB (2006) The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci 61(10):1059–1064
Attaix D, Ventadour S, Codran A, Béchet D, Taillandier D, Combaret L (2005) The ubiquitin–proteasome system and skeletal muscle wasting. Essays Biochem 41:173–186
Lecker SH, Goldberg AL, Mitch WE (2006) Protein degradation by the ubiquitin–proteasome pathway in normal and disease states. J Am Soc Nephrol 17(7):1807–1819
Bodine SC, Latres E, Baumhueter S, Lai VKM, Nunez L, Clarke BA, Poueymirou WT, Panaro FJ, Na E, Dharmarajan K, Pan ZQ, Valenzuela DM, DeChiara TM, Stitt TN, Yancopoulos GD, Glass DJ (2001) Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294(5547):1704–1708
Lambert JD, Elias RJ (2010) The antioxidant and pro-oxidant activities of green tea polyphenols: a role in cancer prevention. Arch Biochem Biophys 501(1):65–72
Luo KW, Lung WY, Chun-Xie XLL, Huang WR (2018) EGCG inhibited bladder cancer T24 and 5637 cell proliferation and migration via PI3K/AKT pathway. Oncotarget 9(15):12261
Oliviero F, Sfriso P, Scanu A, Fiocco U, Spinella P, Punzi L (2013) Epigallocatechin-3-gallate reduces inflammation induced by calcium pyrophosphate crystals in vitro. Front Pharmacol 4:51
Wang H, Lai YJ, Chan YL, Li TL, Wu CJ (2011) Epigallocatechin-3-gallate effectively attenuates skeletal muscle atrophy caused by cancer cachexia. Cancer Lett 305(1):40–49
Dorchies OM, Wagner S, Vuadens O, Waldhauser K, Buetler TM, Kucera P, Ruegg UT (2006) Green tea extract and its major polyphenol (−)-epigallocatechin gallate improve muscle function in a mouse model for Duchenne muscular dystrophy. Am J Physiol Cell Physiol 290(2):C616–C625
Ota N, Soga S, Haramizu S, Yokoi Y, Hase T, Murase T (2011) Tea catechins prevent contractile dysfunction in unloaded murine soleus muscle: a pilot study. Nutrition 27(9):955–959
Tachibana H, Koga K, Fujimura Y, Yamada K (2004) A receptor for green tea polyphenol EGCG. Nat Struct Mol Biol 11(4):380–381
Tachibana H (2011) Green tea polyphenol sensing. Proc Japan Acad Ser B 87(3):66–80
Umeda D, Yano S, Yamada K, Tachibana H (2008) Green tea polyphenol epigallocatechin-3-gallate signaling pathway through 67-kDa laminin receptor. J Biol Chem 283(6):3050–3058
Tsukamoto S, Hirotsu K, Kumazoe M, Goto Y, Sugihara K, Suda T, Tsurudome Y, Suzuki T, Yamashita S, Kim Y, Huang Y, Yamada K, Tachibana H (2012) Green tea polyphenol EGCG induces lipid-raft clustering and apoptotic cell death by activating protein kinase Cδ and acid sphingomyelinase through a 67 kDa laminin receptor in multiple myeloma cells. Biochem J 443(2):525–534
Byun EH, Fujimura Y, Yamada K, Tachibana H (2010) TLR4 signaling inhibitory pathway induced by green tea polyphenol epigallocatechin-3-gallate through 67-kDa laminin receptor. J Immunol 185(1):33–45
Kumazoe M, Yamashita M, Nakamura Y, Takamatsu K, Bae J, Yamashita S, Yamada S, Onda H, Nojiri T, Kanagawa K, Tachibana H (2017) Green tea polyphenol EGCG upregulates Tollip expression by suppressing Elf-1 expression. J Immunol 199(9):3261–3269
Minato KI, Miyake Y, Fukumoto S, Yamamoto K, Kato Y, Shimomura Y, Osawa T (2003) Lemon flavonoid, eriocitrin, suppresses exercise-induced oxidative damage in rat liver. Life Sci 72(14):1609–1616
Yáñez JA, Remsberg CM, Miranda ND, Vega-Villa KR, Andrews PK, Davies NM (2008) Pharmacokinetics of selected chiral flavonoids: hesperetin, naringenin and eriodictyol in rats and their content in fruit juices. Biopharm Drug Dispos 29(2):63–82
Punyasiri PAN, Abeysinghe ISB, Kumar V, Treutter D, Duy D, Gosch C, Martens S, Forkmann G, Fischer TC (2004) Flavonoid biosynthesis in the tea plant Camellia sinensis: properties of enzymes of the prominent epicatechin and catechin pathways. Arch Biochem Biophys 431(1):22–30
Rossato MF, Trevisan G, Walker CIB, Klafke JZ, de Oliveira AP, Villarinho JG, Zanon RB, Royes LF, Athayde ML, Gomez MV, Ferreira J (2011) Eriodictyol: a flavonoid antagonist of the TRPV1 receptor with antioxidant activity. Biochem Pharmacol 81(4):544–551
Lee JK (2011) Anti-inflammatory effects of eriodictyol in lipopolysaccharidestimulated raw 264.7 murine macrophages. Arch Pharm Res 34(4):671–679
Jing X, Shi H, Zhu X, Wei X, Ren M, Han M, Ren D, Lou H (2015) Eriodictyol Attenuates β-Amyloid 25–35 Peptide-Induced Oxidative Cell Death in Primary Cultured Neurons by Activation of Nrf2. Neurochem Res 40(7):1463–1471
Kumazoe M, Fujimura Y, Hidaka S, Kim Y, Murayama K, Takai M, Huang Y, Yamashita S, Murata M, Miura D, Wariishi H, Maeda-Yamamoto M, Tachibana H (2015) Metabolic profiling-based data-mining for an effective chemical combination to induce apoptosis of cancer cells. Sci Rep 5:9474
Sanchez AM, Candau RB, Bernardi H (2014) FoxO transcription factors: their roles in the maintenance of skeletal muscle homeostasis. Cell Mol Life Sci 71:1657–1671
Latres E, Amini AR, Amini AA, Griffiths J, Martin FJ, Wei Y, Lin HC, Yancopoulos GD, Glass DJ (2005) Insulin-like growth factor-1 (IGF-1) inversely regulates atrophy-induced genes via the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin (PI3K/Akt/mTOR) pathway. J Biol Chem 280(4):2737–2744
Kumazoe M, Sugihara K, Tsukamoto S, Huang Y, Tsurudome Y, Suzuki T, Suemasu Y, Ueda N, Yamashita S, Kim Y, Yamada K, Tachibana H (2013) 67-kDa laminin receptor increases cGMP to induce cancer-selective apoptosis. J Clin Invest 123(2):787–799
Butò S, Tagliabue E, Ardini E, Magnifico A, Ghirelli C, Van Den Brûle F, Castronovo V, Colnaghi MI, Sobel ME, Ménard S (1998) Formation of the 67-kDa laminin receptor by acylation of the precursor. J Cell Biochem 69(3):244–251
Sanjuán X, Fernández PL, Miquel R, Muñoz J, Castronovo V, Ménard S, Palacín A, Cardesa A, Campo E (1996) Overexpression of the 67-kD laminin receptor correlates with tumour progression in human colorectal carcinoma. J Pathol 179(4):376–380
Ménard S, Castronovo V, Tagliabue E, Sobel ME (1997) New insights into the metastasis-associated 67 kD laminin receptor. J Cell Biochem 67(2):155–165
Ménard S, Tagliabue E, Colnaghi MI (1998) The 67 kDa laminin receptor as a prognostic factor in human cancer. Breast Cancer Res Treat 52(1–3):137–145
Lee ER, Kim JH, Choi HY, Jeon K, Cho SG (2011) Cytoprotective effect of eriodictyol in UV-irradiated keratinocytes via phosphatase-dependent modulation of both the p38 MAPK and Akt signaling pathways. Cell Physiol Biochem 27(5):513–524
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME (1999) Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96(6):857–868
Stitt TN, Drujan D, Clarke BA, Panaro F, Timofeyva Y, Kline WO, Gonzalez M, Yancopoulos GD, Glass DJ (2004) The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell 14(3):395–403
Rommel C, Bodine SC, Clarke BA, Rossman R, Nunez L, Stitt TN, Yancopoulos GD, Glass DJ (2001) Mediation of IGF-1-induced skeletal myotube hypertrophy by PI (3) K/Akt/mTOR and PI (3) K/Akt/GSK3 pathways. Nat Cell Biol 3(11):1009–1013
Acknowledgements
This research was supported in part by Grants-in-Aid for Scientific Research (KAKENHI) from the Japan Society for the Promotion of Science to H. Tachibana (Grant number: JP15H02448). The authors would like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Murata, M., Shimizu, Y., Marugame, Y. et al. EGCG down-regulates MuRF1 expression through 67-kDa laminin receptor and the receptor signaling is amplified by eriodictyol. J Nat Med 74, 673–679 (2020). https://doi.org/10.1007/s11418-020-01417-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-020-01417-6