Abstract
Purpose
Technological progress and high market demand contributed to a significant interest in the production of fertilizers based on humic acids. The aim of this study was to evaluate the possibilities of using humic acids obtained from lignite in the production of new commercial products. For this purpose, it is necessary to determine the quality standard requirements for such material. Properties of humic acids depend on source of origin as well as method of its extraction.
Materials and methods
The humic acids were extracted from polish deposit of lignite–Sieniawa Lubuska by alkaline extraction using for this purpose six kinds of extractants: 0.1 M NaOH and 0.25 M NaOH, 0.1 M KOH and 0.25 M KOH, and 0.1 M Na4P2O7 and 0.25 M Na4P2O7. The humic samples were used in solid powder form and characterized by UV-Vis spectroscopy, 13C NMR spectroscopy, fluorescence spectroscopy, and thermal analysis.
Results and discussion
The determining factor influencing a degree of humic acids extraction from lignite and their structure is type of extractant. The largest efficiency of extraction (about 50%) was obtained with the use of NaOH solutions. All examined humic acids were generally characterized by simple and heterogeneous molecularly structure with low molecular weight and low aromatic polycondensation. Therefore, it can be concluded that humic acids extracted with NaOH and KOH solutions are less condensed than those extracted with Na4P2O7 solutions. It can suggest that humic acids obtained from lignite using solutions of Na4P2O7 are characterized by a low transformation degree and greater amount of carboxyl groups.
Conclusions
Low rank coal can be successfully used in agriculture as a rich source of humic acids. Reagent used in their extraction, apart from high efficiency should have a neutral impact on their structure. Studies on the physicochemical properties of humic acids can be helpful in predicting behaviors of such fertilizer components in the environment and in inventing new products taking the principles of sustainable development into consideration.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Humic substances (HS) are known to perform many useful functions in soil and are considered to be one of the fundamental factors determining practically all properties and transformations occurring within it (Stevenson 1994; Tan 2003; Ghabbour and Davies 2005). Most published studies of HS show wide range of benefits including a growth of beneficial soil microbes, improvements in soil structure including water retention capacity as well as a soil porosity, aeration, compaction, and adhesion (Piccolo et al. 1993; Nardi et al. 2016; Ouni et al. 2014; Ciarkowska et al. 2017). Another experiment has shown numerous benefits of HS application to plants including root respiration, enzyme activity, and resistance to abiotic stress like salinity and extreme pH. These substances can immobilize and neutralize heavy metals, contributing to the process of soil detoxification; however, their main use is to enhance nutrient and micronutrient uptake including their bioavailability and, as a consequence, increase growth, yield, and product quality (Piccolo 1996; Tipping 2002; Canellas et al. 2002, 2014, 2015; Nardi et al. 2016). For these reasons, it seems that the use of humic acids as agents which improve soil properties and affect the amount and quality of crops, it may be a perspective direction in the development of modern agriculture.
One of the most important factors regulating the humus content in soil is organic fertilization. Humic substances (HS) are natural organic compounds which are sourced not only from soil but also from compost, manure, peat and, e.g., coals such as lignite and leonardite (Canellas et al. 2015; Fuentes et al. 2016; Yakimenko et al. 2016; Ciarkowska et al. 2017). Increased production and continuous expansion of the range of such products have been one of the main trends on fertilizer market. Preparations of humic acids derived from natural organic materials, when they are applied to the soil, should be characterized by similar features. Today, the value of application of commercial products containing humic substances in agriculture is less certain. There is currently not enough research to explain possible mechanisms and accurately predict when humic materials might prove beneficial (Lamar et al. 2014; Lyons and Genc, 2016; Yakimenko et al. 2016). It is recommended that such products should be well characterized physically and chemically. This is not an easy task, even though there are many papers on examination of the structure and properties of humic compounds, not all features of these particles can be clearly specified. For this purpose, it is necessary to determine the quality standard requirements for such material.
In Poland, due to economic and technological developments, it seems real to use lignite as a material for humic acids preparation. Poland is currently the second lignite producer in the European Union as well as the seventh in the word. Generally, the lignite-bearing areas cover about one-third of the Poland’s total territory and is concentrated in five regions of central and western Poland (Piwocki 1992; Pietraszewski 2012; Widera et al. 2016). Lignite production in Poland reached up to 62.9 Mt in 2011, comprising 38.6 Mt in Bełchatów, 10.4 Mt in Turów, 9.3 Mt in Konin, 4.5 Mt in Adamów, and 0.09 Mt in Sieniawa (Szuflicki et al. 2012; Widera 2013). Polish lignite seams are of Early and Middle Miocene age and among the genetically most differentiated in the word. Generally, three stratigraphically distinct lignite seams exploited in Poland, namely the third Ścinawa lignite seam (ŚLS-3), the second Lusatian lignite seam (LLS-2), and the first Mid-Polish lignite seam (MPLS-1) represent humic low rank coal, i.e., ortholignite (Widera 2016). For agricultural purpose, lignite with low degree of coalification, especially the earthy sort of them, is the most beneficial. Application of such material as a rich source of humic substances is also a rational way of managing it.
The direct interaction of humic substances with the soil environment is related to their structure and amount of these compounds contained within it. The main elements forming the humic compounds are carbon, hydrogen, nitrogen, and oxygen. From a chemical point of view, humic substances can be described generally as a concentration of aromatic and aliphatic rings, aliphatic chains, bridges, and numerous functional groups. Among the most important functional groups of humus compounds are carboxyl, hydroxyl, phenolic, methoxy, and carbonyl groups (Stevenson 1994; Chen et al. 2002; Tipping 2002; Tan 2003).
Chemical and physical properties of humic substances in commercial products obtained from various resources should be characterized by similar features. A determining factor influencing chemical character of obtained humic acids, as well as the efficiency of their extraction from potential organic sources, is not only a selection of raw material but also the isolation procedure. Industrial technologies of commercial humic compounds production differ from research analytical techniques used for humic substances isolation from soil. Based on literature data, known analytical techniques and patent descriptions related to humic acids formation from organic materials it can be stated that different methods for the isolation of humic acids exist, in which however, the basic operations are similar: the extraction of the humic substances from potential organic material and then treatment of obtained extract by using inorganic acid solution to isolate humic acids (Stevenson 1994; Tan 2003; Ghabbour and Davies, 2005; Zaccone et al. 2007; Lamar et al. 2014). These methods differ from one another by kind of extractants, their concentration and amount in relation to substances subjected to extraction, time of extraction and temperature, as well as technique of proceeding, kind of inorganic acid, way of purification of obtained substances, etc. The type of extraction liquid has however the most significant impact. It can be expected that can expect that both chemical characteristics and, in consequence, behavior of commercial humic products will be different depending on isolation procedure. Moreover, from a technological point of view, the high efficiency of humic acids extraction is a decisive factor influencing on the choice of extraction parameters, which is especially important for economic reasons. As potential fertilizer components, humic acids derived from different sources under different conditions, should be well characterized. For this purpose, it is necessary to determine the quality standard requirements for such material. This approach could not only contribute to understanding the chemical nature of commercial humic products, but might also be useful for the development of theoretical considerations on their application in agriculture or environmental technologies (Lyons and Genc, 2016; Yakimenko et al. 2016). For this purpose, UV-Vis, 13C NMR, fluorescence spectroscopy, and thermal analysis can be used as diagnostic criteria.
2 Materials and methods
A determining factor influencing on the degree of humic acids extraction from lignite and their physicochemical properties is the selection of raw material and alkaline agent. The aim of present examinations was the assessment of physicochemical properties of humic acids obtained from lignite using 0.1 M NaOH, 0.25 M NaOH, 0.1 M KOH, 0.25 M KOH, 0.1 M Na4P2O7, and 0.25 M Na4P2O7. Humic acids used in the present work were obtained from polish deposit of lignite–Sieniawa Lubuska. Obtained results were compared with results of analyzes of technical humic acids (KHtech) and their salt (KHsalt) from Sigma-Aldrich.
2.1 Characterization of lignite
The specificity of the Sieniawa Lignite Mine is the lack of connection of the mine with the energy sector, for which the lignite would be the basic raw material for energy production. This lignite mine is the smallest in Poland and the only one where all production is supplied to the local heat boiler station, individual customers and to companies using lignite for non-energy purpose. The second group of Lusatian lignite seams of Early and Middle Miocene age is exploited in Sieniawa opencast mine and represents humic ortholignite of low-coal rank (Macuda et al. 2011; Widera 2013).
For characterization of Sieniawa lignite, sample in air-dry state, crushed to a grain size below 0.5 mm were used. As a part of physicochemical characteristic of lignite content of organic carbon (Cdorg, calculated as moist-free), hydrogen (in the analytical state, Ha), moisture (moisture in the analytical state, Wa and the total moisture in the ash-free state, Wtraf) as well as value of pH in water (pH-H2O) and KCl solution (pH-KCl) were determined. Moreover, calorific value (in analytical state - Qir and in the working state Qir) was determined.
2.2 Extraction of humic acids
For the extraction of humic acids from the carbon material, analytical techniques typically applied in studies of organic matter and soil humic compounds were used. The concept of obtaining humic acids (HA) from lignite involves two stages. In the first step, lignite was treated with one of following extractants (0.1 M NaOH and 0.25 M NaOH,0.1 M KOH and 0.25 M KOH, and 0.1 M Na4P2O7 and 0.25 M Na4P2O7) (1:20 w/v) by constant stirring for 12 h. The mixture was then centrifuged at 2500×g for 10 min and the soluble supernatant (humic substances, HS) was separated from the insoluble sediment (humin). In a second stage, HA were precipitated from the extract by acidification of the environment by adding 2 M HCl solution to pH about 2. The coagulation lasted 24 h and then HA were separated from the soluble fulvic acids (FA) by centrifugation, as above. Obtained gel of humic acids was dissolved in a 2% Na2CO3 solution and transferred to the volumetric flask (100 or 250 cm3).
The extraction method used in the study belongs to indirect methods. It means that the content of individual fractions of humic substances is determined by determining the content of one of its components. Results of the content of humic substances (HS = HA + FA) and humic acids (HA) obtained from Polish lignite depending on extractant type was expressed as a percentage of Corg in this groups of compounds per the total organic carbon content in the output sample. The efficiency of extraction was measured by amount of organic carbon in whole extract (HS) and in only HA’s solution. The content of fulvic acids (FA) was calculated by subtraction of HA content from the HS content. The content of organic carbon in individual fractions was determined using Alten’s method.
2.3 Characterization of humic acids
The humic acids obtained from lignite were characterized by UV-Vis, 13C NMR, fluorescence spectroscopy, and thermal analysis.
UV-Vis analysis was performed on UV-Vis spectrometer Jasco V-600 in order to obtain the A4/A6 absorbances ratio at wavelengths of 465 and 665 nm. To determine this ratio, 0.015 g humic acids were suspended in 100 ml of NaHCO3 0.05 M aqueous solution.
13C NMR spectrum of solid HA sample was determined on Bruker MSL-300 spectrometer operated at room temperature. Based on the areas defined under peaks, the contribution of carbon was calculated: Cal–alifatic bonds (0–95 ppm), Car–aromatic bonds (95–160 ppm); Clig–aromatic lignin bonds (140–160 ppm); C-COOH–carboxyl groups (160–210 ppm).
The fluorescence spectra of excitation-emission-matrix (EEM) were determined on Edinburgh Instrument F900 fluorescence spectrophotometer. EEM spectra were obtained on water extracts of HA at a concentration of 100 mg/l.
The thermal behavior of samples, as a function of temperature, was determined by an air flow simultaneous thermal analyzer NETZSCH STA 449 F3 Jupiter®, coupled with QMS 403 Aëolos® quadrupole mass spectrometer in order to detect the gas evolved from the thermal analyzer in the range m/z = 12–64.Thermogravimetric curves were obtained from approximately 50 mg of solid humic acids sample. Measurements were carried out in air atmosphere (30 ml/min) at 5 °C/min heating rate in the temperature range of 35–800 °C. DTA type S thermocouple and Al2O3 crucibles were used.
3 Results and discussion
The features that indicate unsuitability of lignite as a fuel are simultaneously considered as beneficial in terms of using lignite in agriculture. Low rank coal can be successfully used in agriculture as an indirect fertilizer component. Physicochemical quality indicators of Sieniawa lignite are shown in Table 1. An indicator that indirectly and simply characterizes the degree of lignite coalification is the moisture content. In geological documentation the total moisture in the working state (Wtr) is usually determined. In order to classify lignite by type, the total moisture in the working state converted to the ash-free state (Wtraf) is used. This parameter varies conventionally in the range of 45–60% mass for soft coals and 20–45% mass for hard coals. Based on accepted division, lignite from Sieniawa mine should be classified as soft petrographic variety with a relatively low degree of coalification.
According to the literature, the heat of combustion of brown coals containing from 10 to 75% of the total moisture is on level from 6700 to 25,000 kJ/kg. Other sources of the literature in classification of brown coals report that the group of energetic brown coals may already include those whose calorific value in working condition and when total moisture in the working state–Wtr = 50%, is above 6700 kJ/kg (Qir) (Hoffmann and Huculak-Mączka, 2012). Fuel parameters of examined lignite confirm that it belongs rather to the low-calorie coals. The main criterion for assessing the possibility of using lignite as a raw material for the preparation of humic acids is the content of organic matter expressed as the content of organic carbon. The organic carbon content (Cdorg) calculated as moist-free for Sieniawa lignite was 65.2%.
The efficiency of humic acids extraction is determined not only by raw material which are extracted from but also by the physicochemical parameters of the process. The choice of appropriate extractant is extremely important. In the extraction process of humic acids following solutions were used: 0.1 M NaOH and 0.25 M NaOH, 0.1 M KOH and 0.25 M KOH, and 0.1 M Na4P2O7 and 0.25 M Na4P2O7. The use of various extraction systems for production of humic acids from lignite is associated with obtaining different fractions with varying degree of transformation.
Results of the content of humic (HA) and fulvic acids (FA) obtained from Polish lignite depending on extractant type are showed in Table 2. The largest efficiency of extraction, which was measured by amount of organic carbon in whole extract (HS = HA + FA) and in only HAs solution, was obtained with the use of NaOH solution. Extractions with Na4P207solutions lead to isolation of a considerable part of humic substances. Sodium pyrophosphate solution is however less effective than solutions of bases. Reagent used in the extraction, apart from high efficiency, should have a neutral impact on the physicochemical properties of obtained humic acids. In order to assess the impact of extractant on the isolated humic acids, spectroscopic (UV-Vis, 13C NMR and fluorescence) and thermal analyzes were used. Obtained results were compared with results of analyzes of technical humic acids (KHtech) and their salt (KHsalt) from Sigma-Aldrich.
One of the oldest analytical methods used to describe humus substances is UV-Vis absorption spectroscopy. Simple measurement techniques, high speed and relatively low cost are considered as advantages of using this method. Moreover, optical methods that use absorbance measurements of humic substances in the UV-Vis range are non-destructive and do not require large quantities of output samples. Optical parameters in UV-Vis allow to determine the degree of humification of examined humic acids, some of their structural features and indirectly their quality.
In the qualitative analysis of humus compounds, absorption parameters like A2/4, A2/6, and A4/6 are often used. These optical parameters are absorbance ratios measured at wavelengths of 280, 472, and 664 nm, respectively.
The first parameter, A2/4, determines the content of organic compounds resistant to humification—the lignin type in relation to the content of substances at the beginning of humification. This value is negatively correlated with the progress of the humification process. The A2/6 parameter expresses the ratio of the non-humified and strongly humified material (Kononova 1966; Chen et al. 1977; Yakimenko et al. 2016). The highest values of these parameters are characterized by humic acids obtained from lignite using 0.1 M KOH.
The A4/6 ratio is one of the most commonly used parameter in science of humic substances. Its value does not depend on the content of humus compounds but depends on their construction and correlates with the size of their molecular weight. It is assumed that the A4/6 value is related to the overall carbon content and molecular weight of the humus compounds and this relationship shows a negative correlation. The correlation between the A4/6 and aromaticity as well as degree of humification is a disputable issue and will not be discussed in this paper (Chen et al. 1977; Chin et al. 1994; Cieslewicz and Gonet, 2004; Zbytniewski and Buszewski, 2005).
The obtained A4/6 results (Table 3) for examined humic acids samples are higher than traditional soil humic acids (about 4–5) (Chen et al. 1977, 2002; Pertusatti and Prado 2007). This fact confirms that examined humic acids present rather a low molecular weight. The lower molecular weight of humus compounds obtained from lignite may, in the perspective of their use for fertilization, affect their greater mobility. Humic acids obtained from lignite using solutions of Na2P4O7 with the highest A4/6 values should be abundant in carboxyl groups content and low in aromatic structures.
More specific information concerning the structure of the HA could be obtained from 13C NMR spectroscopy (Qiang et al., 1993; Ricca et al. 2000; Piccolo 2002; Zbytniewski and Buszewski, 2005; Pertusatti and Prado, 2007; Kwiatkowska-Malina 2009). Figure 1 shows solid-state 13C NMR spectra of humic acids extracted from lignite using different solutions together with spectra of technical humic acids (KHtech) and their salt (KHsalt) from Sigma-Aldrich.13C NMR spectra of humic acids obtained from Sieniawa lignite generally are characterized by only one clear peak at around 30 ppm corresponding to aliphatic carbon (Cal), in particular the methyl and methylene groups. There is also some absorption intensity in the range of 95–160 ppm. However, it is not possible to distinguish the characteristic peak in this area, and the intensity of absorption in this range of chemical shifts is significantly lower than for the range of 0–95 ppm. Range between 95 and 160 ppm corresponds to aromatic carbon bonds (Car) and may indicate that examined humic acids are characterized by less condensed structures.
The differences between samples of humic acids were slight but still distinct enough to compare the harms brought about by the various extractants. Hence, base-treated samples appeared to be composed of a higher portion of alkyls, ethers and alcohols at the expense of aromatic components, the alteration getting more distinct with decreasing NaOH and KOH concentration. Therefore, it can be concluded that humic acids extracted with NaOH and KOH solutions are less condensed than those extracted with Na4P2O7 solutions. SP0.1 and SP0.25 samples were also characterized by a certain amount of carboxyl groups, as evidenced by a slight increase in absorption intensity in the range of 160–190 ppm. It can suggest that humic acids obtained from lignite using solutions of Na4P2O7 are characterized by a low transformation degree. Comparison of humic substances from Sigma-Aldrich spectra with those obtained from lignite shows that, depending on the preparation conditions, they have a similar structure. For the KHtech sample, the most similar 13C NMR spectrum was obtained for lignite humic acids using 0.1 M Na4P2O7. Only spectrum of KHsalt has three major peaks of alkyl carbons at around 30 ppm, aromatic carbons at around 125 ppm, and carboxyl groups at around 180 ppm, as depicted in Fig. 2. This finding is also supported by the NMR data—contribution of carbon in various bonds, calculated on the basis of the defined areas under peaks (Table 4). For KHsalt sample, the integration of the NMR spectrum between 0 and 95 ppm showed 39.37% of total carbons, 54.45% between 95 and 160 ppm, and 6.18% between 160 and 190 ppm. In lignite humic acids, the carboxylic groups assigned to peak in the range of 160–190 ppm, constituted from 2.10 to 8.33%.
However, interpretation of the 13C NMR spectra is limited to determining the number of equivalent 13C atoms and, based on their chemical shift, defining what type of molecular structure elements they belong to. Therefore, it is recommended to treat NMR results as reflecting the statistical conditions and to determine the functional groups content in humic acids using more precise methods such as, e.g., potentiometric titration.
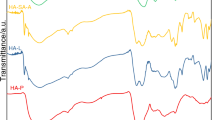
Fluorescence spectroscopy is the sensitive method used in structural investigation of macromolecules. In the case of humic substances, this method reflects their structural processes of intramolecular energy transfer and transmutations. The dependency of relative fluorescence intensity on the wavelength of excitation and/or emission is responsive to the origin of the humic substance, even for humic substances from similar sources. The 3-D EEM spectra of humic acids from lignite feature a unique chromophore centered at low excitation/emission wavelengths pair (Fig. 3). This fact may be attributed to the increased content of simple aromatics in the extracted HA. It is in accordance with the previous studies, and may suggest the presence of simple structural components of small molecular size bearing electron-donating substituents, such as hydroxyl, methoxy, and aromatic polycondensation and conjugated chromophores in humic acids (Senesi et al. 1991; Chen et al. 2002; Sierra et al. 2005; Kwiatkowska et al., 2008; Wang et al. 2014). All spectra were characterized by two peaks: one at 330ex/440em and second, smaller at 340ex/450em with the exception of spectrum for SP0.25, which was distinguished by the fact that the first peak was shifted to shorter excitation wavelengths and centered at 310ex/440em.
Among the EEM spectra of lignite humic acids, the SP0.1 spectrum should be distinguished, where the delicate peak contour at 470ex/540em can be observed and which with higher fluorescence intensity occurs in the spectrum of technical humic acids (KHtech) and their salt (KHsalt) from Sigma-Aldrich (Fig. 4). The presence of a peak in this area may indicate the presence of lignin structures in these humic acids. According to 13C NMR spectra, from all examined samples, humic acids obtained using Na4P2O7 solutions had the largest number of carbon contribution in aromatic lignin bonds (the integration of the NMR spectrum between 140 and 160 ppm).
It must be remembered that fluorescence analysis of such complex macromolecular biopolymers, like humic acids, encounter significant interpretive difficulties. It is caused by the strong interaction of molecules with the environment and formation of a complex structure of excited states.
Thermo-oxidative behavior of examined humic substances was investigated employing the thermogravimetry. The TG/DTA curves for lignite humic acids were obtained in order to determine their thermal stability, as shown in Figs. 5, 6, and 7 and Fig. S1–S3 in the Electronic Supplementary Material. All TG/DTA curves for lignite humic acids were characterized by an endothermic peak at a temperature range of 100–120 °C. This peak is the result of dehydration and the difference in the amount of accompanying mass loss results from the different degree of water binding through the sample. Water may be present in free form or chemically bound with porous humic acids. Its large part (physically bound) was removed from it during the drying process at 105 °C before analysis. The peak at about 170 °C is associated with mass loss due to the removal of crystallization water. The first significant temperature range for the transformation of organic matter is attributed to a range of 200 to 400 °C and is related to the degradation of less thermally stable structures, generally characterized by a less condensed structure. The loss of mass in this temperature range is attributed to changes such as degradation of carbohydrates, dehydration of aliphatic structures, and decarboxylation of carboxylic groups (Francioso et al. 2005; Pertusatti and Prado, 2007; Rotaru et al. 2008; Klavins et al. 2010; Sirbu et al., 2010). These changes in the DTA graph are reflected in the form of exothermic peaks at a temperature close to 300 °C, especially sharp for samples of humic acids obtained using NaOH solutions. The resulting mass loss for both samples—SN0.1 and SN0.25—was comparable and amounted to 14.03 and 15.84%, respectively, and can be attributed to the loss of simple hydrocarbon compounds. Changes occurring at temperatures higher than 400 °C affect the degradation of more condensed structures and oxidation of aromatic components and breakdown of C-C bonds. These reactions are probably related to the occurrence of long-chain hydrocarbons and nitrogen compounds. Above 500 °C, a pyrolysis of aromatic components of lignin and other polyphenols occurs and the aromatic core of humic acids structure is destroyed. Exothermic effect occurring at temperatures above 600 °C is probably due to high-temperature changes associated with polycondensation and depolymerization reactions.
In the TG/DTA graph for lignite humic acids (except for samples SN0.1 and SN0.25), one high exothermic peak over a wide range of temperatures can be observed. Determination of a detailed construction of these humic acids is practically impossible on the basis of TG/DTA curves. However, curves give insight on how complex humic substances are and the temperature range of the peak indicates a contribution of both aliphatic and aromatic structure. For SN0.1 and SN0.25 samples, a significant mass loss above 700 °C was observed, which in both cases was approximately 17%. This is probably the result of the decomposition processes of minerals (inorganic compounds), mainly silicates, and shows the possibility of also undesirable compounds extraction under these conditions.
The mass losses of lignite humic acids in the temperature ranges are shown in Table S1 (Electronic Supplementary Material). For comparison, results for technical humic acids (KHtech) and their salt (KHsalt) from Sigma-Aldrich are also presented in the table below.
The TG/DTA curves for these samples were shown in Figs. 8 and 9. Comparing TG/DTA curves for KHtech with those obtained by extraction from lignite, a relatively high similarity with sample SP0.1 was noticed. For these samples, TG/DTA curves were characterized by two similar exothermic effects at about 100 °C and between 400 and 600 °C. The mass losses for KHtech at mentioned temperature range were respectively m1 = 6.28%, m2 = 47.67%, and for SP0.1 m1 = 5.84%, m2 = 42.16%. The basic difference between the samples was observed in the range of 200–400 °C, in which a clear peak was observed only for the KHtech sample. Comparing the results of thermal analysis of humic acids obtained with the use of different extractants, it is difficult to assess which preparations are more advantageous from the agricultural point of view. However, it can be distinguished that those for which significant loss mass in the lower temperature range is observed, have less condensed structures with aliphatic character and carboxyl groups. Peaks shift toward a high temperature means that humic acids are characterized by more thermally stable structure as is the case for aromatic compounds.
4 Conclusions
Number of physicochemical properties of humic substances, the changing trends on the fertilizer market, innovation factor and ecological conditions recently contributed to the increasing interest in humic acids. Over the last decades, commercial humic products from various resources found multiple applications in agriculture and environmental technologies. In Poland, due to economic and technological developments, it seems real to use lignite as a material for humic acids preparation. A determining factor influencing the degree of humic acids extraction from lignite is, with exception of raw material, the choice of extractant. The largest efficiency of extraction (about 50%) was obtained with the use of NaOH solutions. Extraction liquid used in isolation process of humic acids from lignite should be characterized by high efficiency and selectivity of extraction and neutral influence on the physicochemical properties of the obtained compounds. When using more aggressive extractant the greater efficiency of the extraction process can be expected, however, there is a risk that it may simultaneously cause partial degradation of humic substances structure and contribute to the contamination of the final product with undesirable compounds. Less destructive extractant may provide a good quality product at the expense of process efficiency. Humic acids derived from different sources under different conditions, as a potential fertilizer component, should be well characterized. For this purpose, UV-Vis, 13C NMR, fluorescence spectroscopy, and thermal analysis as a diagnostic criteria were used. The structure of obtained humic acids was undoubtedly influenced by the type and concentration of extractants used for their isolation. Aliphatic connections dominated in the structure of humic acids derived from lignite. Their participation in these samples ranged from 58.52% for sample obtained using 0.25 M Na4P2O7 to 78.90% for this obtained using 0.1 M NaOH. All examined humic acids were generally characterized by simple and heterogeneous structure with low molecular weight and low aromatic polycondensation.
Participation of aliphalic and aromatic units in the structure of humic acids, after introduction into the soil, has a double meaning in terms of generalized transformations occurring within it. The presence of functional groups is related to the ion exchange capacity and the possibility of formation of mineral-organic connections by commercial humic products. On the other hand, the aromatic structures guarantee the stability of the organic substance and are a valuable source of energy and matter for microorganisms.
References
Canellas LP, Olivares FL (2014) Physiological responses to humic substances as plant growth promoter. Chem Biol Technol Agric 1:1–11
Canellas LP, Olivares FL, Aguiar NO, Jones DL, Nebbioso A, Mazzei P, Piccolo A (2015) Humic and fulvic acids as biostimulants in horticulture. Sci Hortic 196:15–27. https://doi.org/10.1016/j.scienta.2015.09.013
Canellas LP, Olivares FL, Okorokova-Facanha AL, Facanha AR (2002) Humic acids isolated from earthworm compost enhance root elongation, lateral root emergence, and plasma membrane H+-ATPase activity in maize roots. Plant Phys 130(4):1951–1957. https://doi.org/10.1104/pp.007088
Chen J, Gu B, LeBoeuf EJ, Pan H, Dai S (2002) Spectroscopic characterization of the structural and functional properties of natural organic matter fractions. Chemosphere 48(1):59–68. https://doi.org/10.1016/S0045-6535(02)00041-3
Chen Y, Senesi N, Schnitzer M (1977) Information provided on humic substances by E4/E6 ratios. Soil SciSoc Am J 41(2):352–358. https://doi.org/10.2136/sssaj1977.03615995004100020037x
Chin YP, Alken G, O’Loughlin E (1994) Molecular weight, polydispersity, and spectroscopic properties of aquatic humic substances. Environ Sci Technol 28(11):1853–1858. https://doi.org/10.1021/es00060a015
Ciarkowska K, Sołek-Podwika K, Filipek-Mazur B, Tabak M (2017) Comparative effects of lignite-derived humic acids and FYM on soil properties and vegetable yield. Geoderma 303:85–92. https://doi.org/10.1016/j.geoderma.2017.05.022
Cieslewicz J, Gonet S (2004) Properties of humic acids as biomarkers of lake catchment management. Aquat Sci 66(2):178–184. https://doi.org/10.1007/s00027-004-0702-0
Francioso O, Montecchio D, Gioacchini P, Ciavatta C (2005) Thermal analysis (TG-DTA) and isotopic characterization (13C-15N) of humic acids from differend origins. Appl Geochem 20(3):537–544. https://doi.org/10.1016/j.apgeochem.2004.10.003
Fuentes M, Baigorri R, González-Gaitano G, García-Mina JM (2016) New methodology to assess the quantity and quality of humic substances in organic materials and commercial products for agriculture. J Soils Sediments. https://doi.org/10.1007/s11368-016-1514-2
Ghabbour EA, Davies G (2005) Humic substances: molecular details and applications in land and water conservation. Taylor & Francis, Inc., New York
Hoffmann K, Huculak-Mączka H (2012) The utilization possibility of waste lignite as a raw material in the process of obtaining humic acids preparations. Pol J. Chem Tech 14:1–6
Klavins M, Ansone L, Tjutrins J, Silamikele I, Purmalis O (2010) Differential thermal analysis of peat and peat humic acids in relation to their origin. In: Klavins M (ed) Mires a nd Peat, 1st edn. University of Latvia Press, Riga pp 207–214
Kononova MM (1966) Soil organic matter, its nature, its role in soil formation and in soil fertility. Pergamon, Oxford
Kwiatkowska-Malina J (2009) Structure and properties of humic substances from brown coal 11 years after addition into soil. Pol. J Soil Sci 42:167–174
Kwiatkowska J, Provenzano MR, Senesi N (2008) Long term effects of a brown coal-based amendment on the properties of soil humic acids. Geoderma 148(2):200–205. https://doi.org/10.1016/j.geoderma.2008.10.001
Lamar RT, Olk DC, Mayhew L, Bloom PR (2014) A new standardized method for quantification of humic and fulvic acids in humic ores and commercial products. J AOAC Int 97(3):721–730. https://doi.org/10.5740/jaoacint.13-393
Lyons G, Genc Y (2016) Commercial humates in agriculture: real substance or smoke and mirrors?—a review. Agron 6(4):50. https://doi.org/10.3390/agronomy6040050
Macuda J, Nodzeński A, Wagner M, Zawisza L (2011) Sorption of methane on lignite from Polish deposits. Int J Coal Geol 87(1):41–48. https://doi.org/10.1016/j.coal.2011.04.010
Nardi S, Pizzeghello D, Schiavon M, Ertani A (2016) Plant biostimulants: physiological responses induced by protein hydrolyzed-based products and humic substances in plant metabolism. Sci Agric 73(1):18–23. https://doi.org/10.1590/0103-9016-2015-0006
Ouni Y, Ghnaya T, Montemurro F, Abdelly C, Lakhdar A (2014) The role of humic substances in mitigating the harmful effects of soil salinity and improve plant productivity. Int J Plant Prod 8:353–374
Pertusatti J, Prado A (2007) Buffer capacity of humic acid: thermodynamic approach. J Colloid Interf Sci 314(2):484–489. https://doi.org/10.1016/j.jcis.2007.06.006
Piccolo A (1996) Humus and soil conservation. In: Piccolo A (ed) In humic substances in terrestrial ecosystems. Elsevier, Amsterdam. https://doi.org/10.1016/B978-044481516-3/50006-2
Piccolo A (2002) The supramolecular structure of humic substances. Adv Agron 75:57–134. https://doi.org/10.1016/S0065-2113(02)75003-7
Piccolo A, Celano G, Pietramellara G (1993) Effectsof fractions of coal-derived humic substances on seedgermination and growth of seedlings (Lactuga sativa and Lycopersicumesculentum). Biol Fertil Soils 16(1):11–15. https://doi.org/10.1007/BF00336508
Pietraszewski A (2012) Polish lignite mining industry—2011WęgielBrunatny 79(http://www.ppwb.org.pl/wb/79/14.php (in polish))
Piwocki M (1992) Extent and correlations of main groups of the tertiary lignite seamson Polish platform area. Prz Geol 40:281–286 (in Polish)
Qiang T, Xiao Quan S, Zhe Ming N (1993) Comparative characteristic studies on soil and commercial humic acids. Fresenius J Anal Chem 347:330–336. https://doi.org/10.1007/BF00323816
Ricca G, Severini F, Silvestro G, Yuan CM, Adani F (2000) Derivatization and structural studiem by spectroscopic methods of humic acids from Leonardite. Geoderma 98(3-4):115–125. https://doi.org/10.1016/S0016-7061(00)00055-0
Rotaru A, Nicolaescu I, Rotaru P, Neaga C (2008) Thermal characterization of humic acids and other components of raw coal. J Therm Anal Calorim 92(1):297–300. https://doi.org/10.1007/s10973-007-8816-y
Senesi N, Miano TM, Provenzano MR, Brunetti G (1991) Characterization, differentiation, and classification of humic substances by fluorescence spectroscopy. Soil Sci 152(4):259–271. https://doi.org/10.1097/00010694-199110000-00004
Stevenson FJ (1994) Humus chemistry: genesis, composition, reactions, 2nd edn. Wiley, New York
Sierra MMD, Giovanela M, Parlanti E, Soriano-Sierra EJ (2005) Fluorescence fingerprint of fulvic and humic acids from varied origins as viewed by single-scan and excitation/emission matrix techniques. Chemosphere 58(6):715–733. https://doi.org/10.1016/j.chemosphere.2004.09.038
Sîrbu C, Cioroianu T, Rotaru P (2010) About the humic acids and thermal behaviour of some humic acids. Physics AUC 20:120–126 http://cis01.central.ucv.ro/pauc/vol/2010_20_part1/2010_part1_120_126.pdf
Szuflicki M, Malon A, Tymiński M (2012) The balance of mineral resources deposits in Poland as of 31.12.2011. Polish Geological Institute 1–447 (http://surowce.pgi.gov.pl (in Polish))
Tan KH (2003) Humic matter in soil and the environment: principles and controversies. Marcel Dekker, New York. https://doi.org/10.1201/9780203912546
Tipping E (2002) Cation binding by humic substances. Cambridge University Press, Cambridge. https://doi.org/10.1017/CBO9780511535598
Wang Z, Cao J, Meng F (2014) Interactions between protein-like and humic-like components in dissolved organic matter revealed by fluorescence quenching. Water Res 68:404–413
Widera M (2016) An overview of lithotype associations of Miocene lignite seams exploited in Poland. Andean Geol 22:213–225
Widera M (2013) Changes of the lignite seam architecture—a case study from Polish lignite deposits. Int J Coal Geol 114:60–73. https://doi.org/10.1016/j.coal.2013.02.004
Widera M, Kasztelewicz Z, Ptak M (2016) Lignite mining and electricity generation in Poland: the current state and future prospects. Energy Policy 92:151–157. https://doi.org/10.1016/j.enpol.2016.02.002
Yakimenko O, Khundzhua D, Izosimov A, Yuzhakov V, Patsaeva S (2016) Source indicator of commercial humic products: UV-Vis and fluorescence proxies. J Soils Sediments. https://doi.org/10.1007/s11368-016-1528-9
Zaccone C, Cocozza C, D’Orazio V, Plaza C, Cheburkin A, Miano TM (2007) Influence of extractant on quality and trace elements content of peat humic acids. Talanta 73(5):820–830. https://doi.org/10.1016/j.talanta.2007.04.052
Zbytniewski R, Buszewski B (2005) Characterization of natural organic matter (NOM) derived from sewage sludge compost. Part 1: chemical and spectroscopic properties. Bioresour Technol 96(4):471–478. https://doi.org/10.1016/j.biortech.2004.05.018
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Elżbieta Jamroz
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Huculak-Mączka, M., Hoffmann, J. & Hoffmann, K. Evaluation of the possibilities of using humic acids obtained from lignite in the production of commercial fertilizers. J Soils Sediments 18, 2868–2880 (2018). https://doi.org/10.1007/s11368-017-1907-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-017-1907-x