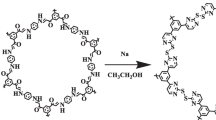
Abstract
Thiosulfate gold leaching is one of the most promising green cyanide-free gold extraction processes; however, the difficulty of recovering Au(I) from the leaching system hinders its further development. This study prepared aminoguanidine-functionalized microspheres (AGMs) via a one-step reaction involving nucleophilic substitution between aminoguanidine hydrochloride and chloromethylated polystyrene microspheres and used AGMs to adsorb Au(I) from thiosulfate solutions. Scanning electron microscopy, Brunauer–Emmett–Teller analysis, Fourier transform infrared spectroscopy, and X-ray photoelectron spectroscopy were used to analyze the structure and properties of AGMs. Experiments were designed to investigate the effects of pH, temperature, initial Au(I), and thiosulfate concentrations on the gold adsorption performance of AGMs. Results demonstrated that AGMs can efficiently adsorb Au(I) from thiosulfate solutions in a wide pH range. The adsorption process conforms to the pseudo-second-order kinetic model and Langmuir isotherm model, with a maximum capacity of 22.03 kg/t. Acidic thiourea is an effective desorbent, and after four adsorption–desorption cycles, the adsorption rate of Au(I) by AGMs is 78.63%, which shows AGMs have good cyclic application potential. Based on the results of characterization, experiments, and density functional theory calculations, the mechanism for the adsorption of [Au(S2O3)2]3− on AGMs involves anion exchange. Importantly, AGMs exhibited satisfactory adsorption property for Au(I) in practical Cu2+–NH3(en)–S2O32− systems. This study provided experimental reference for the recovery of Au(I) from thiosulfate solution.
Graphical Abstract












Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Abbruzzese C, Fomari P, Massidda R, Veglio F, Ubaldini S (1995) Thiosulphate leaching for gold hydrometallurgy. Hydrometallurgy 39:265–276
Awadalla FT, Ritcey GM (1991) Recovery of gold from thiourea, thiocyanate, or thiosulfate solutions by reduction-precipitation with a stabilized form of sodium borohydride. Sep Sci Technol 26(9):1207–1228
Awual MR, Hasan MM, Eldesoky GE, Khaleque MA, Rahman MM, Naushad M (2016) Facile mercury detection and removal from aqueous media involving ligand impregnated conjugate nanomaterials. Chem Eng J 290:243–251
Awual MR, Hasan MM, Islam A, Asiri AM, Rahman MM (2020) Optimization of an innovative composited material for effective monitoring and removal of cobalt(II) from wastewater. J Mol Liq 298:112035
Brion D (1979) Etude par spectroscopie de photoelectrons de la degradation superficielle de FeS2, CuFeS2, ZnS et PbS a l’air et dans l’eau. App Surface Sci 5:133–152
Chen S, Zi F, Hu X, Chen Y, Yang P, Wang Q, Qin X, Cheng H, Liu Y, He Y et al (2020) Interfacial properties of mercaptopropyl-functionalised silica gel and its adsorption performance in the recovery of gold(I) thiosulfate complex. Chem Eng J 393:124547
Dang Q, Li C-G, Jin X-X, Zhao Y-J, Wang X (2019) Heparin as a molecular spacer immobilized on microspheres to improve blood compatibility in hemoperfusion. Carbohyd Polym 205:89–97
Descostes M, Mercier F, Thromat N, Beaucaire C, Gautier-Soyer M (2000) Use of XPS in the determination of chemical environment and oxidation state of iron and sulfur samples: constitution of a data basis in binding energies for Fe and S reference compounds and applications to the evidence of surface species of an oxidized pyrite in a carbonate medium. Appl Surf Sci 165(4):288–302
Dong Z, Jiang T, Xu B, Yang Y, Li Q (2017) Recovery of gold from pregnant thiosulfate solutions by the resin adsorption technique. Metals 7(12):555
Du J, Dong Z, Wen D, Yang X, Zhai M, Hua R, Zhao L (2022) Selective recovery of rhenium from the simulating leaching solutions of uranium ore by amino guanidine functionalized microcrystalline cellulose microsphere. J Mol Liq 360:119399
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, M AR, J RC, Scalmani G, Barone V, G AP, Nakatsuji H, Li X, Caricato M, A VM, Bloino J, B GJ, Gomperts R, Mennucci B, H PH, J VO, A FI, J LS, Williams, Ding F, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, V GZ, Gao JNR, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, J AMJ, J EP, Ogliaro F, M JB, J JH, E NB, K NK, V NS, T AK, Kobayashi R, Normand J, Raghavachari K, A PR, J CB, S SI, Tomasi J, Cossi M, J MM, Klene M, Adamo C, Cammi R, J WO, R LM, Morokuma K, Farkas O, J BF, D JF (2009) Gaussian 09 revision A.02. CT
Gurung M, Adhikari BB, Morisada S, Kawakita H, Ohto K, Inoue K, Alam S (2013) N-aminoguanidine modified persimmon tannin: A new sustainable material for selective adsorption, preconcentration and recovery of precious metals from acidic chloride solution. Biores Technol 129:108–117
Gurung M, Adhikari BB, Gao XP, Alam S, Inoue K (2014) Sustainability in the Metallurgical Industry: Chemically Modified Cellulose for Selective Biosorption of Gold from Mixtures of Base Metals in Chloride Media. Ind Eng Chem Res 53(20):8565–8576
Harunobu A, Toyohisa F, Wan-Tai Y (2002) Gold cementation from ammonium thiosulfate solution by zinc. Copper Aluminium Powders Mater Trans 43:485–493
Hilson G, Monhemius AJ (2006) Alternatives to cyanide in the gold mining industry: what prospects for the future? J Clean Prod 14(12–13):1158–1167
Hossain MS, Shenashen MA, Awual ME, Rehan AI, Rasee AI, Waliullah RM, Kubra KT, Salman MS, Sheikh MC, Hasan MN et al (2024) Benign separation, adsorption, and recovery of rare-earth Yb(III) ions with specific ligand-based composite adsorbent. Process Saf Environ Prot 185:367–374
Huang T, Zhang S-w, Zhou L, Liu L-f (2021) Electrokinetics couples with the adsorption of activated carbon-supported hydroxycarbonate green rust that enhances the removal of Sr cations from the stock solution in batch and column. Sep Purif Technol 265:118531
Huang T, Pan L, Dong J, Zhou L, Tao H, Zhang S-w, Li A (2022) A comprehensive investigation of zeolite-rich tuff functionalized with 3-mercaptopropionic acid intercalated green rust for the efficient removal of HgII and CrVI in a binary system. J Environ Manage 324:116344
Jeffrey MI, Breuer PL, Chu CK (2003) The importance of controlling oxygen addition during the thiosulfate leaching of gold ores. Int J Miner Process 72(1–4):323–330
Jeon S, Ito M, Tabelin CB, Pongsumrankul R, Kitajima N, Park I, Hiroyoshi N (2018) Gold recovery from shredder light fraction of E-waste recycling plant by flotation-ammonium thiosulfate leaching. Waste Management 77:195–202
Jermakowicz-Bartkowiak D, Kolarz BN (2002) Gold sorption on weak base anion exchangers with aminoguanidyl groups. Eur Polymer J 38(11):2239–2246
Ji C, Qu R, Sun C, Wang C, Sun Y, Zhao N, Xie H (2006) Preparation and adsorption selectivity for Hg(II) and Ag(I) of chelating resin immobilizing benzothiazolyl group on crosslinked polystyrene via hydrophilic sulfur-containing PEG spacer. J Appl Polym Sci 100(6):5034–5038
Ji Y, Zhang W, Yang H, Ma F, Xu F (2021) Green synthesis of poly(pyrrole methane) for enhanced adsorption of anionic and cationic dyes from aqueous solution. J Colloid Interface Sci 590:396–406
Kasper AC, Veit HM, García-Gabaldón M, Herranz VP (2018) Electrochemical study of gold recovery from ammoniacal thiosulfate, simulating the PCBs leaching of mobile phones. Electrochim Acta 259:500–509
Kejun L, Yen WT, Shibayama A, Miyazaki T, Fujita T (2004) Gold extraction from thiosulfate solution using trioctylmethylammonium chloride. Hydrometallurgy 73(1–2):41–53
Kenawy IM, Hafez MAH, Ismail MA, Hashem MA (2018) Adsorption of Cu(II), Cd(II), Hg(II), Pb(II) and Zn(II) from aqueous single metal solutions by guanyl-modified cellulose. Int J Biol Macromol 107:1538–1549
Li D-P, Zhang Y-R, Zhao X-X, Zhao B-X (2013) Magnetic nanoparticles coated by aminoguanidine for selective adsorption of acid dyes from aqueous solution. Chem Eng J 232:425–433
Li X, Hu X, Zi F, Chen S, Yang P, Zhang Y, Chen Y, Zhao L, Lin Y, Jiang Y et al (2022) Efficient environmental recovery of Au(I) from thiosulfate solution on triphenylphosphine-oxide-modified vermiculite. J Clean Prod 374:133993
Liu Y, Jia H, Li C, Sun Z, Pan Y, Zheng S (2019) Efficient removal of gaseous formaldehyde by amine-modified diatomite: a combined experimental and density functional theory study. Environ Sci Pollut Res 26(24):25130–25141
Liu C, Mao Y, Yang Y, Song Z, Wang C, Kang H, Yan X, Gu D, Yan X, Wang Z (2022) Modification assisted Cu2O/MCM-41 nanoarchitecture toward efficient recovery of Au (III). Chem Eng J 448:137651
Mayhan WG, Sharpe GM (2000) Aminoguanidine Suppresses Basal Macromolecular Extravasation during Diabetes Mellitus. Microvasc Res 59(11):52–60
Nie Y, Chen J, Wang Q, Shi C, Xing B, Jiang Y, Wang S (2022) Selective Reduction of Au(I) from a High-Concentration Thiosulfate Solution for Gold Recovery Using One-Step Heat-Treated High-Sulfur Coal. ACS Sustainable Chemistry & Engineering 10(18):5875–5887
Rahman MM, Shafiullah AZ, Pal A, Islam MA, Jahan I, Saha BB (2021) Study on Optimum IUPAC Adsorption Isotherm Models Employing Sensitivity of Parameters for Rigorous Adsorption System Performance Evaluation. Energies 14(22):7478
Senanayake G (2012) Gold leaching by copper(II) in ammoniacal thiosulphate solutions in the presence of additives. Part I: A review of the effect of hard–soft and Lewis acid-base properties and interactions of ions. Hydrometallurgy 115–116:1–20
Shen W, Qu Y, Li X, Pei X, You S, Yin Q, Wang J, Ma Q (2018) Comparison of gold nanoparticles biosynthesized by cell-free extracts of Labrys, Trichosporon montevideense, and Aspergillus. Environ Sci Pollut Res 25(14):13626–13632
Tamayo JEAA, Gomez SA, Calderon JA (2017) Selective electrochemical gold recovery from leaching solutions of thiosulfate. J Electrochem Soc 164(7):D363–D370
Wang J, Lu Y, Xu Z (2019a) Identifying extraction technology of gold from solid waste in terms of environmental friendliness. ACS Sustain Chem Eng 7(7):7260–7267
Wang Q, Hu X, Zi F, Yang P, Chen Y, Chen S (2019b) Environmentally friendly extraction of gold from refractory concentrate using a copper – ethylenediamine – thiosulfate solution. J Clean Prod 214:860–872
Watling KM, Hope GA, Woods R, Jeffrey MI (2006) Surface Products on Gold leached in Ammoniacal Copper(II) Thiosulfate Solution. ECS Trans 3(2):121–132
Wei Y, Zhang K, Cheng T, Zhou G (2023) Characteristics and Mechanisms of Cr(VI) Removal from Aqueous Solution by FeSm/BC Composite. Water Air Soil Pollut 234(2):060843
Xiong C, Zheng Y, Feng Y, Yao C, Ma C, Zheng X, Jiang J (2014) Preparation of a novel chloromethylated polystyrene-2-amino-1, 3, 4-thiadiazole chelating resin and its adsorption properties and mechanism for separation and recovery of Pt(iv) from aqueous solutions. J Mater Chem A 2(15):5379–5386
Xiong C, Feng Y, Fang L, Cao Y, Song X, Wang J, Wang H, Zheng J, Zhou H (2017) Preparation of a novel chloromethylated polystyrene-2-mercapto-1,3,4-thiadiazole chelating resin and its adsorption properties and mechanism for separation and recovery of Hg(II) from aqueous solutions. Water Sci Technol 76(7):1915–1924
Xu B, Yang Y, Li Q, Jiang T, Zhang X, Li G (2017) Effect of common associated sulfide minerals on thiosulfate leaching of gold and the role of humic acid additive. Hydrometallurgy 171:44–52
Yu H, Zi F, Hu X, Zhong J, Nie Y, Xiang P (2014) The copper–ethanediamine–thiosulphate leaching of gold ore containing limonite with cetyltrimethyl ammonium bromide as the synergist. Hydrometallurgy 150:178–183
Yu H, Zi F, Hu X, Nie Y, Xiang P, Xu J, Chi H (2015) Adsorption of the gold–thiosulfate complex ion onto cupric ferrocyanide (CuFC)-impregnated activated carbon in aqueous solutions. Hydrometallurgy 154:111–117
Yu H, Zi F, Hu X, Nie Y, Chen Y, Cheng H (2017) Adsorption of gold from thiosulfate solutions with chemically modified activated carbon. Adsorpt Sci Technol 36(1–2):698864
Zhao L, Hu X, Zi F, Chen S, Cheng H, Yang P, Zhang Y, Chen Y, Jiang Y, Li X et al (2022) Development of Stable, Efficient, and Recyclable Amine-Containing Microspheres for Gold(I) Thiosulfate Complex Recovery. ACS Sustainable Chemistry & Engineering 10(44):14624–14635
Acknowledgements
The authors sincerely express gratitude to Professor Guocai Tian of the Kunming University of Science and Technology for his help and support.
Funding
This work was financially supported by the National Natural Science Foundation of China (52264031) and Yunnan Provincial Key Laboratory of Chemical Separation and Enrichment in China (KKPH201907003).
Author information
Authors and Affiliations
Contributions
Yihuai Yang: writing—original draft, validation, formal analysis, visualization, methodology, investigation, conceptualization, data curation. Xianzhi Hu and Futing Zi: methodology, writing—review editing, funding acquisition, resources, supervision, project administration. Shuliang Chen: resources, software, formal analysis. Li Zhao: software, visualization, formal analysis. Xinrong Li: resources, visualization, software. Yue Lin: software, formal analysis, validation.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All the authors of this article have known the content and author signature and ranking and agree to submit the article.
Consent for publication
All the authors of this article have known the content of the article and author signature and ranking and agree to publish it.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. Aminoguanidine functionalized microspheres prepared by one-step reaction, the preparation process is simple.
2. Functionalized microspheres can efficiently adsorb gold in thiosulfate solutions over a wide pH range.
3. Au(I) adsorption onto functionalized microspheres involves anion exchange between Cl− and [Au(S2O3)2]3−.
4. The prepared aminoguanidine functionalized microspheres have good potential for application in the adsorption–desorption cycle.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, Y., Hu, X., Zi, F. et al. Effectively adsorb Au(S2O3)23− using aminoguanidine as trapping group from thiosulfate solutions. Environ Sci Pollut Res 31, 38298–38309 (2024). https://doi.org/10.1007/s11356-024-33710-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-33710-2