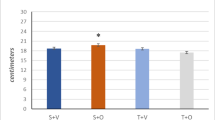
Abstract
Adapting to stress, including cold environmental temperature (eT), is crucial for the survival of mammals, especially small rodents. Long-lived mutant mice have enhanced stress resistance against oxidative and non-oxidative challenges. However, much less is known about the response of those long-lived mice to cold stress. Growth hormone receptor knockout (GHR-KO) mice are long-lived with reduced growth hormone signaling. We wanted to test whether GHR-KO mice have enhanced resistance to cold stress. To examine the response of GHR-KO mice to cold eT, GHR-KO mice were housed at mild cold eT (16 °C) immediately following weaning. Longevity results showed that female GHR-KO and wild-type (WT) mice retained similar lifespan, while both male GHR-KO and WT mice had shortened lifespan compared to the mice housed at 23 °C eT. Female GHR-KO and WT mice housed at 16 °C had upregulated fibroblast growth factor 21 (FGF21), enhanced energy metabolism, reduced plasma triglycerides, and increased mRNA expression of some xenobiotic enzymes compared to females housed at 23 °C and male GHR-KO and WT mice housed under the same condition. In contrast, male GHR-KO and WT mice housed at 16 °C showed deleterious effects in parameters which might be associated with their shortened longevity compared to male GHR-KO and WT mice housed at 23 °C. Together, this study suggests that in response to mild cold stress, sex plays a pivotal role in the regulation of longevity, and female GHR-KO and WT mice are more resistant to this challenge than the males.








Similar content being viewed by others
References
Brown-Borg HM. Longevity in mice: is stress resistance a common factor? Age (Dordr). 2006;28(2):145–62.
Coschigano KT, Clemmons D, Bellush LL, Kopchick JJ. Assessment of growth parameters and life span of GHR/BP gene-disrupted mice. Endocrinology. 2000;141(7):2608–13.
Coschigano KT, et al. Deletion, but not antagonism, of the mouse growth hormone receptor results in severely decreased body weights, insulin, and insulin-like growth factor I levels and increased life span. Endocrinology. 2003;144(9):3799–810.
Salmon AB, et al. Fibroblast cell lines from young adult mice of long-lived mutant strains are resistant to multiple forms of stress. Am J Physiol Endocrinol Metab. 2005;289(1):E23-29.
Hauck SJ, Aaron JM, Wright C, Kopchick JJ, Bartke A. Antioxidant enzymes, free-radical damage, and response to paraquat in liver and kidney of long-living growth hormone receptor/binding protein gene-disrupted mice. Horm Metab Res. 2002;34(9):481–6.
Fang Y, et al. Lifespan of long-lived growth hormone receptor knockout mice was not normalized by housing at 30 degrees C since weaning. Aging Cell. 2020;19(5): e13123.
Klein Hazebroek M, Keipert S. Adapting to the cold: a role for endogenous fibroblast growth factor 21 in thermoregulation? Front Endocrinol (Lausanne). 2020;11:389.
Meyer CW, Ootsuka Y, Romanovsky AA. Body temperature measurements for metabolic phenotyping in mice. Front Physiol. 2017;8:520.
Fang Y, et al. Effects of rapamycin on growth hormone receptor knockout mice. Proc Natl Acad Sci USA. 2018;115(7):E1495–503.
Masternak MM, et al. Metabolic effects of intra-abdominal fat in GHRKO mice. Aging Cell. 2012;11(1):73–81.
Lee P, et al. Mild cold exposure modulates fibroblast growth factor 21 (FGF21) diurnal rhythm in humans: relationship between FGF21 levels, lipolysis, and cold-induced thermogenesis. J Clin Endocrinol Metab. 2013;98(1):E98-102.
Lee P, et al. Irisin and FGF21 are cold-induced endocrine activators of brown fat function in humans. Cell Metab. 2014;19(2):302–9.
Hanssen MJ, et al. Serum FGF21 levels are associated with brown adipose tissue activity in humans. Sci Rep. 2015;5:10275.
Salminen A, Kaarniranta K, Kauppinen A. Regulation of longevity by FGF21: interaction between energy metabolism and stress responses. Ageing Res Rev. 2017;37:79–93.
Potthoff MJ, et al. FGF21 induces PGC-1alpha and regulates carbohydrate and fatty acid metabolism during the adaptive starvation response. Proc Natl Acad Sci USA. 2009;106(26):10853–8.
Makela J, et al. Fibroblast growth factor-21 enhances mitochondrial functions and increases the activity of PGC-1alpha in human dopaminergic neurons via Sirtuin-1. Springerplus. 2014;3:2.
Ji LL, Kang C. Role of PGC-1alpha in sarcopenia: etiology and potential intervention - a mini-review. Gerontology. 2015;61(2):139–48.
Liang H, Ward WF. PGC-1alpha: a key regulator of energy metabolism. Adv Physiol Educ. 2006;30(4):145–51.
Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84(1):277–359.
Kalinovich AV, de Jong JM, Cannon B, Nedergaard J. UCP1 in adipose tissues: two steps to full browning. Biochimie. 2017;134:127–37.
Zhang Y, et al. The starvation hormone, fibroblast growth factor-21, extends lifespan in mice. Elife. 2012;1: e00065.
Schlein C, et al. FGF21 lowers plasma triglycerides by accelerating lipoprotein catabolism in white and brown adipose tissues. Cell Metab. 2016;23(3):441–53.
Steinbaugh MJ, Sun LY, Bartke A, Miller RA. Activation of genes involved in xenobiotic metabolism is a shared signature of mouse models with extended lifespan. Am J Physiol Endocrinol Metab. 2012;303(4):E488-495.
Jarrar YB, Lee SJ. Molecular functionality of cytochrome P450 4 (CYP4) genetic polymorphisms and their clinical implications. Int J Mol Sci. 2019;20(17):4274. https://doi.org/10.3390/ijms20174274.
Hsu MH, Savas U, Griffin KJ, Johnson EF. Human cytochrome p450 family 4 enzymes: function, genetic variation and regulation. Drug Metab Rev. 2007;39(2–3):515–38.
Hardwick JP. Cytochrome P450 omega hydroxylase (CYP4) function in fatty acid metabolism and metabolic diseases. Biochem Pharmacol. 2008;75(12):2263–75.
Huang S, Howington MB, Dobry CJ, Evans CR, Leiser SF. Flavin-containing monooxygenases are conserved regulators of stress resistance and metabolism. Front Cell Dev Biol. 2021;9: 630188.
Conti B, et al. Transgenic mice with a reduced core body temperature have an increased life span. Science. 2006;314(5800):825–8.
Sanchez-Alavez M, Alboni S, Conti B. Sex- and age-specific differences in core body temperature of C57Bl/6 mice. Age (Dordr). 2011;33(1):89–99.
Gagliano-Juca T, et al. Effects of testosterone administration (and its 5-alpha-reduction) on parenchymal organ volumes in healthy young men: findings from a dose-response trial. Andrology. 2017;5(5):889–97.
Nucci RAB, et al. Effects of testosterone administration on liver structure and function in aging rats. Aging Male. 2017;20(2):134–7.
Bartke A, Steele RE, Musto N, Caldwell BV. Fluctuations in plasma testosterone levels in adult male rats and mice. Endocrinology. 1973;92(4):1223–8.
Al-Regaiey KA, Masternak MM, Bonkowski M, Sun L, Bartke A. Long-lived growth hormone receptor knockout mice: interaction of reduced insulin-like growth factor i/insulin signaling and caloric restriction. Endocrinology. 2005;146(2):851–60.
Zhou Y, et al. A mammalian model for Laron syndrome produced by targeted disruption of the mouse growth hormone receptor/binding protein gene (the Laron mouse). Proc Natl Acad Sci USA. 1997;94(24):13215–20.
Basu R, Qian Y, Kopchick JJ. Mechanisms in endocrinology: lessons from growth hormone receptor gene-disrupted mice: are there benefits of endocrine defects? Eur J Endocrinol. 2018;178(5):R155–81.
Allard C, et al. Activation of hepatic estrogen receptor-alpha increases energy expenditure by stimulating the production of fibroblast growth factor 21 in female mice. Mol Metab. 2019;22:62–70.
Hannibal KE, Bishop MD. Chronic stress, cortisol dysfunction, and pain: a psychoneuroendocrine rationale for stress management in pain rehabilitation. Phys Ther. 2014;94(12):1816–25.
Schooling CM, Leung GM. Testosterone and cardiovascular risk. Lancet Diabetes Endocrinol. 2015;3(9):682.
Mariotti A. The effects of chronic stress on health: new insights into the molecular mechanisms of brain-body communication. Future Sci OA. 2015;1(3):FSO23.
Junnila RK, et al. Disruption of the GH receptor gene in adult mice increases maximal lifespan in females. Endocrinology. 2016;157(12):4502–13.
List EO, et al. Removal of growth hormone receptor (GHR) in muscle of male mice replicates some of the health benefits seen in global GHR-/- mice. Aging (Albany NY). 2015;7(7):500–12.
List EO, et al. Liver-specific GH receptor gene-disrupted (LiGHRKO) mice have decreased endocrine IGF-I, increased local IGF-I, and altered body size, body composition, and adipokine profiles. Endocrinology. 2014;155(5):1793–805.
Nagarajan A, Srivastava H, Jablonsky J, Sun LY. Tissue-specific GHR knockout mice: an updated review. Front Endocrinol (Lausanne). 2020;11: 579909.
Young JA, et al. Characterization of an intestine-specific GH receptor knockout (IntGHRKO) mouse. Growth Horm IGF Res. 2019;46–47:5–15.
Acknowledgements
We would like to thank Dr. John J. Kopchick for providing animals to start our colonies and Lisa Hensley for editorial assistance.
Funding
This work was supported by the National Institutes of Health NIA R01-AG057767 (YF, SM, KNH, ERH) and NIA R01-AG061937 (YF, SM, ERH, KNH, AB), Dale and Deborah Smith Center for Alzheimer’s Research and Treatment, Kenneth Stark Endowment (YF, SM, KNH, ERH), NIA R21-AG062985, American Diabetes Association 1–19-IBS-126 (DM, RS, AB), AG031736 and AG051869 (AB).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Fang, Y., Medina, D., Stockwell, R. et al. Resistance to mild cold stress is greater in both wild-type and long-lived GHR-KO female mice. GeroScience 45, 1081–1093 (2023). https://doi.org/10.1007/s11357-022-00706-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-022-00706-0