Abstract
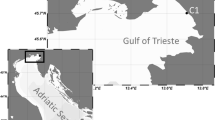
In mid-November 2021, there were large areas of white rot disease on cultivated Saccharina japonica in Rongcheng City, China, and diseases were undetected on Sargassum horneri and Porphyra yezoensis. The disturbance direction of bacterial community in the phycosphere after disease outbreak and the relationship with seawater nutrients remain unclear. Here, in situ studies of bacterial community in the non-diseased and diseased areas (Shawo and Dongchu islands) and seawater nutrient levels were carried out. 16S rRNA sequencing showed that the bacterial richness of the studied seaweeds increased in the diseased area. Only in S. japonica, Algitalea outcompeted abundant primary bacteria with probiotic relationships to the host of the non-diseased area, and dominated in the diseased area (17.6% of the total abundance). Nitrogen and phosphorus levels in seawater were 57.8% and 19.6% higher in the non-diseased area than those in the diseased area, respectively, and were strongly correlated with the phycosphere bacteria at the family level of S. japonica. There was no difference in potential pathogenicity between the two areas, while positive signal communications decreased, and nitrogen cycle, chemoheterotrophy, and cellulolysis increased in the diseased area compared to the non-diseased area. Overall, white rot disease caused a structural disturbance in phycosphere bacterial community of S. japonica that related to seawater nutrient levels. Enriched degraders and altered bacterial community functions may exacerbate the disease. This evaluation will provide information for white rot disease management to prevent and mitigate the occurrence of S. japonica outbreaks.






Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Alfiansah YR, Hassenrück C, Kunzmann A et al (2018) Bacterial abundance and community composition in pond water from shrimp aquaculture systems with different stocking densities. Front Microbiol 9:2457. https://doi.org/10.3389/fmicb.2018.02457
Amin SA, Hmelo LR, van Tol HM et al (2015) Interaction and signalling between a cosmopolitan phytoplankton and associated bacteria. Nature 522:98–101. https://doi.org/10.1038/nature14488
Astafyeva Y, Gurschke M, Qi M et al (2022) Microalgae and bacteria interaction-evidence for division of diligence in the alga microbiota. Microbiol Spectr 10(4):e0063322. https://doi.org/10.1128/spectrum.00633-22
Bell TW, Siegel DA (2021) Nutrient availability and senescence spatially structure the dynamics of a foundation species. Proc Natl Acad Sci U S A 119(1):e2105135118. https://doi.org/10.1073/pnas.2105135118
Bi D, Yang X, Yao L et al (2022) Potential food and nutraceutical applications of alginate: a review. Mar Drugs 20(9):564. https://doi.org/10.3390/md20090564
Bokulich NA, Subramanian S, Faith JJ et al (2013) Quality-filtering vastly improves diversity estimates from Illumina amplicon sequencing. Nat Methods 10(1):57–59. https://doi.org/10.1038/nmeth.2276
Brunet M, de Bettignies F, Le Duff N et al (2021) Accumulation of detached kelp biomass in a subtidal temperate coastal ecosystem induces succession of epiphytic and sediment bacterial communities. Environ Microbiol 23(3):1638–1655. https://doi.org/10.1111/1462-2920.15389
Callahan BJ, McMurdie PJ, Rosen MJ et al (2016) DADA2: High-resolution sample inference from Illumina amplicon data. Nat Methods 13(7):581–583. https://doi.org/10.1038/nmeth.3869
Campbell AH, Harder T, Nielsen S et al (2011) Climate change and disease: bleaching of a chemically defended seaweed. Global Change Biol 17:2958–2970. https://doi.org/10.1111/j.1365-2486.2011.02456.x
Christie-Oleza JA, Sousoni D, Lloyd M et al (2017) Nutrient recycling facilitates long-term stability of marine microbial phototroph-heterotroph interactions. Nat Microbiol 2:17100. https://doi.org/10.1038/nmicrobiol.2017.100
Dai S, Zhou Y, Li N et al (2022) Why do red tides occur frequently in some oligotrophic waters? Analysis of red tide evolution history in Mirs Bay, China and its implications. Sci Total Environ 844:157112. https://doi.org/10.1016/j.scitotenv.2022.157112
Dudek M, Dieudonné A, Jouanneau D et al (2020) Regulation of alginate catabolism involves a GntR family repressor in the marine Flavobacterium Zobellia galactanivorans DsijT. Nucleic Acids Res 48(14):7786–7800. https://doi.org/10.1093/nar/gkaa533
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10(10):996–998. https://doi.org/10.1038/nmeth.2604
Egan S, Fernandes ND, Kumar V et al (2014) Bacterial pathogens, virulence mechanism and host defence in marine macroalgae. Environ Microbiol 16(4):925–938. https://doi.org/10.1111/1462-2920.12288
Enke TN, Datta MS, Schwartzman J et al (2019) Modular assembly of polysaccharide-degrading marine microbial communities. Curr Biol 29(9):1528–1535. https://doi.org/10.1016/j.cub.2019.03.047
FAO (2022) The state of world fisheries and aquaculture 2022 towards blue transformation. Rome. https://doi.org/10.4060/cc0461en
Fernández PA, Gaitán-Espitia JD, Leal PP et al (2020) Nitrogen sufficiency enhances thermal tolerance in habitat-forming kelp: implications for acclimation under thermal stress. Sci Rep 10(1):3186. https://doi.org/10.1038/s41598-020-60104-4
Gai C, La X, Diao J et al (2017) Establishment and utilization of quick retrieval table of kelp common disease. Chinese Fishery Quality and Standards. 7(5):51–55. www.fisheryqs.com/zgyyzlybz/article/abstract/2017102648223?st. Accessed 26 Oct 2017
Grice EA, Kong HH, Conlan S et al (2009) Topographical and temporal diversity of the human skin microbiome. Science 324(5931):1190–1192. https://doi.org/10.1126/science.1171700
Hudson J, Egan S (2022) Opportunistic diseases in marine eukaryotes: Could Bacteroidota be the next threat to ocean life? Environ Microbiol 24(10):4505–4518. https://doi.org/10.1111/1462-2920.16094
Kappelmann L, Krüger K, Hehemann JH et al (2019) Polysaccharide utilization loci of North Sea Flavobacteriia as basis for using SusC/D-protein expression for predicting major phytoplankton glycans. ISME J 13(1):76–91. https://doi.org/10.1038/s41396-018-0242-6
Kumar V, Zozaya-Valdes E, Kjelleberg S et al (2016) Multiple opportunistic pathogens can cause a bleaching disease in the red seaweed Delisea pulchra. Environ Microbiol 18(11):3962–3975. https://doi.org/10.1111/1462-2920.13403
Li X, Su L, Li X et al (2023) Comprehensive analysis of large-scale Saccharina japonica damage in the principal farming area of Rongcheng in Shandong province from 2021 to 2022. J Arg Sci Tech 25(1):206–222. https://doi.org/10.13304/j.nykjdb.2022.0728
Ling F, Egan S, Zhuang Y et al (2022) Epimicrobiome shifts with bleaching disease progression in the brown seaweed Saccharina japonica. Front Mar Sci 9:865224. https://doi.org/10.3389/fmars.2022.865224
MARA (2022) Ministry of Agriculture and Rural Affairs and Shandong Province actively responded to the major kelp disaster. Ministry of Agriculture and Rural Affairs of the People's Republic of China. http://www.moa.gov.cn/xw/zwdt/202201/t20220129_6388014.htm. Accessed 29 Jan 2022
Parks DH, Tyson GW, Hugenholtz P et al (2014) STAMP: statistical analysis of taxonomic and functional profiles. Bioinformatics 30(21):3123–3124. https://doi.org/10.1093/bioinformatics/btu494
Perini V, Bracken ME (2014) Nitrogen availability limits phosphorus uptake in an intertidal macroalga. Oecologia 175(2):667–676. https://doi.org/10.1007/s00442-014-2914-x
Piontek J, Meeske C, Hassenrück C et al (2022) Organic matter availability drives the spatial variation in the community composition and activity of Antarctic marine bacterioplankton. Environ Microbiol 24(9):4030–4048. https://doi.org/10.1111/1462-2920.16087
Qu T, Hou C, Zhao X et al (2021) Bacteria associated with Ulva prolifera: a vital role in green tide formation and migration. Harmful Algae 108:102104. https://doi.org/10.1016/j.hal.2021.102104
Quast C, Pruesse E, Yilmaz P et al (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:D590–D596. https://doi.org/10.1093/nar/gks1219
Serebryakova A, Aires T, Viard F et al (2018) Summer shifts of bacterial communities associated with the invasive brown seaweed Sargassum muticum are location and tissue dependent. PLoS ONE 13(12):e0206734. https://doi.org/10.1371/journal.pone.0206734
Seymour J, Amin S, Raina JB et al (2017) Zooming in on the phycosphere: the ecological interface for phytoplankton–bacteria relationships. Nat Microbiol 2:17065. https://doi.org/10.1038/nmicrobiol.2017.65
Spoerner M, Wichard T, Bachhuber T et al (2012) Growth and thallus morphogenesis of Ulva mutabilis (Chlorophyta) depends on a combination of two bacterial species excreting regulatory factors. J Phycol 48(6):1433–1447. https://doi.org/10.1111/j.1529-8817.2012.01231.x
Sunagawa S, DeSantis TZ, Piceno YM et al (2009) Bacterial diversity and white plague disease-associated community changes in the Caribbean coral Montastraea faveolata. ISME J 3(5):512–521. https://doi.org/10.1038/ismej.2008.131
Ward T, Larson J, Meulemans J et al (2017) BugBase predicts organism level microbiome phenotypes. bioRxiv 133462. https://doi.org/10.1101/133462
Zhang N, Song C, Wang M et al (2017) Diversity and characterization of bacteria associated with the deep-sea hydrothermal vent crab Austinograea sp. comparing with those of two shallow-water crabs by 16S ribosomal DNA analysis. PloS One 12(11):e0187842. https://doi.org/10.1371/journal.pone.0187842
Zhang R, Chang L, Xiao L et al (2020) Diversity of the epiphytic bacterial communities associated with commercially cultivated healthy and diseased Saccharina japonica during the harvest season. J Appl Phycol 32:2071–2080. https://doi.org/10.1007/s10811-019-02025-y
Funding
This study was supported by National Natural Science Foundation of China (42206131).
Author information
Authors and Affiliations
Contributions
Chen Ma: conceptualization, methodology, formal analysis, data curation, writing—original draft. Chengxiang Peng, Longwen Fu, Chenggang Ren, and Xintian Liu: resources, methodology, formal analysis, data curation. Zhihai Zhong, Zhengyi Liu, and Song Qin: conceptualization, supervision, writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: V.V.S.S. Sarma
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, C., Peng, C., Fu, L. et al. Phycosphere bacterial disturbance of Saccharina japonica caused by white rot disease relates to seawater nutrients. Environ Sci Pollut Res (2024). https://doi.org/10.1007/s11356-024-33707-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-024-33707-x