Abstract
This review elucidated the recent advances in integrating microalgal communities in monitoring metal pollution in aquatic ecosystems of sub-Saharan Africa (SSA). It also highlighted the potential of incorporating microalgae as bioindicators in emerging technologies, identified research gaps, and suggested directions for further research in biomonitoring of metal pollution. Reputable online scholarly databases were used to identify research articles published between January 2000 and June 2023 for synthesis. Results indicated that microalgae were integrated either individually or combined with other bioindicators, mainly macroinvertebrates, macrophytes, and fish, alongside physicochemical monitoring. There was a significantly low level of integration (< 1%) of microalgae for biomonitoring aquatic metal pollution in SSA compared to other geographical regions. Microalgal communities were employed to assess compliance (76%), in diagnosis (38%), and as early-warning systems (38%) of aquatic ecological health status. About 14% of biomonitoring studies integrated microalgal eDNA, while other technologies, such as remote sensing, artificial intelligence, and biosensors, are yet to be significantly incorporated. Nevertheless, there is potential for the aforementioned emerging technologies for monitoring aquatic metal pollution in SSA. Future monitoring in the region should also consider the standardisation and synchronisation of integrative biomonitoring and embrace the “Citizen Science” concept at national and regional scales.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stream ecosystems play an ecologically significant role in supporting aquatic biodiversity and providing beneficial ecosystem services that sustain the environment and promote human well-being (Limburg 2009; Maes et al. 2020). Stream ecosystem services include fresh water and food provisioning, sediment retention and transport, pollution control, recreation and ecotourism, flood regulation, disease prevention, nutrient cycling, and cultural heritage preservation (MEA 2005). In addition, both stream and riparian systems serve as biodiversity banks for aquatic and semi-aquatic biota as well as developmental stages of several terrestrial fauna such as arthropods, amphibians, and reptiles, among others (Mccabe 2010; Steward et al. 2022). Streams are also critical in transferring nutrients, matter, and energy, thus acting as sources and sinks of pollutants and disease vectors (Limburg et al. 2013; Wohl 2018; Bashir et al. 2020).
However, the negative impact of both natural and anthropogenic pressures has reduced the ability of stream ecosystems to supply aquatic ecosystem services (Khatri and Tyagi 2015). Natural factors such as climate change, droughts, floods, and other natural disasters impact aquatic ecosystems by altering water availability, water quality, and aquatic biodiversity (Costanza et al. 2014; Talbot et al. 2018; Culhane et al. 2019). Anthropogenic factors such as waste disposal, urbanisation, demand for agricultural land and expansion of industrial activities such as metal mining and fossil fuel combustion, and habitat destruction are also significant drivers of deterioration in aquatic ecosystems (Borgwardt et al. 2019; Cormier et al. 2019; Kimirei et al. 2021; Ferreira et al. 2023). Metal pollution of aquatic ecosystems from natural and anthropogenic sources is particularly an environmental and health concern in metal mining regions due to metals being persistent, non-biodegradable, and toxic (Yahya et al. 2018; Ali and Khan 2019; Amoatey and Baawain 2019; Zhou et al. 2020). In the stream ecosystems of Sub-Saharan Africa (SSA), aquatic metal pollution is an ever-growing environmental concern (Biney et al. 1994; Fayiga et al. 2018). There has been a steady accumulation of metals in water, sediment, and aquatic biota in rivers and lakes of SSA, mainly from natural and anthropogenic sources (Zhou et al. 2020; Yabe et al. 2010; Fayiga et al. 2018; Ochieng et al. 2009). In southern Africa, aquatic metal pollution above permissible limits has been reported for stream water and sediments from mining, coal use, and other industrial activities (Ouma et al. 2022; Addo-Bediako et al. 2021). Gerber et al. (2015) and Moyo et al. (2015) noted high Cu, Co, Pb, and Mn in the Olifants River associated with anthropogenic activities and posed a high risk to aquatic biota. Furthermore, Chetty and Pillay (2019) linked the influence of anthropogenic activities to elevated Cr, Cu, Pb, and Zn in Palmiet and Sezela rivers in South Africa’s Kwa-Zulu Natal coupled with high mobility and bioavailability. In the Zambian Copperbelt, Cu, Co, Pb, and Zn mining has impacted the water and sediments of Lake Kariba (Chalumba et al. 2021) and Kafue River with increased ecological risks to aquatic life. In the Katangese Copperbelt of the Democratic Republic of Congo, extreme sediment enrichment with Cu (190.2 mg/kg) and Zn (1117 mg/kg) in the Bumbu River draining Kinsasha has been reported (Kayembe et al. 2018). Banze wa Mutombo (2022) also associated the high pollution of the Mura and Kimpulande tributaries of the Congo River with Cu-Co-As-Cd-loaded mining effluents that increased the vulnerability of aquatic communities.
Metal pollution in West Africa’s aquatic systems has similarly reached alarming levels. Gbogbo and Otoo (2015) reported the detrimental impacts of Cd, As, Hg, and Cu pollution on water, macrophytes, algae, and fish in Ghana’s Sakumo II wetland in the Tema Metropolitan area. According to Ngueyep et al. (2021), Cameroon’s Kadey River tributaries had excess Ni, Fe, Cr, Se, As, and Hg in sediments from artisanal and small-scale gold mining. Tyovenda et al. (2019) reported contamination of water and algae and sediment enrichment with Pb, Hg, Ni, and Fe in River Benue, Nigeria. Despite the relatively low mineral deposit in Eastern Africa, alarming aquatic metal pollution has been reported in Kenya’s gold mining belt (Ngure et al. 2014), Tanzania’s Mara River (Nkinda et al. 2020), Awetu watershed in Ethiopia (Astatkie et al. 2021), and Namukombe stream in Uganda (Omara et al. 2019). For instance, Ngure et al. (2014) noted high Hg (355 mg/kg) in fish, while Astatkie et al. (2021) recorded stream sediment contamination with Pb (2,000 mg/kg), As (623 mg/kg), and Cr (375 mg/kg). Based on the representative studies above, there is sufficient evidence of aquatic metal pollution thus, raising the need for monitoring of aquatic ecosystems across SSA.
One of the approaches that can be employed to effectively monitor and assess the magnitude of anthropogenic and natural impacts on stream ecosystems is the bioindicator concept, which utilises sentinel aquatic biota (Lazorchak et al. 2003; Schwacke et al. 2013; Parmar et al. 2016). Bioindicators reflect the bioavailable fractions of pollutants and hence are of potential ecotoxicological significance (Hamza-Chaffai 2014; Lamare 2019; Kumari and Paul 2020) Based on the targeted outcome, three categories of bioindicators commonly used for monitoring environmental health include early warning, compliance, and diagnostic indicators (Hamza-Chaffai 2014). “Early-warning bioindicators” signify the impending deterioration of ecosystem health. Deviations from the acceptable aquatic environmental conditions are detected by “compliance indicators” while the “diagnostic bioindicators” reflect the causes for the deviations from the expected ecological conditions of the aquatic ecosystem (Sumudumali and Jayawardana 2021). Figure 1 illustrates the bioindicator concept of aquatic metal pollution biomonitoring that utilises the compliance, diagnostic, or early-warning aspects of indicator species or communities.
Aquatic microalgal communities have been utilised to monitor the ecosystem health in lotic environments (Yilmaz et al. 2021; Feisal et al. 2023). The vital ecological role of microalgal communities has been recognised through continuous surveillance to establish their status in the aquatic environment (Li et al. 2021; Thompson and Carstensen 2023). The ability of microalgae to accumulate high levels of pollutants, their relative sessile nature, ease of sampling, ease of culture in the lab, and their trophic importance as primary producers in the aquatic food web have positioned them as suitable bioindicators of metal pollution (Zhou et al. 2008; Parmar et al. 2016; Kumari and Paul 2020). Freshwater microalgae occur either as planktonic, which dominate the pelagic zone, or the benthic forms associated with substrates such as sediments, rocks, macrophytes, mud, and organic debris (Bellinger and Sigee 2015).
Globally, the use of microalgae for biomonitoring as early-warning signals has been widely documented. For instance, in Europe, Dokulil et al. (2016) documented long-term historical biomonitoring using microalgae responses and community composition in the extensive Danube River hydro system. Furthermore, biomonitoring of metal pollution in the transboundary Danube River delta aquatic complex reported high concentrations of bioavailable Ni, Cd, and other potentially toxic elements (Burada et al. 2015; Simionov et al. 2021; Calmuc et al. 2021). Metal pollution trends were also observed in the southeastern Brazil river basin impacted by metal contamination from the Mariana dam failure, with increased Hg bio uptake by microalgae (Marques et al. 2022). Silva et al. (2022) further reiterated the significance of using morphological and taxonomic responses of microalgae as bioindicators to environmental changes in river basins of southern Brazil. In India, microalgal communities in the tropical freshwater Godavari River, Cu, and Zn exhibited lethal effects at high concentrations for the dominant cyanobacteria and chlorophytes (Chakraborty et al. 2010). Feng et al. (2021) also noted the detrimental impacts of metal pollution on the microalgal community structure, with certain microalgal species being more sensitive to the bioavailable metals in the Yangtze River in China. In the Sefid Rud River, Iran, changes in microalgal assemblages were suitable bioindicators of environmental variability and corresponded to physical and chemical changes in the south Caspian Sea catchment (Ramezanpour et al. 2014).
Microalgal communities have also been used to monitor metal pollution in Africa’s stream ecosystems. In West Africa’s Niger River system, Ezewudo et al. (2021) noted weak to high potential ecological risks to aquatic communities, including microalgae, from As, Cd and Hg contamination. In the Cameroon Centre Region, the spatial-seasonal changes in algal densities in the streams of the Sanaga lotic system network draining urban and industrial settlements corresponded to changes in the aquatic physicochemical environment (Pascale 2023). According to Mangadze et al. (2019a), several ecological health studies on southern Africa’s rivers have applied benthic diatoms for biomonitoring. Dalu et al. (2014) noted a direct response of microalgal communities to changes in the physicochemical environment of the Kowie system riverine-estuarine continuum in South Africa’s Eastern Cape. Recent studies on South Africa’s urban Molopo River depicted anthropogenic Cu, Cr, Zn, and Pb sediment contamination with potentially deleterious ecological impacts on the benthic algal and macrofauna communities (Mohajane and Manjoro 2022). Additionally, the diatom-based biomonitoring tools (e.g., the “South African Diatom Index (SADI)” and the “Benthic Diatom Index (BDI)”) have been used to detect and quantify the magnitude of natural and anthropogenic impacts on stream ecosystems (Lang et al. 2013; Harding and Taylor 2014; Sirunda et al. 2021).
Microalgae have the potential to be integrated into conventional monitoring programs as complementary tools to increase the resolution in detecting sub-lethal contamination and thus serve as early-warning bio-systems (Cid et al. 2012; Bae and Park 2014). Despite the potential of integrating microalgal communities in the biomonitoring of aquatic ecosystems, this approach remains one of the least explored alternatives to sustainable management of freshwater ecosystems in SSA (Lemley et al. 2016). Therefore, this review seeks to (1) provide insights into recent advances in the integration of microalgae in biomonitoring metal pollution in the SSA lotic systems, (2) highlight the potential of integrating microalgal as bioindicators in the emerging technologies for monitoring aquatic metal pollution of lotic systems, and (3) identify research gaps and suggest directions for further research in the integrating microalgae as bioindicators of metal pollution in lotic systems of SSA.
Methodological approach
Scope of literature search
To ensure that high-quality and relevant articles were selected, our review defined explicit inclusion criteria outlined in Cornelissen et al. (2009). The literature search included articles addressing advances in integrating microalgae for biomonitoring metal pollution in stream ecosystems draining metal-mining landscapes of SSA. The search was restricted to original research, written in English, from articles published between January 2000 and June 2023 to identify ‘gold-standard’ literature on stream biomonitoring of metal pollution with a focus on microalgae as bioindicators.
The article selection process aims to identify the original research papers that present clear evidence of the study objectives (Syeed et al. 2023). Page et al. (2021) preferred reporting items for systematic review and meta-analysis (PRISMA) protocol was followed to ensure a comprehensive and well-defined strategy for the identification, screening, and inclusion of articles for review (Fig. 2). Reputable academic databases, SCOPUS, Taylor and Francis, and Semantic Scholar were searched for authentic articles (Kitchenham and Charters 2007). Furthermore, snowballing or citation-searching from “gold-standard” literature was used to identify more articles for preliminary screening (Wright et al. 2014).
The literature search was conducted based on the article title, abstract, and keywords using key terms such as “bioindicator”, “aquatic biomonitoring”, “metal mining”, “aquatic pollution”, “algae”, “aquatic ecosystems”, “sub-Saharan Africa”, “Africa”, “e-DNA”, “environmental DNA”, “metagenomics”, “microalgae”, “Biosensors”, “Remote sensing”, and “Citizen science”. From each article included, the following information was extracted: (i) title, (ii) authors, (iii) publication year, (iv) regional distribution (v) main objective, (vi) methods (physical, chemical, biological) (vii) indicator organism(s), (viii) environment (sediment, water, biota), (ix) pollutant(s) (x) microalgal metrics used, and (xi) key findings, gaps, or recommendations.
Streamlining article evaluation and selection
Quality assurance and bias reduction
To remove bias in the first stage of the search, the authors searched independently in the digital databases using search terms with slightly varying synonyms to maximise the extraction of articles from the global search. This initial search was followed by the within-results search, where the authors used the same filter criteria specifying the period, the document type, the region of study, and the field of study. In the second stage, the authors verified the extracted articles’ metadata for completeness and originality. Articles that fulfilled the quality assurance process were included for further synthesis.
Article processing
The results from the databases were downloaded and imported into the Mendeley reference software (Mendeley Ltd). The following metadata was checked and updated where necessary for each article: author(s), title, year of publication (and month), volume, page numbers or article number, abstract, keywords, and DOI, if available. However, articles for which pertinent metadata items such as author, title, or publication year that were missing were further excluded from the list.
Exclusion process
An automated keyword-based search was used to explore the database and extract relevant research articles (Beecham et al. 2008). The terms were searched in the article titles, abstracts, and keywords. The exclusion criteria for out-of-scope articles were principally based on the following aspects: (1) studies outside freshwater systems, e.g., oceans and seas; (2) other bioindicator categories used, e.g., non-photosynthetic bacteria, marine plankton, freshwater zooplankton, macrophytes, macroalgae, macroinvertebrates, and vertebrates; (3) clinical and laboratory biomonitoring studies, e.g. humans and wild and domestic animals using water resources; and (4) studies involving aquatic ecosystem pollutants other than metals. Furthermore, manual removal was conducted to ensure that only relevant and complete articles were included in the final review process (Petticrew and Roberts 2008).
Bibliometric analysis
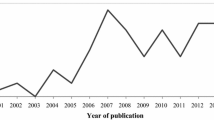
A bibliometric analysis of the extracted information was conducted to classify articles based on the year of publication, authors, region, main objective(s), bioindicator type, environmental matrix, pollutant(s), methods, and the microalgal metric(s). Following the PRISMA filtering protocol, the review included 21 articles (15 from the digital scholarly databases and six from snowballing/citation search) relevant to the research area, geographical location, and study period. From Fig. 3a, between January 2000 and June 2023, there was a notable general cumulative 95% increase in the studies incorporating different microalgal taxa in monitoring metal pollution in aquatic ecosystems in the SSA. This indicates a growing interest in incorporating microalgal taxa in aquatic biomonitoring.
Generally, between January 2000 and June 2023, studies indicate that only South Africa, Namibia, and the Democratic Republic of Congo (DRC) integrated microalgae and microalgal-based eDNA, respectively, in aquatic biomonitoring for metal pollution. Sub-regionally, only 24% of the countries in West Africa, 10% in Eastern Africa, 25% in Central Africa, and 75% in Southern Africa conducted microalgal-based biomonitoring of metal pollution in streams. However, microalgal-eDNA integration in aquatic metal pollution biomonitoring is still in its infancy in SSA, with only Central and Southern Africa accounting for 25% and 75% of aquatic-based research to monitor metal pollution (Fig. 3b). The integration of microalgal-eDNA method in aquatic metal pollution biomonitoring in SSA was first documented by Jordaan et al. (2019), who noted a 6% variability in bacterial community composition and diversity from the anthropogenic Co, As, Cr, Ni, and U pollution in the rivers within the lower Wonderfonteinspruit catchment of South Africa. Since then, the eDNA approach has been seen as a potential approach to accelerate aquatic biomonitoring by supplementing traditional taxomorphological monitoring in the SSA landscape (Perry et al. 2022).
Bioindicator taxa and environmental assessment
The diatoms are the single most preferred microalgal bioindicator taxon (36%) and are also used with benthic macroinvertebrates (18%) to monitor aquatic metal pollution. Considering their specificity and sensitivity to ecological changes in aquatic ecosystems, diatoms have been widely employed to detect perturbations in stream water quality (Lobo et al. 2016; Mangadze et al. 2017). The preference for both taxa could be attributed to their stationary and benthic nature, which makes them suitable for recording long-term pollutant trends compared to the instantaneous physicochemical methods that only consider a “snap-shot” of the environmental water quality (Beyene et al. 2009; Hattikudur et al. 2014). Other single-use taxa of microalgae, including cyanobacteria, had equal preferences (18%), while “algae” and macroinvertebrates comprised 9% each as the bioindicators used to assess metal pollution (Fig. 4a).
Regional studies elsewhere, for example, in North America (e.g., Smucker et al. 2018) and Asia (e.g., Chon et al. 2013), have similarly integrated algae and macroinvertebrates as well as microbial communities to monitor metal pollution of freshwaters while leveraging on the producer–consumer trophic changes as indicators of disturbances at catchment scale. Furthermore, Respondek et al. (2022) integrated mosses and microalgae in monitoring metal pollution in surface water in the smelter area of Ozimek, Poland. They observed diatom taxa as the dominant algal group that indicated responses to metal stress, e.g., the metal-tolerant Achnanthidium sp and Mayamaea sp dominated up to 99% of the algal communities, and served as excellent bioindicators of metal contamination. In addition, Pandey (2020) compared green algae, cyanobacteria, and diatom species and noted an increased relative taxa abundance, indicating increased tolerance to metal pollution. Moreover, increased lipid production and cell-wall teratologies in diatoms, also indicated by Lavoie et al. (2012), were observed under Cu, Cd, Zn, and Pb stress. Pandey and Bergey (2018) also found that diatoms-dominated periphyton biofilms were excellent indicators of metal pollution, thus showing the utility of periphytic diatom communities as an effective tool for biomonitoring of aquatic metal pollution. Gbogbo and Otoo (2015) used the biomonitoring potential of algae, among other bioindicator biota of an urban wetland system in Ghana, to determine the magnitude of metal pollution algae accumulated up to 12 mg/g Cd. Similarly, Leguay et al. (2016) and Solak et al. (2020) reiterated the importance of complementing physicochemical assessment techniques with diatom-dominated biofilm-based proxies, diatom indices (e.g., the Pampean Diatom Index and Specific Pollution Index) to monitor metal contamination in aquatic systems.
In Fig. 4b, water is the most frequently assessed abiotic matrix (28.6%) and in combination with different bioindicators in the same proportion for assessing metal contamination. Studies by Dalu et al. (2017, 2022a) and Tyovenda et al. (2019) included stream sediment plus water, diatoms, algae, and benthic macroinvertebrates in the evaluation of metal pollution to obtain a three-way health status of the aquatic ecosystem. In addition, Mangadze et al. (2017) incorporated a fourth dimension of atmospheric contribution to stream ecosystem metal pollution to assess the potential of using diatoms as suitable bioindicators of ionic metal pollution along a South African temperate river system.
Application of microalgae for biomonitoring tropical stream ecosystems of SSA
In Fig. 5 and Table 1, three studies (by Jordaan et al. 2019 Laffite et al. 2020 Perry et al. 2022) incorporated the microalgal-eDNA to check for environmental compliance with the established national or international guidelines for metal contaminant levels in freshwater aquatic environments. Jordaan et al. (2019) and Pereira‐da‐Conceicoa et al. (2021) used eDNA as a diagnostic tool to determine the causes of deteriorating water quality and changes in microbial communities in South African river catchments. However, no study used microalgal-eDNA for early warning of aquatic ecosystem change, making this a potential area for future research. Most studies (76%) employed various microalgal taxa responses for compliance monitoring, followed by diagnostic and early-warning functions, each at 38% (Fig. 5).
Several studies combined more than one environmental application of algal communities to test for compliance, diagnosis, or early warning to evaluate the overall integrity and potential ecological risks for the respective aquatic ecosystems investigated. Despite its environmental importance, only 30% of the studies used biomonitoring as an early-warning tool, while 40% employed biomonitoring for diagnostic purposes. Nevertheless, 91% of the studies were targeted to determine environmental compliance of anthropogenic activities that introduce metal contaminants to the aquatic environment against the set effluent discharge standards in the pro-active management of aquatic ecosystems in SSA. Table 1 highlights the environmental applications (compliance, diagnostic, and early warning) of microalgal communities based on the main objectives of the reviewed studies. Algal communities are helpful for compliance, diagnostic, or early-warning biomonitoring since they reflect long-term changes in stream water quality (Mangadze et al. 2016) (Table 1). Ugbeyide and Ugwumba (2021) assessed the physicochemical and biological status of the Ibuya River in Nigeria, which was impacted by anthropogenic pollution. Cd (0.003 mg/L) and Pb (3.5 mg/L) levels exceeded permissible limits for surface water quality, while the lower species richness and composition, dominated by Bacillariophyceae, reflected a lotic system impacted by allochthonous pollution. Oberholster et al. (2016) observed increased algal species diversity caused by improved downstream water quality during the rehabilitation of the Grootspruit wetland, South Africa, impacted by acid-mine drainage. The trends concur with a previous study by Ali and Abd el-Salam (1999) that noted changes in the dominance of microalgal species Cyclotella and Nitzschia (Bacillariophyta), Actinastrum and Scenedesmus (Chlorophyta), and Oscillatoria sp (Cyanophyta).
Furthermore, in the Macedonian Maidanska River, the bioconcentration and biomineralisation of Cu, As, Cr, Se, and Cs were observed in Audouinella sp, while the high bioaccumulation of Ba (3 mg/g) and intracellular biomineralisation were evidenced in Spirogyra sp. thereby positioning these algal species as a biological pathfinder for acid-mine drainage deposits (Bermanec et al. 2018). Water and sediment chemistry, including nutrient and metal pollutants, largely influence the stream algal community composition. Dalu et al. (2017) explored the influence of anthropogenic impacts on diatom communities and noted the dominance of pollution-tolerant taxa in an austral temperate stream in South Africa. The tolerance and morphological changes (teratologies) on epilithic diatom communities have also been employed to monitor and quantify the biological effects of metal stress from an abandoned Coval da Mo mine drainage (Ferreira da Silva et al. 2009). The findings agree with Pandey and Bergey (2018), who correlated non-taxonomical parameters, including teratologies and lipid bodies, to indicate metal toxicity and recovery in fluvial systems. Diatom indices, including the GDI (Generic Diatom Index), BDI (Biological Diatom Index), and TDI (Trophic Diatom Index), were successfully employed to monitor the Dongjiang River in China with BDI and GDI showing an apparent response to water quality changes (Deng et al. 2012). Diatoms have also been incorporated in multispecies biomonitoring of the temporal variability of metal pollution in Nigeria’s Calabar River (Hena et al. 2022) and Kebena-Akaki Rivers, Ethiopia (Beyene et al. 2009). In both studies, a significant response was observed between the algal community structure and metal concentrations. Mangadze et al. (2017) similarly reflected the role of diatom assemblages as bioindicators of metal pollutants (e.g., As, Zn, Cu, and Cr), particularly on low pollution tolerant species such as Fragilaria, Cyclostephanos, and Gyrosigma transition to high pollution tolerant forms (e.g., Nitzschia and Gomphonema). This observation is also supported by findings in Dalu et al. (2022b), where changes strongly influenced the structure of diatom communities in water and sediment quality due to the presence of metal contaminants such as B, Cu, and Fe in the Krom River system of the western cape, South Africa. Microalgal communities have also been used to indicate metal pollution in lacustrine systems. For instance, Ogoyi et al (2011) determined metal concentrations (Zn, Pb, Cd, Cr, and Hg) in algal communities alongside water and sediment as an integrative aquatic ecosystem assessment approach.
Integrating microalgal communities into molecular tools for compliance, diagnostic, or early-warning monitoring of streams in mining regions of SSA is also ongoing. Jordaan et al. (2019) studied the influence of anthropogenic pollution on the structure and function of aquatic bacterial communities, using 16S rRNA as a proxy indicator, in South Africa’s Wonderfonteinspruit river catchment. Pereira-da-Conceicoa et al. (2021) demonstrated the merits of incorporating eDNA into existing aquatic biomonitoring metrics with the potential of recovering more diversity and a higher resolution. The ecological advantages of integrating eDNA studies in aquatic biomonitoring above are also evident in other global investigations. Li et al. (2018) noted that the operational taxonomic units of molecular e-DNA data can predict up to 79% of aquatic pollution. Ancion et al. (2010) used 16S rRNA gene libraries to examine the impact of Cu, Zn, and Pb on bacterial communities embedded in freshwater biofilms and recorded higher sensitivities, thereby confirming their potential role as compliance indicators of stream health.
Method integration and environmental and biological metrics used for assessment of metal pollution in aquatic ecosystems of SSA
All the reviewed microalgal-based works combined physicochemical and biological techniques to investigate metal pollution, possibly to enhance the detection of contaminants and their impact on biota (Torrisi et al. 2010). Several environmental and biological indices were used to quantify the magnitude of the impact of metal pollution, including enrichment and contamination factors, pollution indices, species richness, and diversity indices (Bere et al. 2016; Dalu et al. 2022a; Mangadze et al. 2019b; Ugbeyide and Ugwumba 2021) (Table 2). According to Lobo et al. (2016), biotic indices such as Beck’s index and Renberg’s “Index B” developed from the relative abundances of bioindicator species have been employed for biomonitoring of streams and other aquatic ecosystems. The determination of the physicochemical water quality coupled with the estimation of aquatic biodiversity based on biotic indices has been used to infer the ecological health status and as “early-warning” indicators of aquatic ecosystem health changes (Bellinger and Sigee 2015; Forio and Goethals 2020). Geochemical indices such as contamination factor (CF), enrichment factor (EF), geo-accumulation index (Igeo), and pollution load index (PLI) were used to evaluate the occurrence and magnitude of pollution in SSA streams receiving metal(loid) contaminants (Tyovenda et al. 2019; Hena et al. 2022). Changes in algal community composition, abundance, PLI, and metal pollution index (MPI) have also been used to assess aquatic metal pollution stress in aquatic communities of Egypt’s Alexandria coast (Ismail and El Zokm 2023).
Integrating molecular techniques in biomonitoring is a potential approach to revolutionise aquatic pollution assessment (Li et al. 2010; Lobo et al. 2016). In this review, Laffite et al. (2020) observed a significant correlation between metals and 16 s rRNA, suggesting a close link between metal pollution and human-mediated pressures on an urban river in the Democratic Republic of Congo. Pereira-da-Conceicoa et al. (2021) demonstrated the relevance of integrating environmental DNA (eDNA) into existing monitoring metrics to provide additional taxonomic resolution for aquatic biodiversity management in South African streams. The application of molecular methods has also been observed to substantially improve the biomonitoring of streams in France, China, and Switzerland compared to the traditional morphotaxonomic methods (Apothéloz-Perret-Gentil et al. 2021; Keck et al. 2018; Li et al. 2018). However, Perry et al. (2022) noted a significant drawback in the integration of eDNA principally inhibited by inadequate reference data for SSA in the gene banks. The lack of reference eDNA databases, downstream transport, dilution of DNA fragments, and introduction of terrestrial DNA, among other challenges, has also been observed in other regions, e.g., Finland (Norros et al. 2022), Switzerland (Deiner et al. 2016), Canada (Laporte et al. 2022), and globally (Beng and Corlett 2020).
Integrating microalgae into emerging technologies for monitoring metal pollution in stream ecosystems
Several cutting-edge emerging technologies are gaining popularity as complementary approaches to support conventional monitoring and assessments of stream ecosystems, as described below.
Microalgal-eDNA metabarcoding
Based on the current review, recently, limited studies have incorporated microalgal eDNA in biomonitoring aquatic metal pollution in the SSA. For instance, Laffite et al. (2020) investigated the co-contamination and seasonal variability of metal in bed sediments of urban rivers in DRC using bacterial eDNA. Significant correlations were observed between metal concentrations and 165 s rRNA bacterial densities, linking pollution to anthropogenic inputs. In South Africa, Jordaan et al. (2019), using the 16 s rRNA gene profiles, noted a substantial impact of pH and metal contamination from mining on bacterial diversity and community structure in the lower Wonderfonteinspruit catchment rivers. Furthermore, Perry et al. (2022) demonstrated the cost–benefit of using bulk samples and eDNA for multispecies biodiversity monitoring of Namibia’s freshwater systems. However, in most SSA countries, few studies, if any, have integrated microalgal eDNA in aquatic metal pollution biomonitoring. Given the sparsity of eDNA biomonitoring research data in SSA, more effort is needed to develop methods adapted to regional and local conditions and to generate eDNA gene-bank reference data to increase our understanding of SSA aquatic ecosystems (Perry et al. 2022). In addition, the performance of eDNA tools in biomonitoring aquatic metal pollution in SSA lotic ecosystems compared with the conventional monitoring approaches is not adequately investigated. Therefore, further research is needed to address this methodological gap by integrating microalgae-based eDNA biomonitoring of aquatic metal(loid) pollution at the community, species, and molecular level in stream ecosystems of SSA (Stat et al. 2017).
In the recent past, most of the eDNA biomonitoring has been conducted in the global North (Resh 2007). For instance, Cilleros et al. (2019) compared the effectiveness of eDNA metabarcoding and conventional morphotaxonomic techniques while assessing the diversity of fish assemblages in 38 streams of the French Guiana. Their findings revealed that while traditional taxonomic methods offered a more comprehensive inventory of fish taxa, they were spatially limited. In contrast, eDNA metabarcoding, when complemented with classical methods, was a more comprehensive and efficient approach for rapidly assessing and monitoring fish diversity on a larger spatial scale. Similarly, Gleason et al. (2021) conducted a study in southern Ontario, Canada, comparing eDNA metabarcoding techniques with traditional kick-net sampling to monitor lotic macroinvertebrate communities. Their findings demonstrated that eDNA techniques, especially metabarcoding of bulk tissues, provided a better representation of the diversity of macroinvertebrate taxa at a finer spatial resolution than traditional methods. However, in SSA, few studies have integrated eDNA in aquatic biomonitoring of metal pollution (e.g., Laffite et al. 2020; Jordaan et al. 2019; Perry et al. 2022). Therefore, progressive regional research must be strengthened to overcome the current limitations of aquatic eDNA biomonitoring, such as inadequate e-DNA reference data (Perry et al. 2022).
While acknowledging that eDNA is more appropriate for short-term monitoring, eDNA data can be used integratively with long-term monitoring approaches, such as remote-sensing, biosensor, and citizen science (Hansen et al. 2020). For instance, eDNA data can be used to validate or ground-truth remotely sensed data to ensure the reliability of long-term monitoring systems. Additionally, the integration of eDNA can increase the resolution of pollutant detection at sub-lethal and ensure the validity and consistency of sensed data.
Biosensor systems for aquatic biomonitoring
Recently, a variety of biosensors gained high attention and have been employed in in-situ for real-time monitoring and detection of environmental contaminants (Huang et al. 2023). A biosensor typically comprises a biosensing probe and a transducer that detects a contaminant by producing a quantifiable signal (Mishra et al. 2019; Rovira and Domingo 2019). The biosensor probe material can be antibody-, DNA-, whole-cell-, or enzyme-based (Singh et al. 2020). The transducer translates the biological signals to optical or electrical signals via optical, physicochemical, or piezoelectric material (Nguyen et al. 2019). Electrochemical and optical biosensors have been employed to detect and quantify metals, including Hg+, Pb2+, Zn2+, Cu2+, and Cd2+ in water (Wu et al. 2023). Advances in nanotechnology have further improved the performance of biosensors due to the numerous benefits of larger sensing equipment. Nanosensor materials improve biosensor efficiency for colour sensing, target sensitivity, and carrier capacity. Additionally, nanomaterials have high thermal and electrical conductivity (Huang et al. 2021; Abdel-Karim 2024).
Whole-cell microbial biosensors detect metal ions based on the genetic element that responds to target metals (Huang et al. 2023). In aquatic environments, whole-cell bacterial biosensors have been used to detect bioavailable metals with high sensitivity (Cerminati et al. 2015). Alfadaly et al. (2021) applied a complementary target resistive Rhizobium bacteria-based and Rhodotorula fungi-based bioelectrochemical sensor to detect and remove Cr6+ and Cd2+ ions from polluted water. The bacterial component exhibited superior performance for metal resistivity and removal. In another study, Cerminati et al. (2015) confirmed the efficacy of a broad-spectrum whole-cell-based metal biosensor as a screening tool for the presence of bioavailable Au, Hg, Pb, and Cd in water.
Genetically engineered DNA-based microbial biosensors combined with electrochemical transducers broaden the applicability of cell-based biosensors for early monitoring and detection of metal ions in water Jeon et al. (2022). According to Jeon et al. (2022), the mutation of a regulatory protein ZntR in Escherichia coli enhanced the selectivity of Pb2+ ions after metal ion-exporting genes were deleted in the host cells. Furthermore, Nourmohammadi et al. (2020) observed high specificity for Pb2+ bacterial biosensor expressing a luciferase reporter gene controlled by pbr/cadA promoters in Cupriavidus metallidurans in a genetically engineered bacterial system.
According to Huang et al. (2023), biosensors are low-cost, easy to use, and energy-saving and require minimal pre-sample treatment. In addition, biosensor technology uses non-hazardous materials and has considerably low carbon footprints compared to physicochemical methods. Furthermore, the integration of bacteria into biosensor technologies offers numerous benefits in the detection and monitoring of aquatic metal pollution. Biosensors and microalgae serve as complementary tools, offering different perspectives and capabilities. Biosensors enhance the detection and quantification of metal contaminants in real-time or near real-time, thereby allowing for rapid detection and tracking of metal pollution (Wu et al. 2023).
In contrast, microalgae are reliable bioindicators of long-term exposure to metal pollution, reflecting the historical trend. A combination of biosensor data with microalgae assessments reflects a comprehensive understanding of short and long-term metal pollution dynamics in impacted streams. Additionally, biosensors often exhibit high sensitivity and specificity for detecting target metal ions, enabling the detection of sub-lethal metal concentrations (Huang et al. 2023). Microalgae, while sensitive to metal pollution, may not always provide precise measurements of metal concentrations at low levels or in complex environmental matrices. The integration of microalgal DNA into biosensors has the advantage of sensitivity and specificity, especially in natural environments with multi-elemental metal contaminants. Furthermore, biosensors are robust and can simultaneously be deployed at multiple locations within stream ecosystems. This spatial advantage complements the localised application of microalgae per time to monitor metal contamination.
Additionally, biosensors are often portable and easy to deploy, making them accessible for field-based monitoring in remote or challenging environments. However, the high cost and technological requirements of nanomaterials production could impede the production and application of nanobiosensors, particularly in developing countries.
Remote sensing
Satellite-based remote sensing (RS) and hyperspectral imaging is a cost-effective monitoring approach that enhances extensive and rapid spatial coverage of the Earth’s surface with repeatability capabilities for investigating environmental systems (Reddy 2018; Pettorelli et al. 2018). By detecting unique spectral signatures of various substances, including metals, RS can pinpoint the presence and concentration of specific metals in waters via hyperspectral electromagnetic radiation. For instance, Lin et al. (2024) determined the concentration of metals in China’s Dalian Lake using hyperspectral analysis and genetics algorithms. The integration of RS techniques with biosensor data capture probes also improved the spatial mapping of metals and sediments along Egypt’s Red Sea Coast (Mohammed et al. 2024). Bresciani et al. (2016) mapped patterns of cyanobacterial blooms in five Italian lakes using a suite of aerial and space-borne hyperspectral sensors with increased accuracy. Guo et al. (2022) and Cao et al. (2018) integrated RS to model metals and chlorophyll-a concentrations in water. The models provided high retrieval accuracy and realistic information.
Data generated from RS enables the creation of detailed spatiotemporal maps of aquatic metal pollution and hotspots mapping. Integrating RS data with water quality measurements and GIS data provides a comprehensive understanding of metal pollution dynamics in aquatic ecosystems (Yu et al. 2020; Zhu et al. 2022). Furthermore, RS is cost-effective and efficient compared to traditional field-based methods, enabling the rapid collection of extensive data over large areas with lower monitoring costs (Avtar et al. 2020). Overall, RS serves as a valuable tool in monitoring metal pollution in aquatic environments, providing timely and spatially explicit information crucial for informed decision-making and effective environmental management strategies.
The complementarity between RS and microalgae in monitoring metal pollution in aquatic systems is multifaceted. RS provides wide spatial coverage, allowing for the monitoring of large water bodies and the identification of metal pollution hotspots (Chi et al. 2016). However, RS may lack the spatial resolution needed to detect localised pollution events or variations. Microalgae, on the other hand, can be highly sensitive to rapid changes in metal concentrations and hence reflect localised pollution impacts. Furthermore, RS allows for the monitoring of changes in metal pollution over time by capturing images at different intervals. In contrast, the rapid response of microalgae to changes in metal concentrations makes useful indicators of short-term pollution events.
RS data can be validated and calibrated using ground-truthed data, including responses from bioindicators, including microalgae (Cook et al. 2023). This process enables researchers can assess the accuracy of remote-sensing-derived metal pollution estimates and refine remote-sensing algorithms to improve their reliability. Microalgae responses to metal pollution can serve as early warning indicators of environmental degradation. With a combination of RS data with real-time monitoring of microalgae populations, researchers can develop early warning systems to alert authorities to potential pollution events or ecosystem stressors, enabling timely intervention and mitigation efforts.
Citizen science
Citizen science monitoring involves volunteers (i.e., mainly non-professionals), often the riparian communities, and is fundamentally public participation by stakeholders in environmental stewardship (Moharana 2021; Fraisl et al. 2022). Citizen science has the potential of upscaling field studies to a regional or global extent coupled with centralised monitoring efforts that enhance extensive and well-coordinated environmental monitoring, which can produce large datasets rapidly. Miguel-Chinchilla et al. (2019) analysed citizen-sensed catchment data on stream turbidity which contributed nearly 12% value to the study. Babiso et al. (2023) analysed water quality data collected from the Meki River, Ethiopia, by citizen scientists. The study results indicated a good agreement with selected parameters, which implied the accuracy of citizen-collected data. Additionally, Thornhill et al. (2017) used citizen science stream data from the metropolis of China to model and classify predictors of water quality using random forest models with reliable results.
The incorporation of smartphone technology to measure and record environmental data under the citizen science programs has greatly improved the speed, volume, and quality of data. Malthus et al. (2020) examined the impact of citizen science smartphone applications (Apps) on remotely sensed surface reflectance, stream sediment, and algal concentrations in 32 stream sites in eastern Australia. Smartphone Apps provide a friendly interface for citizen scientists to engage with and use sophisticated modern water quality monitoring technology. Smartphones are widely accessible, and the Apps are customised for objective, comprehensive, and accurate data capture (Pattinson et al. 2023).
Citizen science can complement microalgal biomonitoring of aquatic metal pollution in stream ecosystems through increased data collection. Citizen science projects engage a broader range of participants, allowing for more extensive data collection across various locations and times (Njue et al. 2019; Babiso et al. 2023). This can provide a more comprehensive understanding of the spatial and temporal dynamics of metal pollution in stream ecosystems. Furthermore, citizen science involves community engagement and creates social accountability and awareness towards environmental stewardship (Ruppen and Brugger 2022). Involving citizens in scientific monitoring fosters a sense of ownership and stewardship over local environments. This leads to increased awareness of environmental issues such as metal pollution and promotes sustainable behaviours to mitigate them. Citizen science, being a cost-effective metal pollution monitoring technique, can leverage the manpower and resources of volunteers, reducing the costs associated with monitoring efforts (Njue et al. 2019; Ruppen and Brugger 2022). This enables more frequent sampling and monitoring, which is essential for detecting changes in metal pollution levels over time. However, there is a need to identify and address the potential for errors and biases in integrating this approach in the biomonitoring of stream ecosystems (Follett and Strezov 2015).
Challenges and opportunities in integrating microalgae in aquatic biomonitoring of metal pollution
Despite the bottlenecks in the integrative monitoring of aquatic metal pollution in lotic systems of SSA using algal communities as bioindicators, several opportunities also present further room for developing a microalgae-based assessment of stream health status in the region. We highlight the challenges and opportunities for developing higher resolution, site-specific, and species-targeted microalgal-based bioassessment in SSA.
Challenges
From our literature search, while other bioindicator taxa, particularly macroorganisms, are popular options, the use of algal communities to assess aquatic metal pollution, where this has been attempted, has been limited to the morphotaxonomic level. Besides not offering the benefit of higher resolution in detecting sub-lethal metal contamination, the absence of region-specific baseline data in several SSA sub-regions further limits the comprehensiveness of their use as bioindicators. Additionally, accurate identification of microalgal species requires trained morphotaxonomists and special equipment, such as the high-resolution scanning electron microscope, which may be limited in SSA. The uptake and integration of microalgae-based bioindicators into cutting-edge biomonitoring tools such as molecular (eRNA and eDNA), artificial intelligence systems (e.g., biosensors), and geospatial systems are yet to take off significantly. Accurate identification is essential for proper assessment and quantification of the magnitude of metal pollution and potential ecological risks to the provision of stream ecosystem services in SSA.
Microalgal communities and populations exhibit significant seasonal fluctuations, which must be understood to allow the partitioning of metal-pollution-induced impacts. Climate change, seasonality, and natural and anthropogenic factors influence microalgal community composition and species abundance. Seasonal fluctuations, such as variations in rainfall and temperature, significantly influence the hydrology of aquatic ecosystems, affecting the transport and deposition of metals (Maphanga et al. 2024). For instance, intense rainfall during the wet season can remobilise metals from soil and sediment into water bodies, causing elevated metal concentrations (Conrad et al. 2020). Conversely, dry seasons may concentrate metals due to decreased dilution and increased evaporation rates (Edokpayi et al. 2017). Extreme climatic events, such as floods and drought, further exacerbate metal pollution by altering metal transport and sedimentation patterns, which may introduce metals from repositories into water bodies (Xia et al. 2015; Wijngaard et al. 2017). Anthropogenic activities such as mining, industrial effluents, and agricultural runoff are significant sources of metal pollution in sub-Saharan Africa (Laffite et al. 2020). These activities introduce high concentrations of metals such as Pb, Hg, Cu, and Cd into aquatic ecosystems, which are highly toxic to aquatic biota (Hama Aziz et al. 2023; Fatmi et al. 2023). The interaction between seasonal dynamics, extreme climatic events, and anthropogenic activities underscores the complexity of integrating microalgae in monitoring aquatic metal pollution in SSA.
Furthermore, in the aquatic environment, metal species and mixtures interact differently with microalgae community species and vary in toxicity. Understanding these interactions and the ecological impact on microbial bioindicators, including microalgae, in the SSA is poorly understood due to the complexity and, hence, the need for cutting-edge research using advanced methods and sophisticated analytical equivalents. Unfortunately, many regions in SSA suffer from limited monitoring infrastructure, which hampers effective biomonitoring and data availability. Also, SSA, as a low to middle-income subregion, is marked by limited financial capacity to fully support advanced environmental stewardship programs in light of other “critical” financial obligations.
Opportunities
Similar to other tropical regions, SSA has a high diversity of microalgal communities and species richness due to its varied and extensively interconnected aquatic habitats. The biodiversity and richness can be leveraged to select the highly sensitive and most indicative species that respond specifically to individual and mixtures of aquatic metal pollution at sub-lethal concentrations. Since the most suitable microalgal communities and species have high sensitivity to changes in water quality, they can serve as potential early warning indicators of aquatic metal pollution episodes in river systems. Therefore, integrating microalgae in stream health assessment will improve early detection and further inform proactive interventions in managing stream ecosystems in the SSA mining regions.
Collaborative research within the SSA and with international research institutions and partners will address the limitations of access to advanced analytical tools, expertise, and limited funding. In particular, adopting modern research and monitoring tools such as eDNA and molecular biosensors will improve the resolution of detection of aquatic metal pollution. Establishing and expanding baseline data collection and accessible online databases will further accelerate the integration of microalgae for monitoring aquatic metal pollution in aquatic ecosystems across SSA.
Conclusion and future perspectives
Integrating microalgal communities as bioindicators of aquatic metal pollution in rivers of sub-Saharan Africa holds great promise for enhancing water quality monitoring and environmental conservation efforts. However, in the past decade and a half, the inclusion of microalgal taxa for integrative monitoring into aquatic metal pollution monitoring programs in the SSA has been low but gradually improving past 2020. The region is still lagging in the integration of emerging tools, such as environmental DNA, and technological advances, such as artificial intelligence models, remote sensing, and citizen science, that offer potential benefits of high precision, speed, reduced costs, and eco-friendly green technologies in monitoring and assessment of stream ecosystem health across its mining landscapes.
Despite the lack of a standardised, synchronised, and adequately documented microalgal database, inadequate microalgal taxonomic and molecular assessment expertise, limited monitoring and processing infrastructure, and economic constraints, the integrative microalgae-based approach offers significant opportunities for addressing aquatic metal pollution in the SSA lotic ecosystems. The high biodiversity of microalgae in SSA presents a vast pool for selecting suitable site-specific (sensitive and indicative) taxa that respond specifically to individual and mixtures of metal pollutants. Different microalgal taxa have been combined with other bioindicator groups to increase the sensitivity to pollutant detection source tracking and quantification. This uniqueness emphasises the central role of microalgae in aquatic ecosystem biomonitoring initiatives. Moreover, the rapid response to changes in water quality during integrative monitoring positions microalgae as potential early warning indicators for aquatic metal pollution events that impact aquatic ecosystems.
Fundamentally, there is an urgent need to prioritise efforts to institutionalism and strengthen and standardise national and regional baseline data collection on microalgae dynamics in response to metal pollution in aquatic ecosystems across SSA. Such data will serve as a foundation for accuracy and a reference point for improving the assessments of metal pollution and ecological impacts on the region’s aquatic ecosystems.
Collaboration among regional and international research institutions and organisations can lead to the development of integrated monitoring networks. These networks can leverage advanced analytical tools, technologies, and expertise to enhance data collection and analysis. Furthermore, involving riparian communities in data collection and monitoring efforts fosters a sense of collective responsibility and ownership of the stream ecosystem resources. In fact, “citizen science” initiatives are crucial to empowering riparian communities to participate actively in the conservation of stream ecosystems.
Continued research into the interactions between metal pollutants and microalgal species is essential for better understanding the ecological consequences of aquatic metal pollution. Complementing conventional monitoring with innovative techniques, such as artificial intelligence, molecular tools, and remote sensing, must be prioritised to improve the overall efficiency and maximise the productivity of environmental stewardship in metal mining regions.
Data availability
All the data generated are available in this review. Additional data and information can be sourced from the cited references and online databases or sources.
References
Abdel-Karim R (2024) Nanotechnology-enabled biosensors: a review of fundamentals, materials, applications, challenges, and future scope. Biomed Mater Devices. https://doi.org/10.1007/s44174-023-00147-z
Addo-Bediako A, Nukeri S, Kekana M (2021) Heavy metal and metalloid contamination in the sediments of the Spekboom River, South Africa. Appl Water Sci 11:133. https://doi.org/10.1007/s13201-021-01464-8
Alfadaly RA, Elsayed A, Hassan RYA et al (2021) Microbial sensing and removal of heavy metals: bioelectrochemical detection and removal of chromium(VI) and cadmium(II). Molecules 26:2549. https://doi.org/10.3390/molecules26092549
Ali GH, Abd el-Salam NF (1999) Factors controlling bioindicators for industrial pollution detection. Biomed Environ Sci 12:194–200
Ali H, Khan E (2019) Trophic transfer, bioaccumulation, and biomagnification of non-essential hazardous heavy metals and metalloids in food chains/webs—concepts and implications for wildlife and human health. Hum Ecol Risk Assess an Int J 25:1353–1376. https://doi.org/10.1080/10807039.2018.1469398
Amoatey P, Baawain MS (2019) Effects of pollution on freshwater aquatic organisms. Water Environ Res 91:1272–1287. https://doi.org/10.1002/wer.1221
Ancion P-Y, Lear G, Lewis GD (2010) Three common metal contaminants of urban runoff (Zn, Cu & Pb) accumulate in freshwater biofilm and modify embedded bacterial communities. Environ Pollut 158:2738–2745. https://doi.org/10.1016/j.envpol.2010.04.013
Apothéloz-Perret-Gentil L, Bouchez A, Cordier T et al (2021) Monitoring the ecological status of rivers with diatom eDNA metabarcoding: a comparison of taxonomic markers and analytical approaches for the inference of a molecular diatom index. Mol Ecol 30:2959–2968. https://doi.org/10.1111/mec.15646
Astatkie H, Ambelu A, Mengistie E (2021) Contamination of stream sediment with heavy metals in the Awetu watershed of Southwestern Ethiopia. Front Earth Sci 9:1–13. https://doi.org/10.3389/feart.2021.658737
Avtar R, Komolafe AA, Kouser A et al (2020) Assessing sustainable development prospects through remote sensing: a review. Remote Sens Appl Soc Environ 20:100402. https://doi.org/10.1016/j.rsase.2020.100402
Awofolu OR (2005) Determination and seasonal variation of heavy metals in algae and sediments in sewers from industrial areas in Lagos State, Nigeria. Pak J Sci Ind Res 48:28–36
Babiso WZ, Ayano KK, Haile AT et al (2023) Citizen science for water quality monitoring in the Meki River, Ethiopia: quality assurance and comparison with conventional methods. Water 15:238. https://doi.org/10.3390/w15020238
Bae M-J, Park Y-S (2014) Biological early warning system based on the responses of aquatic organisms to disturbances: A review. Sci Total Environ 466–467:635–649. https://doi.org/10.1016/j.scitotenv.2013.07.075
Banze wa Mutombo A, Atibu EK, Mbuya wa Mutombo J et al (2022) Contamination by heavy metals from mining activities: an ecological impact assessment of Mura and Kimpulande Rivers, Democratic Republic of the Congo. Watershed Ecol Environ 4:148–157. https://doi.org/10.1016/j.wsee.2022.10.004
Bashir I, Lone FA, Bhat RA et al (2020) Concerns and threats of contamination on aquatic ecosystems. Bioremediation and biotechnology. Springer International Publishing, Cham, pp 1–26
Beecham S, Baddoo N, Hall T et al (2008) Motivation in software engineering: a systematic literature review. Inf Softw Technol 50:860–878. https://doi.org/10.1016/j.infsof.2007.09.004
Bellinger EG, Sigee DC (2015) Fresh water algae: indentification, enumeration and use as bioincators, 2nd edn. John Wiley & Sons, Chichester
Beng KC, Corlett RT (2020) Applications of environmental DNA (eDNA) in ecology and conservation: opportunities, challenges and prospects. Biodivers Conserv 29:2089–2121. https://doi.org/10.1007/s10531-020-01980-0
Bere T, Mangadze T, Mwedzi T (2016) Variation partitioning of diatom species data matrices: understanding the influence of multiple factors on benthic diatom communities in tropical streams. Sci Total Environ 566–567:1604–1613. https://doi.org/10.1016/j.scitotenv.2016.06.058
Bermanec V, Palinkaš LA, Fiket Ž et al (2018) Interaction of acid mine drainage with biota in the Allchar Carlin-type As-Tl-Sb-Au deposit, Macedonia. J Geochemical Explor 194:104–119. https://doi.org/10.1016/j.gexplo.2018.07.015
Beyene A, Addis T, Kifle D et al (2009) Comparative study of diatoms and macroinvertebrates as indicators of severe water pollution: case study of the Kebena and Akaki rivers in Addis Ababa, Ethiopia. Ecol Indic 9:381–392. https://doi.org/10.1016/j.ecolind.2008.05.001
Biney C, Amuzu ATT, Calamari D et al (1994) Review of heavy metals in the African aquatic environment. Ecotoxicol Environ Saf 28:134–159. https://doi.org/10.1006/eesa.1994.1041
Blanco S, Bécares E (2010) Are biotic indices sensitive to river toxicants? A comparison of metrics based on diatoms and macro-invertebrates. Chemosphere 79:18–25. https://doi.org/10.1016/j.chemosphere.2010.01.059
Borgwardt F, Robinson L, Trauner D et al (2019) Exploring variability in environmental impact risk from human activities across aquatic ecosystems. Sci Total Environ 652:1396–1408. https://doi.org/10.1016/j.scitotenv.2018.10.339
Bresciani M, Giardino C, Lauceri R et al (2016) Earth observation for monitoring and mapping of cyanobacteria blooms. Case studies on five Italian lakes. J Limnol 76. https://doi.org/10.4081/jlimnol.2016.1565
Bunholi IV, Foster NR, Casey JM (2023) Environmental DNA and RNA in aquatic community ecology: toward methodological standardization. Environ DNA 5:1133–1147. https://doi.org/10.1002/edn3.476
Burada A, Ţopa CM, Georgescu LP et al (2015) Heavy metals accumulation in plankton and water of four aquatic complexes from Danube Delta area. Aquac Aquarium Conserv Legis Int J Bioflux Soc 7:301–310
Calmuc VA, Calmuc M, Arseni M et al (2021) Assessment of heavy metal pollution levels in sediments and of ecological risk by quality indices, applying a case study: the lower Danube River, Romania. Water 13:1801. https://doi.org/10.3390/w13131801
Cao Y, Ye Y, Zhao H et al (2018) Remote sensing of water quality based on HJ-1A HSI imagery with modified discrete binary particle swarm optimization-partial least squares (MDBPSO-PLS) in inland waters: a case in Weishan Lake. Ecol Inform 44:21–32. https://doi.org/10.1016/j.ecoinf.2018.01.004
Cerminati S, Soncini FC, Checa SK (2015) A sensitive whole-cell biosensor for the simultaneous detection of a broad-spectrum of toxic heavy metal ions. Chem Commun 51:5917–5920. https://doi.org/10.1039/c5cc00981b
Chakraborty P, Raghunadh Babu PV, Acharyya T, Bandyopadhyay D (2010) Stress and toxicity of biologically important transition metals (Co, Ni, Cu and Zn) on phytoplankton in a tropical freshwater system: an investigation with pigment analysis by HPLC. Chemosphere 80:548–553. https://doi.org/10.1016/j.chemosphere.2010.04.039
Chalumba KS, Eliezer MB, Patricia B et al (2021) Assessment of heavy metals in wild and farmed tilapia (Oreochromis niloticus) on Lake Kariba, Zambia: implications for human and fish health. Food Addit Contam Part A 39:74–91. https://doi.org/10.1080/19440049.2021.1975830
Chetty S, Pillay L (2019) Assessing the influence of human activities on river health: a case for two South African rivers with differing pollutant sources. Environ Monit Assess 191:168. https://doi.org/10.1007/s10661-019-7308-4
Chi G, Ma J, Shi Y, Chen X (2016) Hyperspectral remote sensing of cyanobacterial pigments as indicators of the iron nutritional status of cyanobacteria-dominant algal blooms in eutrophic lakes. Ecol Indic 71:609–617. https://doi.org/10.1016/j.ecolind.2016.06.014
Chmielewská E, Medved’ J (2001) Bioaccumulation of heavy metals by green algae Cladophora gramerata in a refiner sewage lagoon. Croat Chem Acta 74:135–145
Chon T-S, Qu X, Cho W-S et al (2013) Evaluation of stream ecosystem health and species association based on multi-taxa (benthic macroinvertebrates, algae, and microorganisms) patterning with different levels of pollution. Ecol Inform 17:58–72. https://doi.org/10.1016/j.ecoinf.2013.06.004
Cid A, Prado R, Rioboo C et al (2012) Use of microalgae as biological indicators of pollution: looking for new relevant cytotoxicity endpoints. Microalgae: biotechnology, microbiology and energy. Nova Science Publishers, New York, pp 311–323
Cilleros K, Valentini A, Allard L et al (2019) Unlocking biodiversity and conservation studies in high-diversity environments using environmental DNA (eDNA): a test with Guianese freshwater fishes. Mol Ecol Resour 19:27–46. https://doi.org/10.1111/1755-0998.12900
Conrad SR, Santos IR, White SA et al (2020) Elevated dissolved heavy metal discharge following rainfall downstream of intensive horticulture. Appl Geochem 113:104490. https://doi.org/10.1016/j.apgeochem.2019.104490
Cook KV, Beyer JE, Xiao X, Hambright KD (2023) Ground-based remote sensing provides alternative to satellites for monitoring cyanobacteria in small lakes. Water Res 242:120076. https://doi.org/10.1016/j.watres.2023.120076
Cormier R, Elliott M, Rice J (2019) Putting on a bow-tie to sort out who does what and why in the complex arena of marine policy and management. Sci Total Environ 648:293–305. https://doi.org/10.1016/j.scitotenv.2018.08.168
Cornelissen B, Zaidman A, van Deursen A et al (2009) A systematic survey of program comprehension through dynamic analysis. IEEE Trans Softw Eng 35:684–702. https://doi.org/10.1109/TSE.2009.28
Costanza R, De GR, Sutton P et al (2014) Changes in the global value of ecosystem services. Glob Environ Chang 26:152–158. https://doi.org/10.1016/j.gloenvcha.2014.04.002
Culhane F, Teixeira H, Nogueira AJA et al (2019) Environment risk to the supply of ecosystem services across aquatic ecosystems. Sci Total Environ 660:611–621. https://doi.org/10.1016/j.scitotenv.2018.12.346
Dalu T, Froneman PW, Richoux NB (2014) Phytoplankton community diversity along a river-estuary continuum. Trans R Soc South Africa 69:107–116. https://doi.org/10.1080/0035919X.2014.930074
Dalu T, Wasserman RJ, Magoro ML et al (2017) Variation partitioning of benthic diatom community matrices: effects of multiple variables on benthic diatom communities in an Austral temperate river system. Sci Total Environ 601–602:73–82. https://doi.org/10.1016/j.scitotenv.2017.05.162
Dalu T, Cuthbert RN, Weyl OLF, Wasserman RJ (2022a) Community structure and environmental factors affecting diatom abundance and diversity in a Mediterranean climate river system. Sci Total Environ 810:152366. https://doi.org/10.1016/j.scitotenv.2021.152366
Dalu T, Mwedzi T, Wasserman RJ et al (2022b) Land use effects on water quality, habitat, and macroinvertebrate and diatom communities in African highland streams. Sci Total Environ 846:157346. https://doi.org/10.1016/j.scitotenv.2022.157346
Deiner K, Fronhofer EA, Mächler E et al (2016) Environmental DNA reveals that rivers are conveyer belts of biodiversity information. Nat Commun 7:12544. https://doi.org/10.1038/ncomms12544
Deng P, Lei Y, Liu W, Wang X (2012) Exploration of benthic diatom indices to evaluate water quality in rivers in the Dongjiang basin. Shengtai Xuebao/ Acta Ecol Sin 32:5014–5024. https://doi.org/10.5846/stxb201112071867
Dokulil MT, Donabaum U, Vienna LS (2016) Phytoplankton of the Danube River : composition and long-term dynamics. Acta Zool Bulg 7:147–152
Dora SL, Chowdhury A, Maiti SK, Tiwary RK (2021) Assessment of pollution load and identifying bioindicator algal species using multivariate statistical techniques: a case study from Damodar River, India. Int J Environ Pollut 69:151. https://doi.org/10.1504/IJEP.2021.126971
Edokpayi JN, Odiyo JO, Popoola EO, Msagati TAM (2017) Evaluation of temporary seasonal variation of heavy metals and their potential ecological risk in Nzhelele River, South Africa. Open Chem 15:272–282. https://doi.org/10.1515/chem-2017-0033
Ezewudo BI, Mgbenka BO, Islam S, Odo GE (2021) Appraisal of metal contamination in sediments of lower reaches of Niger River, Nigeria, using contamination indices and sediment quality guidelines. Int J Environ Anal Chem 00:1–20. https://doi.org/10.1080/03067319.2021.1895140
Fatmi B, Hazzab A, Ghenaim A, Rahmani A (2023) anthropogenic pollution and ecological risk in urban sediments: assessing heavy metal and organic contamination in the Saida watershed, North-Western Algeria. Water Air Soil Pollut 234:699. https://doi.org/10.1007/s11270-023-06701-1
Fayiga AO, Abioye O, Ipinmoroti MO et al (2018) Environmental pollution in Africa. Environ Dev Sustain 20:41–73. https://doi.org/10.1007/s10668-016-9894-4
Feisal NAS, Kamaludin NH, Abdullah Sani MF et al (2023) Anthropogenic disturbance of aquatic biodiversity and water quality of an urban river in Penang, Malaysia. Water Sci Eng 16:234–242. https://doi.org/10.1016/j.wse.2023.01.003
Feng B, Zhang M, Chen J et al (2021) Reduction in the phytoplankton index of biotic integrity in riverine ecosystems driven by industrial activities, dam construction and mining: a case study in the Ganjiang River, China. Ecol Indic 120:106907. https://doi.org/10.1016/j.ecolind.2020.106907
Ferreira V, Albariño R, Larrañaga A et al (2023) Ecosystem services provided by small streams: an overview. Hydrobiologia 850:2501–2535. https://doi.org/10.1007/s10750-022-05095-1
Ferreira da Silva E, Almeida SFPP, Nunes ML et al (2009) Heavy metal pollution downstream the abandoned Coval da Mó mine (Portugal) and associated effects on epilithic diatom communities. Sci Total Environ 407:5620–5636. https://doi.org/10.1016/j.scitotenv.2009.06.047
Follett R, Strezov V (2015) An analysis of citizen science based research: usage and publication patterns. PLoS One 10:e0143687. https://doi.org/10.1371/journal.pone.0143687
Forio MAE, Goethals PLM (2020) An integrated approach of multi-community monitoring and assessment of aquatic ecosystems to support sustainable development. Sustainability 12:5603. https://doi.org/10.3390/su12145603
Fraisl D, Hager G, Bedessem B et al (2022) Citizen science in environmental and ecological sciences. Nat Rev Methods Prim 2:64. https://doi.org/10.1038/s43586-022-00144-4
Gbogbo F, Otoo SD (2015) The concentrations of five heavy metals in components of an economically important urban coastal wetland in Ghana: public health and phytoremediation implications. Environ Monit Assess 187:655. https://doi.org/10.1007/s10661-015-4880-0
Gerber R, Smit NJ, van Vuren JHJ et al (2015) Application of a sediment quality index for the assessment and monitoring of metals and organochlorines in a premier conservation area. Environ Sci Pollut Res 22:19971–19989. https://doi.org/10.1007/s11356-015-5206-z
Gleason JE, Elbrecht V, Braukmann TWA et al (2021) Assessment of stream macroinvertebrate communities with eDNA is not congruent with tissue-based metabarcoding. Mol Ecol 30:3239–3251. https://doi.org/10.1111/mec.15597
Guo Y, Liang Y, Deng R et al (2022) Development and application of a new sensitivity analysis model for the remote sensing retrieval of heavy metals in water. Heliyon 8:e12033. https://doi.org/10.1016/j.heliyon.2022.e12033
Hama Aziz KH, Mustafa FS, Omer KM et al (2023) Heavy metal pollution in the aquatic environment: efficient and low-cost removal approaches to eliminate their toxicity: a review. RSC Adv 13:17595–17610. https://doi.org/10.1039/d3ra00723e
Hamza-Chaffai A (2014) Usefulness of bioindicators and biomarkers in pollution biomonitoring. Int J Biotechnol Wellness Ind 3:19–26. https://doi.org/10.6000/1927-3037.2014.03.01.4
Hansen BK, Jacobsen MW, Middelboe AL et al (2020) Remote, autonomous real-time monitoring of environmental DNA from commercial fish. Sci Rep 10:13272. https://doi.org/10.1038/s41598-020-70206-8
Harding B, Taylor JC (2014) Diatoms as indicators of historical water quality: a comparison of samples taken in the Wemmershoek catchment (Western Province, South Africa) in 1960 and 2008. Water SA 40:601–606. https://doi.org/10.4314/wsa.v40i4.4
Harrison JB, Sunday JM, Rogers SM (2019) Predicting the fate of eDNA in the environment and implications for studying biodiversity. Proc R Soc B Biol Sci 286:20191409. https://doi.org/10.1098/rspb.2019.1409
Hattikudur RS, Alakananda B, Ramachandra TV (2014) Diatoms as bioindicators. In: LAKE 2014: Conference on Conservation and Sustainable Management of Wetland Ecosystems in Western Ghats, 13–15 November, 2014. Sirsi, India, pp 2012–2015
Hena JS, Magaji JI, Kulawe D (2022) Dsitribution of heavy metal and phytoplankton in Calabar River port terminals, Calabar, Cross River state, Nigeria. Sci World J 17:521–525
Huang X, Zhu Y, Kianfar E (2021) Nano biosensors: properties, applications and electrochemical techniques. J Mater Res Technol 12:1649–1672. https://doi.org/10.1016/j.jmrt.2021.03.048
Huang CW, Lin C, Nguyen MK et al (2023) A review of biosensor for environmental monitoring: principle, application, and corresponding achievement of sustainable development goals. Bioengineered 14:58–80. https://doi.org/10.1080/21655979.2022.2095089
Ismail MM, El Zokm GM (2023) Evaluation of the response of phytoplankton communities to heavy metal stresses using multi-statistical approaches, Alexandria coast, Egypt. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-023-04914-9
Jeon Y, Lee Y, Jang G et al (2022) Design of Pb(II)-Specific E. coli-based biosensors by engineering regulatory proteins and host cells. Front Microbiol 13:1–12. https://doi.org/10.3389/fmicb.2022.881050
Jordaan K, Comeau AM, Khasa DP, Bezuidenhout CC (2019) An integrated insight into the response of bacterial communities to anthropogenic contaminants in a river: a case study of the Wonderfonteinspruit catchment area, South Africa. Plos One 14:e0216758. https://doi.org/10.1371/journal.pone.0216758
Kaonga CC, Chiotha SS, Monjerezi M et al (2008) Levels of cadmium, manganese and lead in water and algae; Spirogyra aequinoctialis. Int J Environ Sci Technol 5:471–478. https://doi.org/10.1007/BF03326043
Kayembe JM, Sivalingam P, Salgado CD et al (2018) Assessment of water quality and time accumulation of heavy metals in the sediments of tropical urban rivers: Case of Bumbu River and Kokolo Canal, Kinshasa City, Democratic Republic of the Congo. J African Earth Sci 147:536–543. https://doi.org/10.1016/j.jafrearsci.2018.07.016
Keck F, Vasselon V, Rimet F et al (2018) Boosting DNA metabarcoding for biomonitoring with phylogenetic estimation of operational taxonomic units’ ecological profiles. Mol Ecol Resour 18:1299–1309. https://doi.org/10.1111/1755-0998.12919
Khatri N, Tyagi S (2015) Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Front Life Sci 8:23–39. https://doi.org/10.1080/21553769.2014.933716
Kimirei IA, Mubaya CP, Ndebele-Murisa M et al (2021) Trends in ecological changes. In: Ndebele-Murisa M, Kimirei IA, Mubaya CP, Bere T (eds) Ecological changes in the Zambezi River Basin. CODESRIA, Dakar, Senegal, pp 49–82. https://doi.org/10.2307/j.ctv1grbbq5.10
Kitchenham B, Charters S (2007) Guidelines for performing systematic literature reviews in software engineering. EBSE Technical Report, EBSE-2007-01. Durham, UK
Kock A, Taylor JC, Malherbe W (2019) Diatom community structure and relationship with water quality in Lake Sibaya, KwaZulu-Natal, South Africa. South Afr J Bot 123:161–169. https://doi.org/10.1016/j.sajb.2019.03.013
Kumari D, Paul DK (2020) Assessing the role of bioindicators in freshwater ecosystem. J Interdiscipl Cycle Res 12:58–74
Laffite A, Al Salah DMM, Slaveykova VI et al (2020) Impact of anthropogenic activities on the occurrence and distribution of toxic metals, extending-spectra β-lactamases and carbapenem resistance in sub-Saharan African urban rivers. Sci Total Environ 727:138129. https://doi.org/10.1016/j.scitotenv.2020.138129
Lamare M (2019) Plankton: a guide to their ecology and monitoring for water quality, 2nd edition. New Zeal J Mar Freshw Res 53:480–480. https://doi.org/10.1080/00288330.2019.1625497
Lang P, Taylor JCC, Bertolli L et al (2013) Proposed procedure for the sampling, preparation and analysis of benthic diatoms from Zambian rivers: a bioassessment and decision support tool applicable to freshwater ecoregions in tropical southern Africa. SAFRASS Project (AFS/2009/219013) Report. East Kilbride, Scotland
Laporte M, Berger CS, García-Machado E et al (2022) Cage transplant experiment shows weak transport effect on relative abundance of fish community composition as revealed by eDNA metabarcoding. Ecol Indic 137:0–7. https://doi.org/10.1016/j.ecolind.2022.108785
Lavoie I, Lavoie M, Fortin C (2012) A mine of information: benthic algal communities as biomonitors of metal contamination from abandoned tailings. Sci Total Environ 425:231–241. https://doi.org/10.1016/j.scitotenv.2012.02.057
Lazorchak JM, Hill BH, Brown BS et al (2003) USEPA biomonitoring and bionindicator concepts needed to evaluate the biological integrity of aquatic ecosystems. In: Markert BA, Breure AM, Zechmeister HG (eds) Bioindicators & biomonitors : principles, concepts, and applications, 1st edn. Elsevier Science Ltd, New York, pp 123–152
Lee K-L, Choi JS, Lee J-H et al (2021) Response of epilithic diatom assemblages to weir construction on the Nakdong River, Republic of Korea. Ecol Indic 126:107711. https://doi.org/10.1016/j.ecolind.2021.107711
Leguay S, Lavoie I, Levy JL, Fortin C (2016) Using biofilms for monitoring metal contamination in lotic ecosystems: the protective effects of hardness and pH on metal bioaccumulation. Environ Toxicol Chem 35:1489–1501. https://doi.org/10.1002/etc.3292
Lemley DA, Adams JB, Bate GC (2016) A review of microalae as indicators in South African estuaries. South Afr J Bot 107:12–20. https://doi.org/10.1016/j.sajb.2016.04.008
Li L, Zheng B, Liu L et al (2010) Metagenomic applications in environmental monitoring and bioremediation. J Ind Microbiol Biotechnol 43:1345–1354. https://doi.org/10.1016/j.tibtech.2010.03.005
Li F, Peng Y, Fang W et al (2018) Application of environmental DNA metabarcoding for predicting anthropogenic pollution in rivers. Environ Sci Technol 52:11708–11719. https://doi.org/10.1021/acs.est.8b03869
Li Y, Zhou Q, Zhang Y et al (2021) Research trends in the remote sensing of phytoplankton blooms: results from bibliometrics. Remote Sens 13:4414. https://doi.org/10.3390/rs13214414
Lin Y, Gao J, Tu Y et al (2024) Estimating low concentration heavy metals in water through hyperspectral analysis and genetic algorithm-partial least squares regression. Sci Total Environ 916:170225. https://doi.org/10.1016/j.scitotenv.2024.170225
Limburg KE (2009) Aquatic ecosystem services. In: Likens GE (ed) Encyclopedia of inland waters, 1st edn. Elsevier, Millbrook, pp 25–30. https://doi.org/10.1016/B978-012370626-3.00004-1
Limburg KE, Swaney DP, Strayer DL (2013) River ecosystems. In: Levin SA (ed) Encyclopedia of biodiversity, 2nd edn. Elsevier, Millbrook, pp 469–484. https://doi.org/10.1016/B978-0-12-384719-5.00222-7
Lobo EA, Heinrich CG, Schuch M et al (2016) Diatoms as bioindicators in rivers. River algae. Springer International Publishing, Cham, pp 245–271
Maes J, Liquete C, Teller A et al (2020) An indicator framework for assessing ecosystem services in support of the EU biodiversity strategy to 2020. 17:14–23. https://doi.org/10.1016/j.ecoser.2015.10.023
Malthus TJ, Ohmsen R, Woerd HJ van der (2020) An evaluation of citizen science smartphone apps for inland water quality assessment. Remote Sens 12:1578. https://doi.org/10.3390/rs12101578
Mangadze T, Bere T, Mwedzi T (2016) Choice of biota in stream assessment and monitoring programs in tropical streams: a comparison of diatoms, macroinvertebrates and fish. Ecol Indic 63:128–143. https://doi.org/10.1016/j.ecolind.2015.11.029
Mangadze T, Wasserman RJ, Dalu T (2017) Use of diatom communities as indicators of conductivity and ionic composition in a small austral temperate river system. Water Air Soil Pollut 228:428. https://doi.org/10.1007/s11270-017-3610-3
Mangadze T, Dalu T, William Froneman P (2019a) Biological monitoring in southern Africa: a review of the current status, challenges and future prospects. Sci Total Environ 648:1492–1499. https://doi.org/10.1016/j.scitotenv.2018.08.252
Mangadze T, Taylor JC, Froneman WP, Dalu T (2019b) Water quality assessment in a small austral temperate river system (Bloukrans River system, South Africa): application of multivariate analysis and diatom indices. South Afr J Bot 125:353–359. https://doi.org/10.1016/j.sajb.2019.08.008
Maphanga T, Chidi BS, Phungela TT et al (2024) The interplay between temporal and seasonal distribution of heavy metals and physiochemical properties in Kaap River. Int J Environ Sci Technol 21:6053–6064. https://doi.org/10.1007/s13762-023-05401-x
Marcel R, Berthon V, Castets V, et al (2017) Modelling diatom life forms and ecological guilds for river biomonitoring. Knowl Manag Aquat Ecosyst 1. https://doi.org/10.1051/kmae/2016033
Marques JA, Costa SR, Maraschi AC et al (2022) Biochemical response and metals bioaccumulation in planktonic communities from marine areas impacted by the Fundão mine dam rupture (southeast Brazil). Sci Total Environ 806:150727. https://doi.org/10.1016/j.scitotenv.2021.150727
Masouras A, Karaouzas I, Dimitriou E et al (2021) Benthic diatoms in river biomonitoring-present and future perspectives within the water framework directive. Water (Switzerland) 13. https://doi.org/10.3390/w13040478
Mccabe DJ (2010) Rivers and streams: life in flowing water streams and rivers: habitats partitioned at different spatial scales. Nat Educ Knowl 1:1–14
MEA (2005) Ecosystems and human well-being: opportunities for business and industry. A report of the millennium ecosystem assessment. Island Press, Washington, DC
Miguel-Chinchilla L, Heasley E, Loiselle S, Thornhill I (2019) Local and landscape influences on turbidity in urban streams: a global approach using citizen scientists. Freshw Sci 38:303–320. https://doi.org/10.1086/703460
Mishra S, Bharagava RN, More N et al (2019) Heavy metal contamination: an alarming threat to environment and human health. In: Sobti RC, Arora NK, Kothari R (eds) Environmental biotechnology: for sustainable future. Springer, Singapore, pp 103–125. https://doi.org/10.1007/978-981-10-7284-0_5
Mohajane C, Manjoro M (2022) Sediment-associated heavy metal contamination and potential ecological risk along an urban river in South Africa. Heliyon 8:e12499. https://doi.org/10.1016/j.heliyon.2022.e12499
Mohammed AH, Khalifa AM, Mohamed HM et al (2024) Assessment of heavy metals at mangrove ecosystem, applying multiple approaches using in-situ and remote sensing techniques, Red Sea, Egypt. Environ Sci Pollut Res 31:8118–8133. https://doi.org/10.1007/s11356-023-31625-y
Moharana T (2021) The Science of Citizen Science. Proc Nov Scotian Inst Sci 51:4. https://doi.org/10.15273/pnsis.v51i2.11167
Moyo S, McCrindle R, Mokgalaka N, Myburgh J (2015) Heavy metal partitioning in sediments from rivers flowing through coal fields in Mpumalanga, South Africa. CLEAN - Soil, Air, Water 43:892–900. https://doi.org/10.1002/clen.201300656
Mwedzi T, Siziba N, Odume ON et al (2020) Responses of macroinvertebrate community metrics to urban pollution in semi-arid catchments around the city of Bulawayo, Zimbabwe. Water SA 46:583–592. https://doi.org/10.17159/wsa/2020.v46.i4.9071
Ngueyep MLL, Kingni KS, Ngounouno NM et al (2021) The impact of gold mining exploitation on the physicochemical quality of water: case of Batouri (Cameroon). Int J Energy Water Resour 5:159–173. https://doi.org/10.1007/s42108-020-00106-0
Nguyen TH, Helm B, Hettiarachchi H et al (2019) The selection of design methods for river water quality monitoring networks: a review. Environ Earth Sci 78:96. https://doi.org/10.1007/s12665-019-8110-x
Ngure V, Davies T, Kinuthia G et al (2014) Concentration levels of potentially harmful elements from gold mining in Lake Victoria Region, Kenya: environmental and health implications. J Geochem Explor 144:511–516. https://doi.org/10.1016/j.gexplo.2014.04.004
Njue N, Stenfert Kroese J, Gräf J et al (2019) Citizen science in hydrological monitoring and ecosystem services management: state of the art and future prospects. Sci Total Environ 693:133531. https://doi.org/10.1016/j.scitotenv.2019.07.337
Nkinda MS, Rwiza MJ, Ijumba JN, Njau KN (2020) Quantitative assessment of metal contamination and associated pollution risk in sediments from the Mara River in Tanzania. Environ Monit Assess 192:721. https://doi.org/10.1007/s10661-020-08681-9
Norros V, Laamanen T, Meissner K et al (2022) Roadmap for implementing environmental DNA (eDNA) and other molecular monitoring methods in Finland. Reports of the Finnish environment institute. Finnish Environment Institute, Helsinki
Nourmohammadi E, Hosseinkhani S, Nedaeinia R et al (2020) Construction of a sensitive and specific lead biosensor using a genetically engineered bacterial system with a luciferase gene reporter controlled by pbr and cadA promoters. Biomed Eng Online 19:1–13. https://doi.org/10.1186/s12938-020-00816-w
Oberholster PJ, De Klerk AR, De Klerk L et al (2016) Algal assemblage responses to acid mine drainage and steel plant wastewater effluent up and downstream of pre and post wetland rehabilitation. Ecol Indic 62:106–116. https://doi.org/10.1016/j.ecolind.2015.11.025
Oberholster PJ, Botha A-M, Hill L, Strydom WF (2017) River catchment responses to anthropogenic acidification in relationship with sewage effluent: an ecotoxicology screening application. Chemosphere 189:407–417. https://doi.org/10.1016/j.chemosphere.2017.09.084
Ochieng EZ, Lalah JO, Wandiga SO (2009) Anthropogenic sources of heavy metals in the Indian Ocean coast of Kenya. Bull Environ Contam Toxicol 83:600–607. https://doi.org/10.1007/s00128-009-9807-4
Ogoyi DO, Mwita CJJ, Nguu EK, Shiundu PM (2011) Determination of heavy metal content in water, sediment and microalgae from Lake Victoria, East Africa. Open Environ Eng J 4:156–161. https://doi.org/10.2174/1874829501104010156
Omara T, Karungi S, Kalukusu R et al (2019) Mercuric pollution of surface water, superficial sediments, Nile tilapia (Oreochromis nilotica Linnaeus 1758 [Cichlidae]) and yams ( Dioscorea alata) in auriferous areas of Namukombe stream, Syanyonja, Busia, Uganda. PeerJ 7:e7919. https://doi.org/10.7717/peerj.7919
Ouma KO, Shane A, Syampungani S (2022) Aquatic ecological risk of heavy-metal pollution associated with degraded mining landscapes of the Southern Africa River Basins: a review. Minerals 12:225. https://doi.org/10.3390/min12020225
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLOS Med 18:e1003583. https://doi.org/10.1371/journal.pmed.1003583
Pandey LK (2020) In situ assessment of metal toxicity in riverine periphytic algae as a tool for biomonitoring of fluvial ecosystems. Environ Technol Innov 18:100675. https://doi.org/10.1016/j.eti.2020.100675
Pandey LK, Bergey EA (2018) Metal toxicity and recovery response of riverine periphytic algae. Sci Total Environ 642:1020–1031. https://doi.org/10.1016/j.scitotenv.2018.06.069
Parmar TK, Rawtani D, Agrawal YK (2016) Bioindicators: the natural indicator of environmental pollution. Front Life Sci 9:110–118. https://doi.org/10.1080/21553769.2016.1162753
Pascale BM (2023) Spatio-temporal distribution of phytoplankton and water quality of some rivers of Haute Sanaga department ( Central Africa ). Biol LIfe Sci 14:1–18. https://doi.org/10.5296/jbls.v14i1.20181
Pereira-da-Conceicoa L, Elbrecht V, Hall A et al (2021) Metabarcoding unsorted kick-samples facilitates macroinvertebrate-based biomonitoring with increased taxonomic resolution, while outperforming environmental DNA. Environ DNA 3:353–371. https://doi.org/10.1002/edn3.116
Pattinson NB, Taylor J, Dickens CWS, Graham PM (2023) Digital innovation in citizen science to enhance water quality monitoring in developing countries. Colombo, Sri Lanka: International Water Management Institute (IWMI). 37. (IWMI working paper 210). https://doi.org/10.5337/2024.201
Perry I, Jâms IB, Casas-Mulet R et al (2022) Challenges to implementing environmental-DNA monitoring in Namibia. Front Environ Sci 9:773991. https://doi.org/10.3389/fenvs.2021.773991
Petticrew M, Roberts H (2008) Systematic reviews in the social sciences: a practical guide. Blackwell Publishing, Oxford. https://doi.org/10.1002/9780470754887
Pettorelli N, Schulte to Bühne H, Tulloch A et al (2018) Satellite remote sensing of ecosystem functions: opportunities, challenges and way forward. Remote Sens Ecol Conserv 4:71–93. https://doi.org/10.1002/rse2.59
Rajfur M, Kłos A, Wacławek M (2010) Sorption properties of algae Spirogyra sp. and their use for determination of heavy metal ions concentrations in surface water. Bioelectrochemistry 80:81–86. https://doi.org/10.1016/j.bioelechem.2010.03.005
Ramezanpour Z, Pourafrasyabi M, Ramezanpour Z (2014) Phytoplankton as bio-indicator of water quality in Sefid Rud River – Iran (South Caspian Sea). Casp J Environ Sci 12:31–40
Reddy GPO (2018) Satellite remote sensing sensors: principles and applications. In: Singh SK (ed) Reddy GPO. Springer International Publishing, Cham, pp 21–43
Resh VH (2007) Multinational, freshwater biomonitoring programs in the developing world: lessons learned from African and Southeast Asian River surveys. Environ Manag 39:737–748. https://doi.org/10.1007/s00267-006-0151-8
Respondek Z, Jerz D, Świsłowski P, Rajfur M (2022) Active biomonitoring of heavy metal concentrations in aquatic environment using mosses and algae. Water 14:3335. https://doi.org/10.3390/w14203335
Rivera SF, Vasselon V, Bouchez A, Rimet F (2020) Diatom metabarcoding applied to large scale monitoring networks: optimization of bioinformatics strategies using Mothur software. Ecol Indic 109:105775. https://doi.org/10.1016/j.ecolind.2019.105775
Rovira J, Domingo JL (2019) Human health risks due to exposure to inorganic and organic chemicals from textiles: a review. Environ Res 168:62–69. https://doi.org/10.1016/j.envres.2018.09.027
Ruppen D, Brugger F (2022) “I will sample until things get better – or until I die.” Potential and limits of citizen science to promote social accountability for environmental pollution. World Dev 157:105952. https://doi.org/10.1016/j.worlddev.2022.105952
Schwacke LH, Gulland FM, White S (2013) Sentinel species in oceans and human health. In: Laws EA (ed) environmental toxicology. Springer New York, New York, pp 503–528
Silva TT, Medeiros G, Amaral MWW et al (2022) Taxonomic and morphofunctional phytoplankton response to environmental variability in rivers from different hydrographic basins in Southern Brazil. Acta Limnol Bras 34:e23. https://doi.org/10.1590/S2179-975X1222
Simionov IA, Cristea DS, Petrea SM et al (2021) Preliminary investigation of lower Danube pollution caused by potentially toxic metals. Chemosphere 264:128496. https://doi.org/10.1016/j.chemosphere.2020.128496
Singh S, Kumar V, Dhanjal DS et al (2020) Biological biosensors for monitoring and diagnosis. In: Singh J, Vyas A, Wang S, Prasad R (eds) Microbial biotechnology: basic research and applications. Environmental and Microbial Biotechnology. Springer, Singapore, pp 317–335. https://doi.org/10.1007/978-981-15-2817-0_14
Sirunda J, Oberholster P, Wolfaardt G et al (2021) The assessment of phytoplankton dynamics in two reservoirs in Southern Africa with special reference to water abstraction for inter-basin transfers and potable water production. Water 13:3045. https://doi.org/10.3390/w13213045
Smucker NJ, Kuhn A, Cruz-Quinones CJ et al (2018) Stable isotopes of algae and macroinvertebrates in streams respond to watershed urbanization, inform management goals, and indicate food web relationships. Ecol Indic 90:295–304. https://doi.org/10.1016/j.ecolind.2018.03.024
Solak CN, Peszek Ł, Yilmaz E et al (2020) Use of diatoms in monitoring the Sakarya River basin, Turkey. Water (switzerland) 12:1–20. https://doi.org/10.3390/w12030703
Stat M, Huggett MJ, Bernasconi R et al (2017) Ecosystem biomonitoring with eDNA: metabarcoding across the tree of life in a tropical marine environment. Sci Rep 7:12240. https://doi.org/10.1038/s41598-017-12501-5
Steward AL, Datry T, Langhans SD (2022) The terrestrial and semi-aquatic invertebrates of intermittent rivers and ephemeral streams. Biol Rev 97:1408–1425. https://doi.org/10.1111/brv.12848
Sumudumali RGI, Jayawardana JMCK (2021) A review of biological monitoring of aquatic ecosystems approaches: with special reference to macroinvertebrates and pesticide pollution. Environ Manag 67:263–276. https://doi.org/10.1007/s00267-020-01423-0
Syeed MMM, Hossain MS, Karim MR et al (2023) Surface water quality profiling using the water quality index, pollution index and statistical methods: a critical review. Environ Sustain Indic 18:100247. https://doi.org/10.1016/j.indic.2023.100247
Talbot CJ, Bennett EM, Cassell K et al (2018) The impact of flooding on aquatic ecosystem services. Biogeochemistry 141:439–461. https://doi.org/10.1007/s10533-018-0449-7
Tapia PM (2008) Diatoms as bioindicators of pollution in the Mantaro River, Central Andes. Peru Int J Environ Heal 2:82. https://doi.org/10.1504/IJENVH.2008.018674
Thompson PA, Carstensen J (2023) Global observing for phytoplankton? A perspective. J Plankton Res 45:221–234. https://doi.org/10.1093/plankt/fbab090
Thornhill I, Ho JG, Zhang Y et al (2017) Prioritising local action for water quality improvement using citizen science; a study across three major metropolitan areas of China. Sci Total Environ 584–585:1268–1281. https://doi.org/10.1016/j.scitotenv.2017.01.200
Tolotti R, Consani S, Carbone C et al (2019) Benthic diatom community response to metal contamination from an abandoned Cu mine: case study of the Gromolo Torrent (Italy). J Environ Sci (china) 75:233–246. https://doi.org/10.1016/j.jes.2018.03.034
Torrisi M, Scuri S, Dell’Uomo A, Cocchioni M (2010) Comparative monitoring by means of diatoms, macroinvertebrates and chemical parameters of an Apennine watercourse of central Italy: the river Tenna. Ecol Indic 10:910–913. https://doi.org/10.1016/j.ecolind.2010.01.010
Tyovenda AA, Ikpughul SI, Sombo T (2019) Assessment of heavy metal pollution of water, sediments and algae in river Benue at Jimeta-Yola, Adamawa State, Nigeria. Niger Ann Pure Appl Sci 1:186–195. https://doi.org/10.46912/napas.44
Ugbeyide JA, Ugwumba OA (2021) Water quality and phytoplankton as indicators of pollution in Ibuya River. Br J Environ Sci 9:26–39
Vetrivel SA, Diptanghu M, Ebhin MR et al (2017) Green algae of the genus Spirogyra: a potential absorbent for heavy metal from coal mine water. Remediation 27:81–90. https://doi.org/10.1002/rem.21522
Wijngaard RR, van der Perk M, van der Grift B et al (2017) The impact of climate change on metal transport in a lowland catchment. Water Air Soil Pollut 228:107. https://doi.org/10.1007/s11270-017-3261-4
Wohl E (2018) Rivers as ecosystems. In: Wohl E (ed) Sustaining river ecosystems and water resources. SpringerBriefs in environmental science. Springer International Publishing, Cham, pp 11–58. https://doi.org/10.1007/978-3-319-65124-8_2
Wright K, Golder S, Rodriguez-Lopez R (2014) Citation searching: a systematic review case study of multiple risk behaviour interventions. BMC Med Res Methodol 14:73. https://doi.org/10.1186/1471-2288-14-73
Wu B, Ga L, Wang Y, Ai J (2023) Recent advances in the application of bionanosensors for the analysis of heavy metals in aquatic environments. Molecules 29:34. https://doi.org/10.3390/molecules29010034
Xia XH, Wu Q, Mou XL, Lai YJ (2015) Potential impacts of climate change on the water quality of different water bodies. J Environ Inf 25:85–98. https://doi.org/10.3808/jei.201400263
Yabe J, Ishizuka M, Umemura T (2010) Current levels of heavy metal pollution in Africa. J Vet Med Sci 72:1257–1263. https://doi.org/10.1292/jvms.10-0058
Yahya AN, Mohamed SK, Mohamed AG (2018) Environmental pollution by heavy metals in the aquatic ecosystems of Egypt. Open Access J Toxicol 3:555603. https://doi.org/10.19080/OAJT.2018.03.555603
Yilmaz N, Ozyigit II, Demir HH, Yalcin IE (2021) Assessment on phytoplankton composition and heavy metal pollution in a drinking water resource: Lake Terkos (Istanbul, Turkey). Desalin Water Treat 225:265–274. https://doi.org/10.5004/dwt.2021.27221
Yu H, Kong B, He Z-W et al (2020) The potential of integrating landscape, geochemical and economical indices to analyze watershed ecological environment. J Hydrol 583:124298. https://doi.org/10.1016/j.jhydrol.2019.124298
Zhou Q, Zhang J, Fu J et al (2008) Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal Chim Acta 606:135–150. https://doi.org/10.1016/j.aca.2007.11.018
Zhou Q, Yang N, Li Y et al (2020) Total concentrations and sources of heavy metal pollution in global river and lake water bodies from 1972 to 2017. Glob Ecol Conserv 22:e00925. https://doi.org/10.1016/j.gecco.2020.e00925
Zhu M, Wang J, Yang X et al (2022) A review of the application of machine learning in water quality evaluation. Eco-Environ Heal 1:107–116. https://doi.org/10.1016/j.eehl.2022.06.001
Acknowledgements
This research was supported by the Chair-Environment and Development, Oliver R. Tambo of the Africa Research Initiative (ORTARChI) Project at the Copperbelt University, Zambia. ORTARChI is an initiative of South Africa’s National Research Foundation (NRF) and the Department of Science and Innovation (DSI) in partnership with the Oliver and Adelaide Tambo Foundation (OATF), Canada’s International Development Research Centre (IDRC), and the National Science and Technology Council (NSTC) of Zambia. The findings and conclusions in this publication are those of the authors and should not be construed to represent any official position of the organisations that funded the study.
Funding
Open access funding provided by University of Pretoria. This review was funded by the Chair-Environment and Development, Oliver R. Tambo of the Africa Research Chair Initiative (ORTARChI), Copperbelt University, Zambia.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.M., C.M., and K.O.; methodology, M.M., K.O., and S.S; validation, M.M., C.M., K.O., T.J., and S.S.; formal analysis, M.M., C.M., and K.O.; investigation, M.M and K.O.; literature and data curation, M.M. and K.O.; writing—original draft, M.M., C.M., K.O., T.J., and S.S.; writing—review and editing, M.M., C.M., K.O, T.J., and S.S.; supervision, C.M., T.J., and S.S. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Robert Duran
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mulenga, M., Monde, C., Johnson, T. et al. Advances in the integration of microalgal communities for biomonitoring of metal pollution in aquatic ecosystems of sub-Saharan Africa. Environ Sci Pollut Res 31, 40795–40817 (2024). https://doi.org/10.1007/s11356-024-33781-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-33781-1